- Home
- »
- Biotechnology
- »
-
CAR T-cell Therapy Market Size, Industry Report, 2033GVR Report cover
![CAR T-cell Therapy Market Size, Share & Trends Report]()
CAR T-cell Therapy Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Abecma, Breyanzi, Carvykti, Kymriah), By Disease Indication (Lymphoma, Leukemia, Multiple Myeloma), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-090-4
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
CAR T-cell Therapy Market Summary
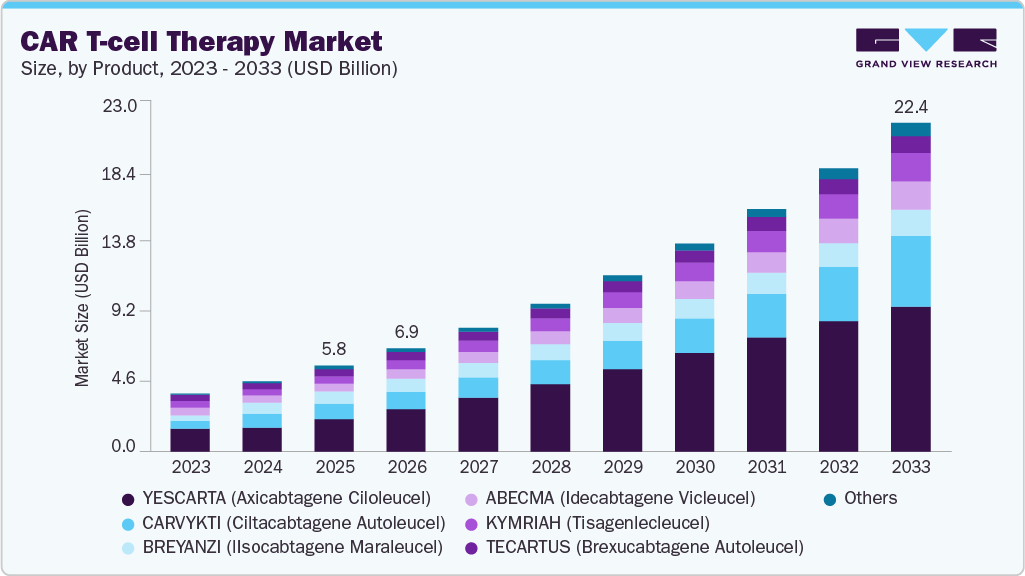
The global CAR T-cell therapy market size was estimated at USD 5.82 billion in 2025 and is projected to reach USD 22.36 billion by 2033, growing at a CAGR of 18.06% from 2026 to 2033. Market growth is driven by rising cancer prevalence, adoption of personalized cell therapies, advances in gene editing, and supportive regulations.
Key Market Trends & Insights
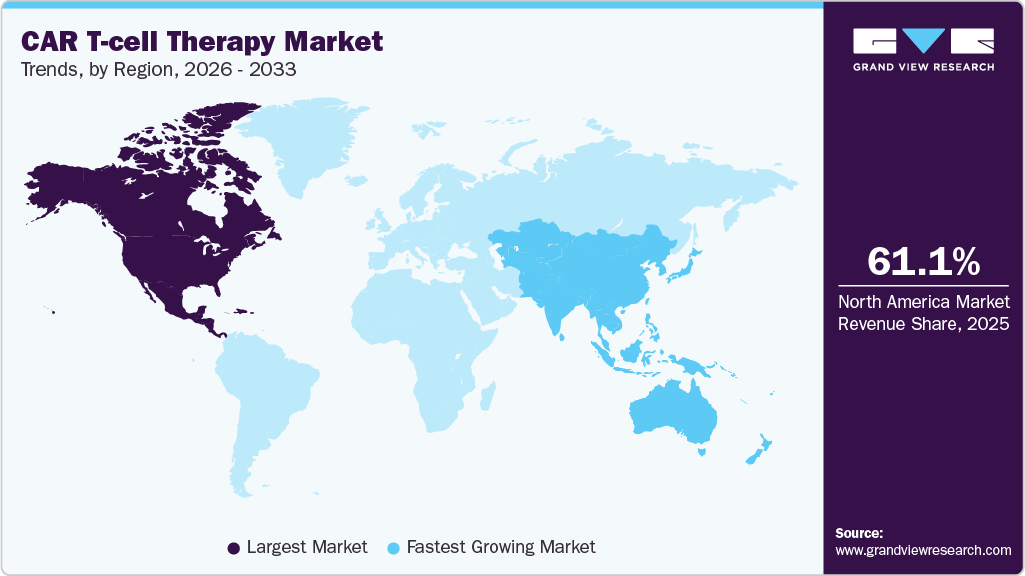
- North America dominated the global CAR T-cell therapy market with the largest revenue share of 61.14% in 2025.
