- Home
- »
- Clinical Diagnostics
- »
-
Cardiac Biomarkers Market Size, Industry Report, 2033GVR Report cover
![Cardiac Biomarkers Market Size, Share & Trends Report]()
Cardiac Biomarkers Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Troponin, Creatine kinase - MB (CK-MB), Myoglobin, BNP and NT-proBNP), By Application (Acute Coronary Syndrome, Myocardial Infarction, Congestive Heart Failure), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-1-68038-083-5
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Cardiac Biomarkers Market Summary
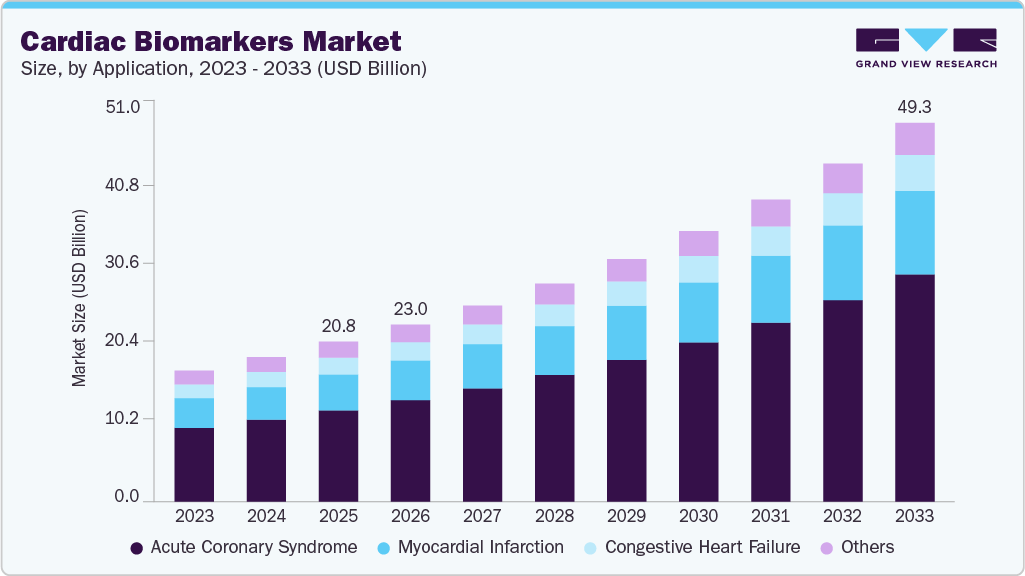
The global cardiac biomarkers market size was estimated at USD 20.81 billion in 2025 and is expected to reach USD 49.31 billion by 2033, growing at a CAGR of 11.5% from 2026 to 2033. The market is driven by the rising prevalence of cardiovascular diseases such as acute coronary syndrome, myocardial infarction, and heart failure, increasing the demand for early and accurate diagnosis.
Key Market Trends & Insights
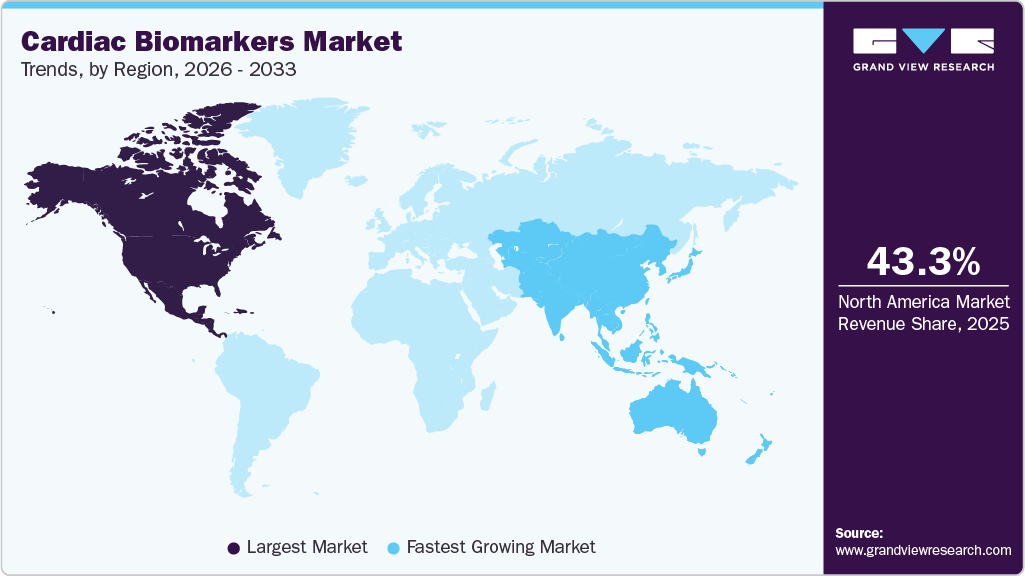
- North America dominated the cardiac biomarkers market and held the largest revenue share of 43.32% in 2025.
