- Home
- »
- Medical Devices
- »
-
Australia Custom Procedure Kits Market Size Report, 2033GVR Report cover
![Australia Custom Procedure Kits Market Size, Share & Trends Report]()
Australia Custom Procedure Kits Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Disposable, Reusable), By Procedure (Colorectal, Thoracic, Orthopedic, Ophthalmology), By End Use (Hospitals, Clinics), And Segment Forecasts
- Report ID: GVR-4-68040-821-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Australia Custom Procedure Kits Market Summary
The Australia custom procedure kits market size was estimated at USD 165.33 million in 2024 and is projected to reach USD 490.46 million by 2033, growing at a CAGR of 12.84% from 2025 to 2033. This growth is attributed to the growing need for greater efficiency, safety, and cost optimization across surgical and interventional workflows.
Key Market Trend and Insights
- By product, the disposable segment led the market with the largest revenue share in 2024.
- By procedure kits, the orthopedic segment led the market with the largest revenue share in 2024.
- By end use, the hospitals segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 165.33 Million
- 2033 Projected Market Size: USD 490.46 Million
- CAGR (2025-2033): 12.84%
As hospitals and ambulatory surgical centers face increasing patient volumes and rising procedural complexity, pre-configured sterile kits tailored to specific surgeries enable faster operating room turnover, reduced infection risk, and fewer supply management errors. In addition, the shift toward value-based care has led facilities to prioritize solutions that lower labor requirements and eliminate inventory waste, further strengthening demand for procedure-ready custom packs. The Australia custom procedure kit industry is primarily driven by the healthcare sector’s strong focus on improving procedural efficiency and cost optimization. Hospitals and day-surgery facilities are increasingly opting for customized sterile procedure kits, as they minimize preparation time, reduce the need for manual gathering of individual consumables, and streamline surgical workflows. This results in faster operating room turnover, lower labor burden, and reduced inventory and procurement complexity, all of which align with the national push toward higher operational productivity in healthcare delivery.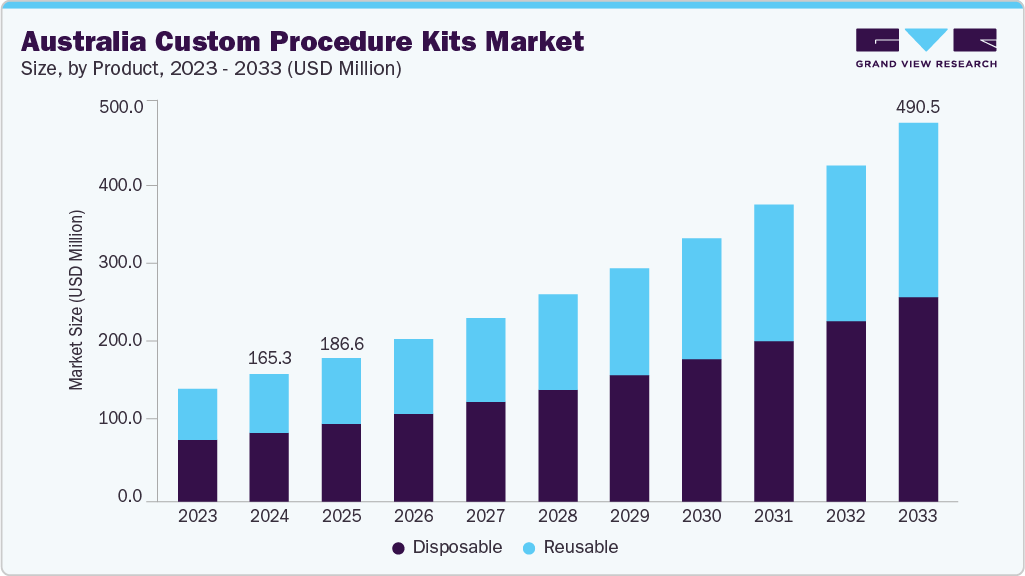
Another key driver is the increasing number of surgical and minimally invasive procedures, fueled by an aging population, the prevalence of chronic diseases, and the expansion of specialty treatments in orthopedics, cardiovascular care, wound therapy, and gynecology. Custom procedure kits offer pre-sterilized components specifically designed for various procedures, enabling busier surgical teams to operate more efficiently and maintain consistency across treatments. As private surgical centers continue to expand across Australia, the demand for ready-to-use kits that enhance standardization and minimize the risk of human error is increasing. For Instance, private hospitals in Australia play a vital role in national healthcare, accounting for around 70% of all planned surgeries, which results in approximately 1.83 million procedures annually. They deliver state-of-the-art treatment using advanced technologies while easing the burden on the public hospital system. Private facilities perform the majority of life-saving and life-enhancing procedures that Australians require throughout their lives.