- The CAR T-cell therapy industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product, the yescarta segment led the market with the largest revenue share of 37.74% in 2025.
- By disease indication, the lymphoma segment accounted for the largest market revenue share in 2025.
- By end use, the hospital segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 5.82 Billion
- 2033 Projected Market Size: USD 22.36 Billion
- CAGR (2026-2033): 18.06%
- North America: Largest market share in 2025
- Asia Pacific: Fastest growing market
Rising Cancer Burden
The rising prevalence of cancer, especially blood cancers like leukemia, lymphoma, and multiple myeloma, is driving the CAR T-cell therapy industry. Traditional treatments often fail to achieve long-term remission, while CAR T-cell therapies, which engineer a patient’s T-cells to target cancer, offer higher efficacy and durable responses. Increasing global cancer incidence is boosting demand for these advanced, personalized treatments.
Key U.S. Cancer Incidence, Mortality, and Survival Statistics (2026 Projection)
Category
Statistic
Projected new cancer cases in the U.S. (2026)
2,114,850
Projected cancer deaths in the U.S. (2026)
626,140
Overall 5-year relative survival (2015-2021)
70%
Leading cause of cancer deaths (2026)
Lung cancer (more than colorectal + pancreatic combined)
Threats to continued progress
Proposed federal cuts to cancer research and health insurance access
Source: American Cancer Society, Secondary Research, Grand View Research
Limited options for aggressive or treatment-resistant blood cancers support CAR T-cell market growth. These therapies address critical unmet needs, and rising awareness among healthcare professionals and patients is driving adoption. Expanding clinical trials into other cancers and solid tumors is expected to increase demand further, making CAR T-cell therapy a central focus in oncology.
Personalized Treatment Advantage
The personalized nature of CAR T-cell therapy is a major market driver, shifting oncology away from conventional treatments. By engineering a patient’s T-cells to target specific cancer antigens, these therapies offer higher efficacy and durable responses, particularly in relapsed or refractory blood cancers. The expanding adoption of precision medicine is further accelerating demand for targeted, cell-based therapies.
Advances in genomics, biomarker discovery, and cell engineering further support demand for CAR T-cell therapies. Personalized approaches reduce off-target toxicity and improve safety and quality of life compared to systemic treatments. As clinicians and payers recognize the long-term value of durable responses and lower relapse rates, CAR T-cell therapy adoption is expected to sustain strong market growth.
Market Concentration & Characteristics
The CAR T-cell therapy industry exhibits a high degree of innovation, supported by advances in genetic engineering, next-generation CAR designs, and improved manufacturing technologies that enhance safety, efficacy, and scalability.
The CAR T-cell therapy industry shows moderate to high M&A activity, as biopharmaceutical companies pursue acquisitions and partnerships to access innovative technologies, manufacturing capabilities, and oncology pipelines.