- By type, the troponin segment held the highest market share of 35.82% in 2025.
- By application, the acute coronary syndrome segment held the highest market share of 56.62% in 2025.
- By end use, the laboratory testing segment held the highest market share of 63.90% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 20.81 Billion
- 2033 Projected Market Size: USD 49.31 Billion
- CAGR (2026-2033): 11.5%
- North America: Largest market in 2025
Advancements in high-sensitivity troponin assays and multi-marker testing have improved clinical decision-making and patient outcomes. In addition, expanding use of point-of-care testing, supportive reimbursement frameworks, and an aging population with higher cardiac risk continue to support market expansion globally.Cardiovascular diseases are one of the major causes of death globally. According to the World Health Organization (WHO) survey in 2023, it was stated that approximately 17.9 million deaths due to cardiac disorders were reported each year. The prevalence of these conditions continues to rise due to an aging population, obesity, diabetes, and hypertension, leading to an increasing number of individuals at risk of heart attack or heart failure. The growing burden of cardiovascular diseases has created a critical need for early detection, accurate diagnosis, and effective patient monitoring. This is where cardiac biomarkers play a vital role. Biomarkers such as troponin, Creatine Kinase-MB (CK-MB), and myoglobin are essential for detecting myocardial injury and assessing disease severity and therefore help in making treatment decisions.
The rising demand for point-of-care (POC) cardiac testing kits is further expected to propel the growth in the cardiac biomarkers Market. Traditionally, the diagnosis of cardiovascular diseases relied on centralized laboratory testing, which requires specialized instruments and trained personnel. These conventional systems can delay diagnosis and treatment, particularly in acute settings such as emergency department admissions for chest pain or suspected myocardial infarction. Such delays can have serious clinical consequences, as timely identification of cardiac events is critical for initiating interventions and improving patient survival rates. POC cardiac testing kits enable quick diagnostic results for making timely decisions directly at the site of patient care, including emergency rooms, cardiology clinics, and in-home-based settings. Key biomarkers such as troponin, natriuretic peptide (BNP), and creatine kinase-MB (CK-MB) are now measurable at the point of care, allowing clinicians to make faster treatment decisions. For instance, POC cTn tests typically provide results within 10 to 20 minutes.
Emerging biomarkers, including soluble suppression of tumorigenicity-2 (sST2), heart-type fatty acid-binding protein (H-FABP), Galectin-3, and growth differentiation factor-15 (GDF-15), are gaining attention for their potential clinical utility. For instance, a study published by MDPI in August 2025 reported that the biomarkers GDF-15, sST2, suPAR, and H-FABP demonstrated promising potential across a range of cardiovascular diseases.
These novel biomarkers not only reflect different aspects of cardiovascular physiology but also offer the potential to improve early diagnosis and monitoring of treatment response. Advances in technology, including high-sensitivity assays and nanotechnology-based sensors, have further enhanced the ability to detect these biomarkers at very low concentrations, enabling faster and more accurate clinical decision-making. In addition, combining multiple biomarkers into panels allows clinicians to gain a more comprehensive understanding of a patient’s cardiovascular status, moving toward more personalized and precise approaches to care. As research continues and more evidence emerges, these novel cardiac biomarkers are expected to play an important role in improving outcomes for patients with heart disease.
