- Home
- »
- Medical Devices
- »
-
Covered Stent Market Size And Share, Industry Report, 2033GVR Report cover
![Covered Stent Market Size, Share & Trends Report]()
Covered Stent Market (2026 - 2033) Size, Share & Trends Analysis Report By Device Type (Balloon-Expandable Covered Stent, Self-Expanding Covered Stent), By Anatomy, By Region (North America, Europe, Asia Pacific, Latin America, MEA), And Segment Forecasts
- Report ID: GVR-4-68040-421-3
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Covered Stent Market Summary
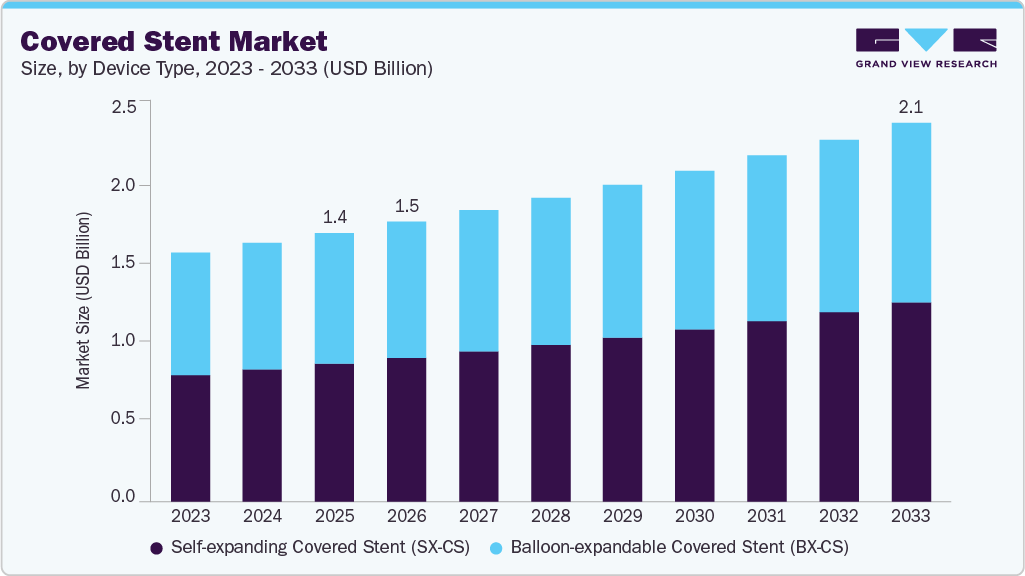
The global covered stent market size was estimated at USD 1.4 billion in 2025 and is projected to reach USD 2.1 billion by 2033, growing at a CAGR of 4.6% from 2026 to 2033. The growth is driven by the increasing prevalence of peripheral artery disease and complex aortoiliac occlusive conditions, rising volumes of endovascular procedures, and expanding preference for minimally invasive revascularization.
Key Market Trends & Insights
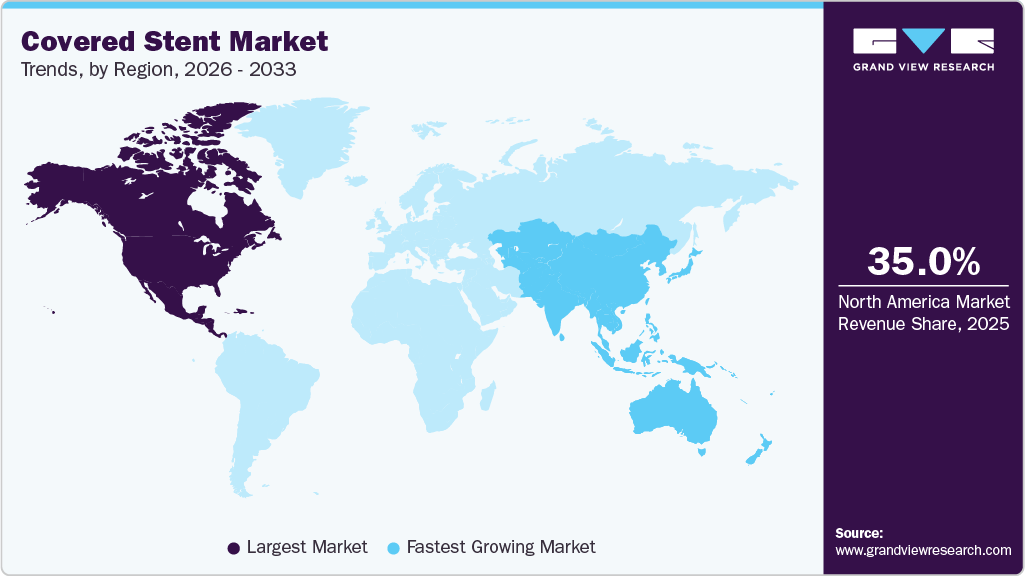
- The North America covered stent market dominated the global market in 2025 and accounted for the largest revenue share of over 35%.