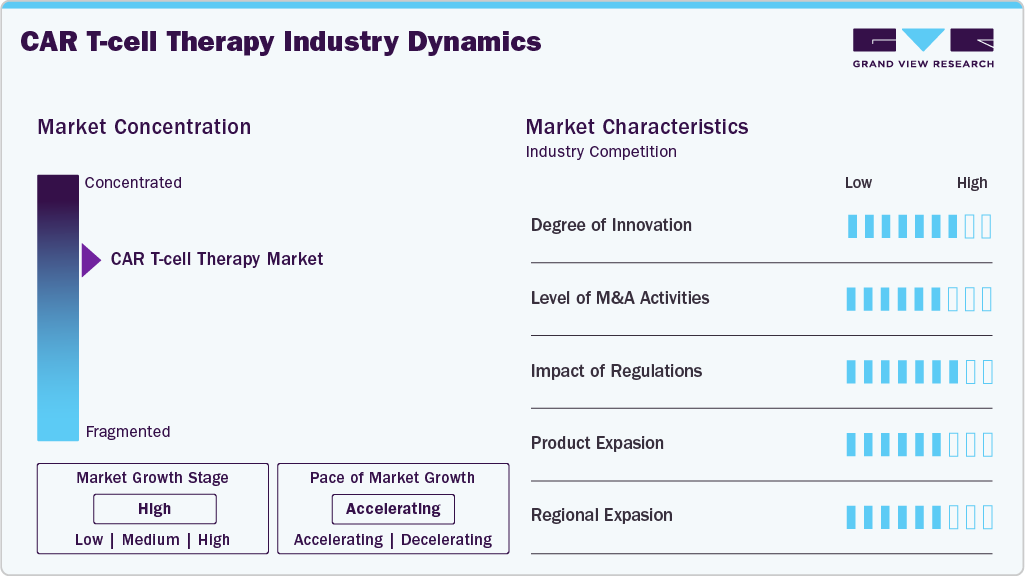
The CAR T-cell therapy industry is largely governed by regulations that impose stringent preconditions for clinical development, production, and marketing. Nonetheless, in convergence with such measures, the fast-track approvals, orphan drug statuses, and favorable reimbursement practices in major countries are on the plus side, helping to achieve quicker market access and wider use while preserving safety and efficacy measures.
Next-generation therapies for new indications drive product expansion in the CAR T-cell therapy industry. Companies are broadening portfolios through pipeline diversification, label extensions, and allogeneic or off-the-shelf CAR T-cell products to enhance accessibility and reach.
The CAR T-cell therapy industry regional expansion is mainly due to the increasing number of regulatory approvals, better healthcare infrastructure, and funding in cell and gene therapy. Firms are moving their operations beyond North America and Europe into Asia-Pacific and other fast-growing areas through partnerships, local manufacturing, and clinical trials.
Product Insights
The yescarta segment led the market with the largest revenue share of 37.74% in 2025. , Yescarta also known as axicabtagene ciloleucel, targets the CD19 antigen and is primarily used for relapsed or refractory large B-cell and follicular lymphoma, with a strong market share driven by improved survival rates, favorable clinical outcomes, and increasing adoption across oncology centers. Its robust efficacy profile, coupled with ongoing research in additional hematologic malignancies, further reinforces its leadership in the CAR T-cell therapy landscape.
The carvykti segment is expected to witness at the fastest CAGR during the forecast period, driven by its effectiveness in treating multiple myeloma, favorable clinical outcomes, and increasing adoption of novel CAR T-cell therapies in hematologic cancers. In addition, ongoing clinical trials exploring combination therapies and expanding indications are expected to boost its market penetration further and strengthen its competitive position.
Disease Indication Insights
The lymphoma segment led the market with the largest revenue share of 52.86% in 2025 and is expected to grow at the fastest CAGR during the forecast period, driven by the high prevalence of B-cell lymphomas and strong clinical efficacy of CAR T-cell therapies in relapsed or refractory cases.
The leukemia segment is expected to grow at the fastest CAGR during the forecast period, driven by rising prevalence, advancements in CAR T-cell therapies, favorable clinical outcomes, increasing adoption of novel immunotherapies, and ongoing research into next-generation treatment options.
End Use Insights
The hospital segment accounted for the largest market revenue share in 2025, driven by advanced facilities, strong purchasing power, and the ability to perform in-house CAR T-cell procedures, which help reduce overall therapy costs.