Market Concentration & Characteristics
The degree of innovation in the market is high, as multi-biomarker panels represent a significant opportunity for innovation. They allow diagnostic companies to offer advanced, comprehensive solutions that go beyond conventional single-marker assays. For instance, in January 2025, the U.S. Food and Drug Administration cleared Abbott’s Alere NT-proBNP assay for the Alinity i system for aiding heart failure diagnosis, enhancing laboratory workflow efficiency and expanding cardiac testing menus in many hospital labs across the country. Furthermore, he development of high-sensitivity cardiac troponin (hs-cTn) tests has significantly improved early detection of myocardial infarction, enabling faster clinical decision-making in emergency settings.
The level of mergers and acquisitions (M&A) activities in market is medium. While there have been some strategic acquisitions by major diagnostic companies to strengthen their product portfolios and expand market reach, for instance, in 2025, bioMérieux agreed to acquire SpinChip Diagnostics ASA, a company developing a rapid point-of-care immunoassay platform focused on high-sensitivity cardiac troponin I, NT-proBNP and D-dimer assays, with first products expected in 2026. These strategic moves help companies accelerate product development timelines and reinforce competitive positioning without significantly disrupting overall market structure.

The impact of regulation on the cardiac biomarkers market is high, as products must comply with stringent clinical validation, quality assurance, and regulatory approval standards. Regulatory agencies require robust evidence of sensitivity, specificity, and clinical utility, particularly for high-sensitivity troponin assays used in acute cardiac care. Compliance with evolving in vitro diagnostic (IVD) regulations, post-market surveillance requirements, and quality management systems adds complexity to product launches.
Product expansion remains a key growth factor, with manufacturers increasingly launching cardiac biomarkers into broader applications such as in September 2025, Roche announced primary results from its TSIX Study Program showing that the new Elecsys Troponin T hs Gen 6 assay (sixth-generation high-sensitivity troponin T test) demonstrated high precision in identifying acute myocardial infarction and reliable exclusion of non-AMI cases, following its recent CE Mark approval. Whereas multiplex panels combining cardiac markers with inflammatory or metabolic biomarkers are gaining attention for comprehensive cardiovascular risk profiling. This expansion reduces dependency on single-test applications and enhances the clinical value proposition of biomarker platforms across diverse care settings.
Regional expansion in the cardiac biomarkers market is accelerating as emerging economies invest in improving access to decentralized and rapid diagnostic services. Growth in Asia Pacific, Latin America, and parts of the Middle East is supported by rising cardiovascular disease prevalence, expanding hospital infrastructure, and increased healthcare spending. The adoption of point-of-care testing solutions in secondary and rural healthcare facilities is particularly strong, enabling faster diagnosis in resource-limited settings.
Type Insights
The troponin segment dominated the cardiac biomarkers market and held the largest revenue share of 35.82% in 2025. Cardiac troponin, especially Troponin I (cTnI), is considered the gold-standard biomarker for detecting myocardial infarction because of its high sensitivity and specificity for myocardial damage. Cardiovascular diseases are one of the major causes of mortality worldwide, driving continuous demand for accurate and early diagnostic tools. Regulatory approval boosts clinical confidence and encourages healthcare providers to upgrade from conventional assays. The assay’s compatibility with existing diagnostic systems lowers implementation costs and supports rapid uptake, especially in emergency and acute care settings. For instance, in November 2025, QuidelOrtho Corporation received U.S. FDA 510(k) clearance for its VITROS high-sensitivity Troponin I Assay. The test is designed for quantitative measurement of cardiac troponin I in human plasma to support the diagnosis of myocardial infarction.
However, CK-MB segment is anticipated to grow at a considerable growth rate over the forecast period, driven by the fact that Creatine kinase (CK) has largely been replaced by troponin as the preferred cardiac biomarker. However, CK may still be used in specific situations, such as detecting a second heart attack occurring shortly after an initial event. CK-MB, a particular creatine isoenzyme kinase found predominantly in heart muscle, increases when heart muscle cells are damaged. The creatine kinase-MB test is relatively lower in cost as compared to high-sensitivity troponin tests, making CK-MB an attractive option for hospitals and diagnostic laboratories in cost-sensitive and emerging markets. For instance, the cost of a high-sensitivity Troponin-I test at Tata 1mg is approximately USD 19.6, whereas a CK-MB test is priced around USD 5.4.