- The U.S. covered stent market is anticipated to register the fastest growth rate during the forecast period.
- In terms of the device type segment, the self-expanding covered stent (SX-CS) segment held the largest revenue share in 2025.
- In terms of the anatomy segment, the SFA segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.4 Billion
- 2033 Projected Market Size: USD 2.1 Billion
- CAGR (2026-2033): 4.6%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Advancements in covered stent design, including enhanced deliverability, radial strength, and long-term patency performance, continue to support broader clinical adoption. The key drivers of the market are the rising prevalence of peripheral artery and aortoiliac occlusive diseases, technological advancements in covered stent design and materials, increasing adoption of minimally invasive endovascular procedures and increasing adaptation of minimally invasive endovascular procedures.Market Drivers
Rising Prevalence of Peripheral Artery and Aortoiliac Occlusive Diseases
The rising prevalence of peripheral artery disease (PAD) and aortoiliac occlusive disease (AIOD) is a major driver for the industry. With aging populations and increasing incidence of comorbidities such as diabetes and hypertension, the need for effective endovascular interventions is growing. Covered stents provide durable treatment for complex lesions and reduce restenosis risks compared with conventional therapies. For instance, in June 2023, the American Heart Association reported that PAD affects over 12 million Americans, with higher prevalence and complication risks among minority populations, driven by disparities in care access and treatment.
Technological Advancements in Covered Stent Design and Materials
Enhanced imaging compatibility and radiopacity in new stent designs further support procedural accuracy. Clinicians can navigate tortuous or calcified vessels effectively, minimizing complications. This technological evolution encourages hospitals and intervention centers to invest in advanced covered stent platforms. In November 2024, Medical Murray highlighted innovations in covered stent and frame technology, emphasizing advanced materials such as ePTFE, polyester, polyurethanes, and silicones for minimally invasive peripheral arterial procedures.
Funding and Strategic Investments in Covered Stent
Funding and strategic investments in covered stents are driven by rising demand for advanced endovascular solutions, with companies focusing on product innovation, clinical validation, and geographic expansion. Investments are directed toward R&D for improved durability and deliverability, strategic partnerships, and acquisitions to strengthen market presence, particularly in high-growth regions and complex vascular applications.
Following are some of the recent funding initiatives by key players in the market
Date
Company
Description
In February 2026
Artivion, Inc.
Extended USD 25 million loans to Endospan to support the FDA IDE study of Nexus aortic arch stent graft system, which aims to accelerate clinical evaluation and provide a minimally invasive alternative for aortic arch repairs.
In December 2025
W.L.Gore and Associates, Inc
Gore received FDA approval for a new venous stent in the VIabahn family, designed for treating deep venous disease in the inferior vena cava and iliac veins.
Market Concentration & Characteristics
The industry is witnessing advancement towards device engineering, which is focused on enhanced deliverability, conformability, and long-term patency due to a medium degree of innovation. The innovations in this sector include thinner strut architecture, improved expanded ePTE and polyurethane coverings, dual-layer and hybrid scaffold designs, and optimized radial force profiles that address complex anatomies. In November 2025, enrollment of the Iliav cohort in the AGILITY IDE Study evaluating the Revello self-expanding covered stent, designed with a low-profile delivery system and enhanced confirmability, was completed by BD.
Players in the balloon-expandable covered stent industry form partnerships with healthcare providers, research institutions, and pharmaceutical or technology firms to enhance product development, integrate innovative delivery systems, and expand their reach across both domestic and export markets in March 2024, Getinge AB partnered with Cook under an exclusive U.S. distribution agreement for the iCast covered stent system, enabling broader patient access for iliac arterial occlusive disease while maintaining manufacturing through Getinge AB’s Atrium Medical entity.