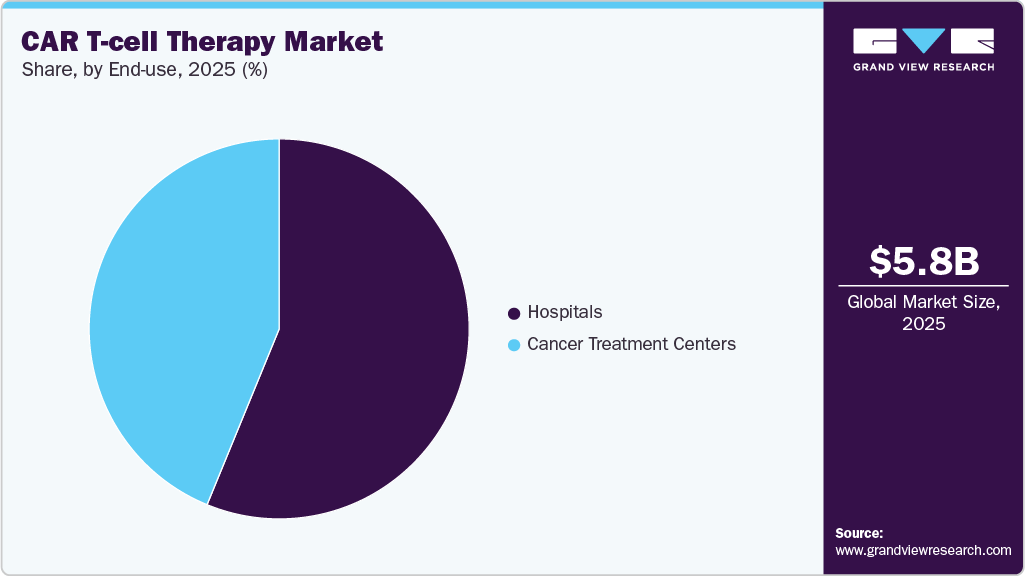
The cancer treatment centers segment is expected to register at the fastest CAGR from 2026 to 2033, primarily due to increasing patient access, specialized oncology services, growing adoption of CAR T-cell therapies in dedicated treatment facilities, and rising investments in advanced cancer care infrastructure.
Regional Insights
North America dominated the global CAR T-cell therapy market with the largest revenue share of 61.14% in 2025, driven by high adoption of advanced therapies, well-established healthcare infrastructure, strong R&D investment, and early regulatory approvals for CAR T-cell therapies. The region’s strong player presence, rising hematologic cancer cases, physician and patient awareness, and favorable reimbursement policies support sustained market growth.
U.S. CAR T-cell Therapy Market Trends
The CAR T-cell therapy market in the U.S. is experiencing significant growth. The growing prevalence rate of various diseases, including lymphoma and multiple myeloma, and the increasing geriatric population are driving market growth. In addition, continuous technological advancements in diagnostics and therapeutics, along with improvements in healthcare infrastructure, are further contributing to the expansion of the cancer treatment landscape.
Europe CAR T-cell Therapy Market Trends
The CAR T-cell therapy market in Europe is expected to grow at a significant CAGR during the forecast period, driven by an aging population, rising cancer prevalence, early diagnosis, adoption of personalized medicine, and strong healthcare infrastructure.
The UK CAR T-cell therapy market is expected to witness growth over the forecast period, driven by increased investments from major companies and governments, including the NHS & research laboratories.
The CAR T-cell therapy market in Germany held a significant share in 2025, driven by strong clinical infrastructure, government-backed centers, strategic pharma-academic collaborations, supportive reimbursement, and rising blood cancer cases.
Asia Pacific CAR T-cell Therapy Market Trends
The CAR T-cell therapy market in the Asia Pacific is projected to expand at the fastest CAGR of 22.35% over the forecast period, driven by a rising patient population, supportive regulatory frameworks, and increasing cancer prevalence. For instance, in December 2025, India launched its first indigenous CAR-T cell therapy, NexCAR19, developed by ImmunoACT, targeting blood cancers and demonstrating the country’s advancing capacity in cell and gene therapy.
The China CAR T-cell therapy market continues to dominate the regional landscape, supported by a large patient pool, strong government support, advanced healthcare infrastructure, and growing investment in CAR T-cell therapy development.
The CAR T-cell therapy market in Japan is growing rapidly, supported by streamlined regulatory pathways, active clinical trials, industry-academic collaborations, and initiatives to enhance patient access to cutting-edge treatments.