Surgery Statistics Performed in Private Hospitals (Australia)
Procedure Category
% Performed in Private Hospitals
Obesity & Overweight Procedures
95%
Skin Grafts
77%
Coronary Procedures
76%
Knee Replacements
75%
Hip Replacements
74%
Eye Disorder Procedures
72%
Malignant Skin Cancers
69%
Carpal Tunnel Syndrome
66%
Spinal Disorders
65%
Hernias
63%
Malignant Breast Cancers
53%
Source: Australian Institute of Health and Welfare, 2025
In addition, the market is gaining momentum due to increasing emphasis on infection control and patient safety, driven by hospital-acquired infection (HAI) awareness and evolving regulatory standards. Single-use procedure packs not only reduce contamination risks compared to reusable product handling but also support compliance with sterilization and traceability requirements. Workforce shortages, particularly among nurses, further reinforce the adoption of custom kits, which help eliminate manual preparation steps and free clinical staff to focus on patient care. Collectively, these factors continue to strengthen market demand across both public and private healthcare segments in Australia.
Market Concentration & Characteristics
The degree of innovation in the Australian market is high, driven by strong demand for workflow efficiency, infection-control safety, and customization in surgical and diagnostic environments. Manufacturers are increasingly incorporating procedure-specific configurations, antimicrobial and eco-friendly materials, RFID-enabled tracking, and ergonomically designed single-use instruments to reduce operating room time and enhance sterility assurance. Digital collaboration with clinicians for kit design and real-time supply analytics has further accelerated innovation, enabling faster turnaround and reduced procurement complexity for hospitals and day surgery centers.
The level of M&A activity in the market is moderate but steadily increasing, driven by the need to expand surgical product portfolios, strengthen hospital networks, and improve supply chain scale. Established global players and regional manufacturers are pursuing acquisitions and strategic partnerships to enhance customization capabilities, integrate sterilization services, and secure long-term contracts with private and public healthcare facilities. Consolidation is also influenced by the rising dominance of single-vendor procurement models, which encourages players to acquire niche suppliers to prevent market fragmentation and maintain competitive leverage.

Regulatory frameworks in Australia have a significant and positive influence on the market, primarily through the strict oversight of the Therapeutic Goods Administration (TGA), which governs sterility standards, material safety, infection control compliance, and the traceability of surgical consumables. While these regulations increase the operational and documentation burden on manufacturers, they enhance market credibility and drive the adoption of high-quality, pre-configured kits across hospitals and day surgery centers. Requirements for validated sterilization processes, clinical evidence of component compatibility, and mandatory reporting of adverse events encourage vendors to invest in quality assurance and technological upgrades.
Substitutes for custom procedure kits in Australia primarily include individually sourced sterile components and generic standard surgical packs, which allow facilities to assemble consumables based on clinician preference rather than pre-configured sets. While these substitutes can appear cost-effective in low-volume or highly variable procedural settings, they introduce drawbacks such as longer preparation time, greater risk of missing items, increased inventory complexity, and higher procedural waste. Some hospitals also rely on reusable instrument trays paired with separately purchased disposables as an alternative, though this model faces rising pressure due to infection-control compliance requirements and reprocessing costs.