Application Insights
The acute coronary syndrome segment dominated the cardiac biomarkers market and held the largest revenue share of 56.62% in 2025, owing to the rising disease burden and the need for early and accurate diagnosis. Coronary artery disease (CAD) is the leading cause of death among adults and one of the most common reasons for emergency department (ED) visits worldwide. Standard diagnostic tools such as the 12-lead electrocardiogram (ECG), while essential, have limited sensitivity of only about 35-50% for detecting acute myocardial infarction (AMI), especially in early stages. As a result, there is a strong need for additional diagnostic methods that can improve accuracy and speed of diagnosis. Cardiac biomarkers fulfill this role by providing measurable and quantifiable indicators of heart muscle injury through blood tests.
On the other hand, the myocardial infarction segment is anticipated to grow at the fastest rate over the forecast period. Myocardial infarction remains one of the leading causes of death worldwide, accounting for a large share of cardiovascular disease related mortality. Millions of people experience heart attacks each year, creating sustained demand for accurate and timely diagnostic tools in emergency and hospital settings. Early detection of myocardial infarction is critical because prompt treatment significantly improves survival and reduces long-term complications. This has increased reliance on cardiac biomarkers, particularly troponin, which is now the gold standard for diagnosing myocardial infarction diagnosis due to its high sensitivity and specificity for heart muscle injury.
End Use Insights
The laboratory testing segment dominated the market and held the largest revenue share of 63.90% in 2025. Laboratory-based assays provide precise, quantitative, and reproducible results, which are essential for accurate diagnosis and risk stratification. Physicians rely on these results to make critical treatment decisions, especially in emergency and acute care settings. Biomarkers such as cardiac troponin are widely regarded as the gold standard for diagnosing myocardial infarction due to their high sensitivity and specificity for myocardial injury.
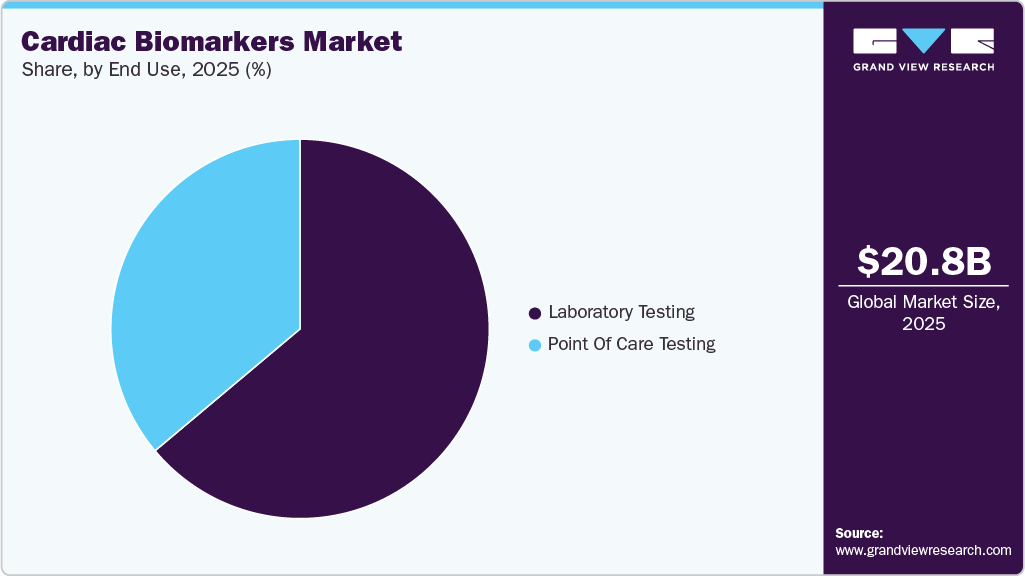
The point of care testing segment is anticipated to grow at the fastest CAGR over the forecast period. The segment growth is driven by the increasing need for rapid and accurate diagnosis of cardiovascular diseases. Cardiovascular conditions such as heart attacks and acute coronary syndromes require immediate clinical decisions, and point-of-care testing enables healthcare providers to obtain test results within minutes rather than hours. This faster turnaround time supports early diagnosis, quicker treatment initiation, and improved patient outcomes.