Key CAR T-cell Therapy Company Insights
The CAR T-cell therapy industry is dominated by major players such as Bristol-Myers Squibb Company, Novartis AG, Gilead Sciences, Inc., Johnson & Johnson Services, Inc., JW Therapeutics (Shanghai) Co., Ltd., bluebird bio, Inc., Merck & Co., Inc., Sangamo Therapeutics, Sorrento Therapeutics, Inc., and GSK plc.
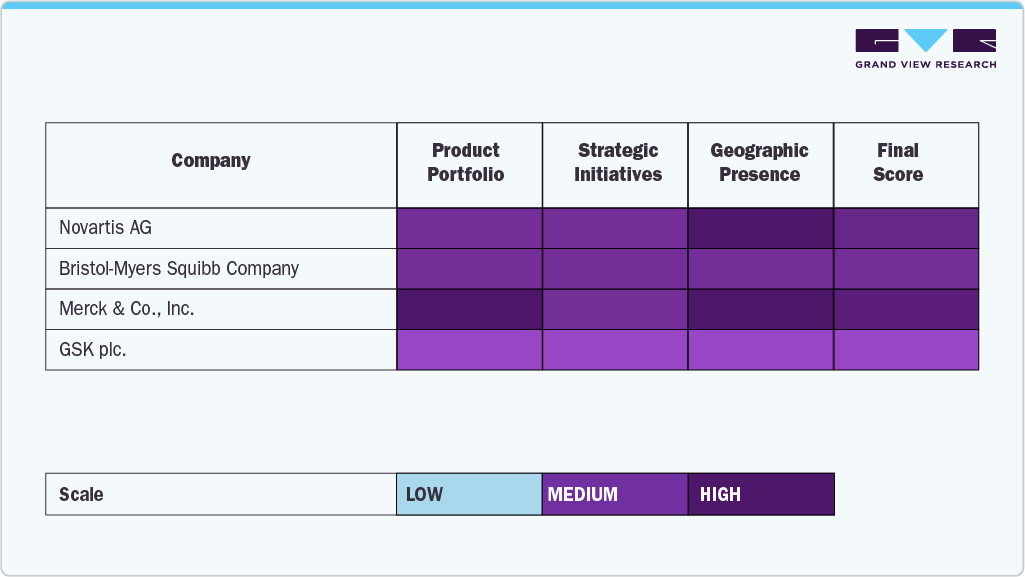
Major players are continually expanding their portfolios through innovative CAR T-cell therapies, clinical-stage assets, and partnerships. Firms that combine the latest tech with the patient-first method will not only pull the market in their direction but also determine its course.
Key CAR T-cell Therapy Companies:
The following key companies have been profiled for this study on the CAR T-cell therapy market.
- Bristol-Myers Squibb Company
- Novartis AG
- Gilead Sciences, Inc.
- Johnson & Johnson Services, Inc.
- JW Therapeutics (Shanghai) Co., Ltd.
- bluebird bio, Inc.
- Merck & Co., Inc.
- Sangamo Therapeutics
- Sorrento Therapeutics, Inc.
- GSK plc.
Recent Developments
-
In January 2026, Australia-based AdAlta and SHcell launched a collaboration to develop and commercialize the CAR‑T therapy BZDS1901, targeting solid tumors, with manufacturing and Phase 1 trials planned in Australia.
-
In December 2025, the U.S. FDA approved Bristol Myers Squibb’s Breyanzi as the first CAR T-cell therapy for adults with relapsed or refractory marginal zone lymphoma, demonstrating high efficacy and manageable safety.
-
In April 2025, Johnson & Johnson announced that the European Commission approved an expanded indication for Darzalex (daratumumab) subcutaneous formulation in combination with bortezomib, lenalidomide, and dexamethasone for the treatment of newly diagnosed multiple myeloma (NDMM) patients, now including those
CAR T-cell Therapy Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 6.99 billion
Revenue forecast in 2033
USD 22.36 billion
Growth rate
CAGR of 18.06% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, disease indication, end use, region
Regional scope
North America; Europe; Asia Pacific; Rest of World
Country scope
U.S.; Canada; UK; Germany; Japan; China.