Product Insights
The disposable segment held the largest share of the Australian market in 2024. This dominance is driven by hospitals increasing focus on infection prevention, sterility assurance, and workflow efficiency in perioperative care. Rising surgical volumes, particularly in orthopedics, cardiovascular, and general surgery, have accelerated demand for single-use consumables that reduce the risk of cross-contamination and eliminate the time and cost associated with reprocessing reusable items. In addition, the surge in day-surgery centers and private hospitals adopting “ready-to-use” procedural packs further boosted disposable kit utilization, as they help minimize turnaround time between procedures, support standardization of surgical workflows, and ensure greater supply consistency.
Reusable are expected to grow at the fastest CAGR over the forecast period due to the healthcare sector’s increasing focus on long-term cost efficiency and sustainability. While disposable kits continue to dominate high-volume surgical settings, hospitals and surgical centers are increasingly adopting reusable instruments and components to reduce medical waste generation and lower recurring procurement costs, especially for procedures requiring high-value surgical tools that can withstand multiple sterilization cycles. Advances in sterilization technologies and durable biocompatible materials have further strengthened the viability of reusable kits, offering improved performance, extended life span, and better return on investment.
Procedure Insights
The orthopedic segment dominated the Australian market in 2024 due to the high volume of orthopedic surgeries performed across the country and the growing aging population prone to degenerative musculoskeletal disorders. Procedures such as knee and hip replacements, spinal surgeries, and fracture repairs require highly standardized instrumentation and implants, making custom procedure kits essential for achieving surgical precision, controlling infections, and ensuring operational efficiency.
Ophthalmology is expected to grow at a significant CAGR over the forecast period, driven by the surge in cataract, glaucoma, and retinal surgeries associated with an aging population and increasing prevalence of vision-related disorders. Ophthalmic procedures require high-precision, sterile, and micron-level surgical tools, making custom kits highly valuable for ensuring accuracy, safety, and reduced operating room setup time. Growing adoption of day-surgery models in private eye hospitals and clinics, combined with demand for infection-control compliance and faster patient turnover, further accelerates kit utilization.
End Use Insights
The hospitals segment held the largest share of the market in 2024. This dominance is attributed due to their high surgical volumes, wide adoption of standardized protocols, and demand for infection-controlled, procedure-ready supplies to support operational efficiency. Hospitals increasingly rely on custom procedure kits to minimize operating room preparation time, reduce labor costs, and ensure consistency across complex and multidisciplinary surgeries, including orthopedics, cardiac, ophthalmology, and general surgery.
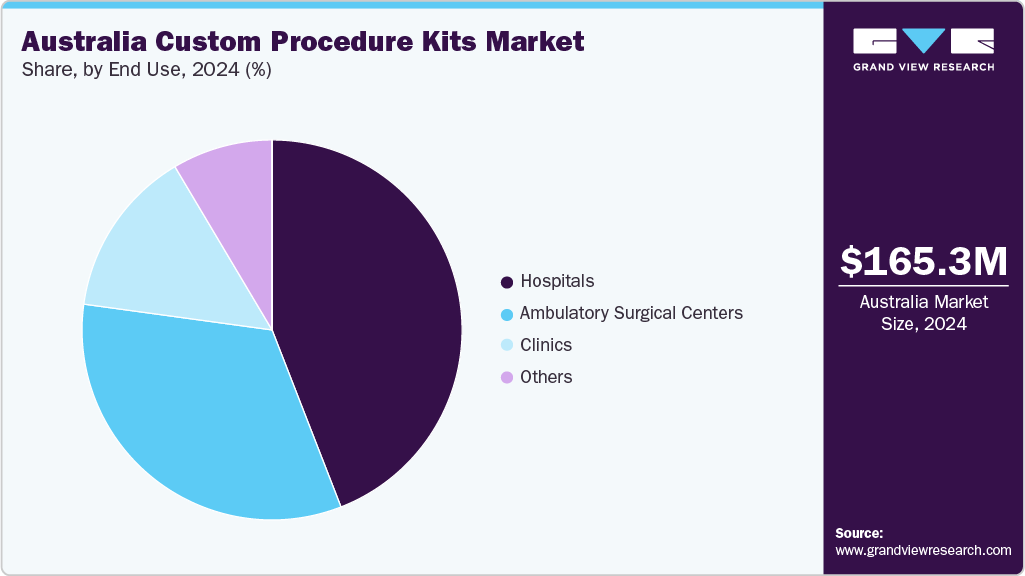