Regional Insights
North America cardiac biomarkers market dominated the global industry and accounted for the largest revenue share of 43.32% in 2025. The growth can be attributed to technological advancement and regulatory support, which have played a key role in strengthening adoption across the region. High-sensitivity assays have become increasingly embedded in clinical workflows, enabling clinicians to make informed diagnostic and prognostic decisions within hours of patient presentation. Regulatory clearances in the U.S. have encouraged innovation in assay design, analytical stability, and workflow efficiency, allowing laboratories to expand testing menus without compromising turnaround time. In parallel, healthcare institutions across North America continue to invest in automated analyzers and integrated diagnostic platforms to manage growing patient volumes.
U.S. Cardiac Biomarkers Market Trends
The U.S. held the largest market share of the cardiac biomarkers market in North America. Regulatory advancements related to assay clearance have also contributed to market growth in the U.S., encouraging innovation and adoption of next-generation diagnostics. For instance, in January 2025, the U.S. Food and Drug Administration cleared Abbott’s Alere NT-proBNP assay for the Alinity i system for aiding heart failure diagnosis, enhancing laboratory workflow efficiency, and expanding cardiac testing menus in many hospital labs across the country. The clearance of this widely used cardiac peptide assay reflects ongoing enhancements in biomarker performance and analytical stability, supporting clinicians in evaluating patients with suspected congestive heart failure
Europe Cardiac Biomarkers Market Trends
Europe accounts for a considerable share of the cardiac biomarkers market due to the cardiovascular conditions remain among the leading causes of hospitalization, morbidity, and mortality, particularly as Europe’s population continues to age. Hospitals, emergency departments, and outpatient clinics increasingly rely on cardiac biomarkers, including high-sensitivity troponins, BNP, and NT-proBNP, for rapid diagnosis, risk stratification, and monitoring of myocardial infarction and heart failure. Rising prevalence of comorbidities such as hypertension, diabetes, and obesity further increases testing demand.
Cardiac biomarkers market in the UK is driven by the increasing prevalence of cardiovascular diseases, estimated at approximately 10,900 per 100,000 people, and the rising demand for early, accurate diagnostic tools. Technological innovation and research evidence have further reinforced market expansion. For instance, April 2025, London School of Hygiene & Tropical Medicine, reported that a simple £5 blood test measuring troponin levels could improve prediction of a person’s risk of heart attack and stroke within 10 years more accurately than standard risk assessments, based on anonymized data from over 62,000 people across 15 studies in Europe, North America, and Australia. The model improved accuracy up to four times compared with cholesterol alone and reclassified up to 8% of intermediate-risk individuals, enabling targeted preventive treatment and demonstrating the clinical value of biomarker-guided risk assessment in the UK.
The Germany cardiac biomarkers market is driven due to the rising burden of cardiovascular diseases, with a prevalence of approximately 11,647 per 100,000 people, and the increasing clinical demand for early, precise diagnostic tools. Research, investment and innovation are also driving market growth by expanding testing capabilities and improving clinical outcomes. For instance, in October 2025, Deutsche Herzstiftung, reported that it had allocated a special research budget of more than 1.1 million USD to support twelve innovative projects selected from 62 applications, targeting the diagnosis and treatment of KHK and heart attack. Individual project funding ranged from 65,857 USD to 140,910 USD, covering topics such as inflammation, thromboinflammation, prognostic biomarkers, and AI-enhanced diagnostics
Asia Pacific Cardiac Biomarkers Market Trends
Asia Pacific is estimated to show the fastest growth in the market over the forecast period. Guideline-driven care and standardized clinical pathways have significantly accelerated adoption in several countries. In Japan and Australia, cardiac biomarkers are deeply integrated into routine practice, supported by well-defined diagnostic thresholds and national clinical guidelines. Lower biomarker cut-offs and serial testing algorithms promote earlier detection, repeat testing, and proactive disease management, particularly among elderly and high-risk population.