Key companies profiled
Bristol-Myers Squibb Company; Novartis AG; Gilead Sciences, Inc.; Johnson & Johnson Services, Inc.; JW Therapeutics (Shanghai) Co., Ltd.; bluebird bio, Inc.; Merck & Co., Inc.; Sangamo Therapeutics; Sorrento Therapeutics, Inc.; GSK plc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
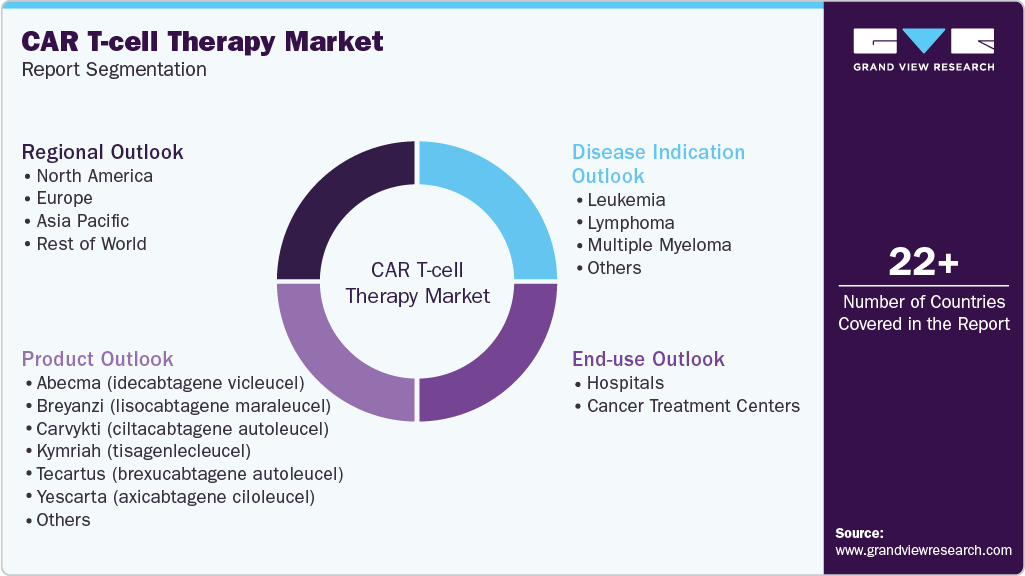
Global CAR T-cell Therapy Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global CAR T-cell therapy market report based on product, disease indication, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Abecma (idecabtagene vicleucel)
-
Breyanzi (lisocabtagene maraleucel)
-
Carvykti (ciltacabtagene autoleucel)
-
Kymriah (tisagenlecleucel)
-
Tecartus (brexucabtagene autoleucel)
-
Yescarta (axicabtagene ciloleucel)
-
Others
-
-
Disease Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Leukemia
-
Lymphoma
-
Multiple Myeloma
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Cancer Treatment Centers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
Germany
-
UK
-
-
Asia Pacific
-
Japan
-
China
-
-
Rest of World
-
Frequently Asked Questions About This Report
b. The global CAR T-cell therapy market size was valued at USD 5.82 billion in 2025 and is expected to reach USD 6.99 billion in 2026.
b. The rising prevalence of cancer such as multiple myeloma across the world and the rampant regulatory support in the form of product approvals and an intensive product pipeline of drugs are some of the key factors driving the growth of the market.
b. The lymphoma segment dominated the CAR T-cell therapy market with 52.86% in 2025 and is expected to grow at the fastest rate during the forecast period, driven by the high prevalence of B-cell lymphomas and strong clinical efficacy of CAR T-cell therapies in relapsed or refractory cases.
b. Some of the key players in the global CAR T-cell therapies market include Bristol-Myers Squibb Company; Novartis AG; Gilead Sciences, Inc.; Johnson & Johnson Services, Inc.; JW Therapeutics (Shanghai) Co., Ltd.; bluebird bio, Inc.; Merck & Co., Inc.; Sangamo Therapeutics; Sorrento Therapeutics, Inc.; GSK plc.
b. The global CAR T-cell therapy market is expected to grow at a compound annual growth rate of 18.06% from 2026 to 2033 to reach USD 22.36 billion by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