Ambulatory surgical centers are expected to grow at the fastest CAGR over the forecast period, due to the rapid shift of elective and minimally invasive surgeries from hospitals to outpatient settings. ASCs prioritize fast turnaround times, cost efficiency, and streamlined workflows, making custom procedure kits highly attractive for reducing setup time, minimizing instrument variability, and supporting predictable scheduling. Growing patient preference for shorter hospital stays, expanding reimbursement support for day surgeries, and increasing investment in specialized standalone centers are further accelerating kit adoption in ASCs, positioning them as the fastest-expanding end use segment in the market.
Key Australia Custom Procedure Kits Company Insights
The industry players are undertaking several strategic initiatives such as acquisitions, partnerships and collaborations. Moreover, the launch of novel products is anticipated to boost the competitive rivalry in the Australia custom procedure kits industry.
Key Australia Custom Procedure Kits Companies:
- Mölnlycke Health Care AB
- Medline (Medline International Two Australia Pty Ltd)
- Teleflex Incorporated
- Medtronic
- Cardinal Health
- ICU Medical, Inc.
- Terumo
- Santex S.p.A.
Australia Custom Procedure Kits Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 186.64 million
Revenue forecast in 2033
USD 490.46 million
Growth Rate
CAGR of 12.84% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Segments covered
Product, procedure, end use
Regional scope
Australia
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
Mölnlycke Health Care AB; Medline (Medline International Two Australia Pty Ltd); Teleflex Incorporated; Medtronic; Cardinal Health; ICU Medical, Inc.; Terumo; Santex S.p.A.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Australia Custom Procedure Kits Market Report Segmentation
This report forecasts revenue growth at a country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Australia custom procedure kits market report based on product, procedure, and end use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Disposable
-
Reusable
-
-
Procedure Outlook (Revenue, USD Million, 2021 - 2033)
-
Colorectal
-
Thoracic
-
Orthopedic
-
Ophthalmology
-
Neurosurgery
-
Cardiac Surgery
-
Gynecology
-
General Surgery
-
Urology
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers
-
Clinics
-
Others
-
Frequently Asked Questions About This Report
b. Some key players operating in the Australia custom procedure kits market include Mölnlycke Health Care AB; Medline (Medline International Two Australia Pty Ltd); Teleflex Incorporated; Medtronic; Cardinal Health; ICU Medical, Inc.; Terumo; Santex S.p.A.
b. Key factors that are driving the market growth include the growing need for greater efficiency, safety, and cost optimization across surgical and interventional workflows. As hospitals and ambulatory surgical centers face increasing patient volumes and rising procedural complexity, pre-configured sterile kits tailored to specific surgeries enable faster operating room turnover, reduced infection risk, and fewer supply management errors.
b. The Australia custom procedure kits market size was estimated at USD 165.33 million in 2024 and is expected to reach USD 186.64 million in 2025.
b. The Australia custom procedure kits market is expected to grow at a compound annual growth rate of 12.84% from 2025 to 2033 to reach USD 490.46 million by 2033.
b. Disposable segment dominated the Australia custom procedure kits market with a share of 54.53% in 2024. This dominance is driven by hospitals increasing focus on infection prevention, sterility assurance, and workflow efficiency in perioperative care.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