The cardiac biomarkers market in China is growing rapidly due to the country’s exceptionally large cardiovascular disease burden, with an estimated prevalence of approximately 8,500 per 100,000 people, and the growing emphasis on early diagnosis and risk stratification. Cardiovascular conditions affect hundreds of millions of people, creating sustained demand for laboratory-based tools that can support screening, diagnosis, and long-term monitoring. Biomarkers such as troponins and natriuretic peptides are increasingly used to identify myocardial injury, heart failure, and disease progression in both hospital and outpatient settings.
Cardiac biomarkers market in Japan is among the most advanced in the region. Regulatory and guideline-driven refinements have further accelerated market growth by increasing testing frequency and broadening eligible patient populations. For instance, October 2023, Japanese Heart Failure Society (JHFS), published a revised Statement on the Use of Blood BNP and NT-proBNP in Heart Failure Care that lowered diagnostic cut-offs from 40 pg/mL to 35 pg/mL for BNP and from 400 pg/mL to 300 pg/mL for NT-proBNP, while defining normal values of BNP ≤18.4 pg/mL and NT-proBNP ≤55 pg/mL. It also specified intermediate ranges of BNP 35-100 pg/mL and NT-proBNP 125-300 pg/mL and high-risk thresholds of BNP ≥200 pg/mL or NT-proBNP ≥900 pg/mL, reinforcing routine biomarker use for diagnosis, prognosis, and treatment guidance across Japan.
Latin America Cardiac Biomarkers Market Trends
The Latin America cardiac biomarkers market has exhibited significant growth in the past few years., due to the high and persistent burden of cardiovascular diseases across countries such as Brazil and Argentina. Conditions including myocardial infarction, acute coronary syndrome, and heart failure remain leading causes of mortality and hospital admissions, driving demand for rapid and reliable diagnostic tools. Cardiac biomarkers play a critical role in confirming myocardial injury, enabling risk stratification, and guiding treatment decisions, particularly in emergency and critical care settings.
Brazil cardiac biomarkers market is witnessing steady growth in the region. As the country the largest population in Latin America, rising emphasis on preventive healthcare and improved access to diagnostic services further contribute to the growth of the Brazilian cardiac biomarkers market. Public health initiatives and physician-led education programs encourage early screening and monitoring of individuals at high cardiovascular risk. As preventive cardiology gains importance, cardiac biomarker testing is increasingly incorporated into routine assessments and follow-up care.
Middle East and Africa Cardiac Biomarkers Market Trends
The MEA region includes South Africa, Saudi Arabia, and other Middle Eastern countries. Healthcare infrastructure development and expansion of diagnostic networks strongly support market growth across the region. Countries such as Saudi Arabia, the UAE, and Kuwait are investing heavily in modern hospitals, laboratories, and specialized cardiac centers equipped with advanced immunoassay platforms and high-sensitivity biomarker tests.
Saudi Arabia’s cardiac biomarkers market is expanding, with investment in healthcare infrastructure and adoption of advanced diagnostic technology are further supporting market growth. Under national health initiatives, the government is upgrading hospitals, laboratories, and specialized cardiac centers, enabling the use of modern biomarker assays. Major cities such as Riyadh, Jeddah, and Dammam are leading in adoption due to higher concentrations of tertiary hospitals and research facilities, while smaller regions are gradually gaining access through digital health platforms and automated laboratory networks.
Key Cardiac Biomarkers Company Insights
The major market players operating in the cardiac biomarkers market are actively adopting strategic initiatives such as new product launches, mergers and acquisitions, partnerships, and geographic expansions to strengthen their competitive positioning. Companies are focusing on the development of high-sensitivity troponin assays, expanded biomarker panels, and integrated point-of-care solutions to address the growing demand for rapid and accurate cardiac diagnostics. Strategic collaborations with hospitals, research institutes, and technology providers are enabling faster clinical validation and commercialization of innovative assays. In addition, acquisitions of niche diagnostic firms and assay developers are helping leading players broaden their product portfolios and enhance technological capabilities.
Key Cardiac Biomarkers Companies:
The following key companies have been profiled for this study on the cardiac biomarkers market.
- Abbott
- Quidel Corporation
- Siemens Healthineers AG
- F. Hoffmann-La Roche Ltd.
- Danaher Corporation
- bioMérieux SA
- Bio-Rad Laboratories, Inc.
- Randox Laboratories
- Creative Diagnostics
- Life Diagnostics, Inc.
Recent Developments
-
In November 2025, the U.S. Food and Drug Administration had granted QuidelOrtho Corp. 510(k) clearance for the VITROS hs Troponin I Reagent Pack, an assay intended for the quantitative measurement of cardiac troponin I in human plasma to aid in diagnosing myocardial infarction, and that the commercial rollout for U.S. laboratories operating VITROS systems was set to begin later in that year, marking a new product availability following clearance on November 3, 2025 of the high-sensitivity troponin test.
-
In September 2025, Roche announced primary results from its TSIX Study Program showing that the new Elecsys Troponin T hs Gen 6 assay (sixth-generation high-sensitivity troponin T test) demonstrated high precision in identifying acute myocardial infarction and reliable exclusion of non-AMI cases, following its recent CE Mark approval.
-
In January 2025, bioMérieux agreed to acquire SpinChip Diagnostics ASA, a company developing a rapid point-of-care immunoassay platform focused on high-sensitivity cardiac troponin I, NT-proBNP and D-dimer assays, with first products expected in 2026.
-
In October 2024, the Atellica IM High-Sensitivity Troponin I (TnIH) test received U.S. FDA clearance with a prognostic claim to help predict major cardiac events and death up to one year after presenting with acute coronary syndrome, expanding its clinical utility beyond standard acute myocardial infarction diagnosis. This indication aids clinicians in risk stratification and long-term management.
Cardiac Biomarkers Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 23.02 billion
Revenue forecast in 2033
USD 49.31 billion
Growth rate
CAGR of 11.5% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in (USD Billion/Million) and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Spain; Italy; Denmark; Norway; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; Quidel Corporation; Siemens Healthineers; F. Hoffmann-La Roche Ltd; Danaher Corporation; Biomérieux SA; Bio-Rad Laboratories Inc.; Randox Laboratories Ltd.; Creative Diagnostics; Life Diagnostics
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Cardiac Biomarkers Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021-2033. For this report, Grand View Research has segmented the cardiac biomarkers market based on type, application, end use, and region.
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Troponin
-
CK-MB
-
Myoglobin
-
BNP and NT-proBNP
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Acute Coronary Syndrome
-
Myocardial Infarction
-
Congestive Heart Failure
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Laboratory Testing
-
Point of Care Testing
-
-
Regional Outlook (Revenue, USD Million, 2021-2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the cardiac biomarkers market include Abbott, Siemens Healthineers, Thermo Fisher Scientific, Inc., Becton, Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories, Inc., F. Hoffmann-La Roche Ltd, and Randox Laboratories Ltd.
b. Key factors that are driving the cardiac biomarkers market growth include the high prevalence of cardiovascular diseases such as Acute Coronary Syndrome (ACS), Acute Myocardial Infarction (AMI), ischemia, and Congestive Heart Failure (CHF) globally and the rising adoption of biomarkers as a prognostic tool.
b. The global cardiac biomarkers market size was estimated at USD 20.81 billion in 2025 and is expected to reach USD 23.02 billion in 2026.
b. The global cardiac biomarkers market is expected to grow at a compound annual growth rate of 11.5% from 2026 to 2033 to reach USD 49.31 billion by 2033.
b. North America dominated the cardiac biomarkers market with a share of 43.32% in 2025. This is attributable to the significant disease burden stemming from the high incidence of cardiovascular diseases in North America and the increasing popularity of PoC diagnostics. High awareness levels associated with advanced diagnostic & prognostic tools and supportive reimbursement policies facilitate adoption of novel technologies in the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