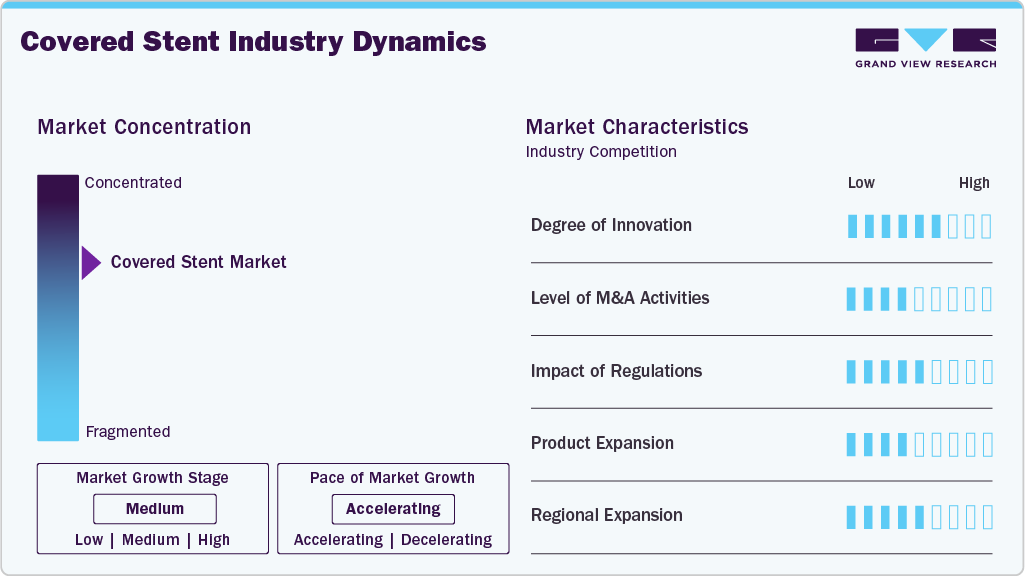
The regulatory environment has a high impact as it is stringent, particularly for Class III implantable vascular devices. The American PMA and Europe (MDR) have strict and stringent polices such as the requirement of clinical evidence, robust post-market surveillance, long-term follow-up, and comprehensive clinical evidence, risk management, documentation, structured post-market clinical follow-up (PMCF), respectively. In December 2025, the Center for Devices and Radiological Health and the Center for Biologics Evaluation and Research issued a guide for how the FDA evaluates real-world data for generating real-world experience.
Players of covered stent markets are forming strategic partnerships and collaborations with healthcare professionals, research institutes, and pharmaceutical technology firms, which enhance product development. In March 2024, Getinge AB partnered with Cook under a U.S. distribution agreement for iCast covered stent system, which enabled broader patient access for iliac arterial occlusive disease and maintained manufacturing through Getinge AB’s Atrium Medical entity.
Regional expansion is significant, with companies actively implementing strategies to enhance product capabilities and extend the reach by tapping new and existing markets. In November 2024, Solaris Endovascular launched in the U.S. to develop stent graft solutions for dialysis and peripheral artery disease. The MD Chairman and Chief Medical and Scientific Officer at Solaris Endovascular stated that, “Solaris Endovascular’s advanced covered stent platforms will not only reduce the burden of repeated interventions but will also dramatically enhance the quality of life and life expectancy for those who suffer from these debilitating conditions”.
Device Type Insights
By device type, the Self-expanding Covered Stent (SX-CS) segment held the largest revenue share of over 51.5% in 2025. This dominance is attributed to the increasing incidence of complex vascular diseases and the launch of technologically advanced stents by key market players. In March 2026, BD introduced the Revello vascular covered stent, which is a self-expanding covered stent engineered for delivering robust radial support and optimized vessel conformity for treating iliac artery atherosclerotic lesions. The device consists of a nitinol scaffold with an advanced ePTE covering to maintain arterial patency and adapt to challenging anatomies. It is used in common and external artery disease, which has enabled its broader clinical adaptation. The device has received CE approval in Europe.
The Balloon-expandable Covered Stent (BX-CS) segment is anticipated to grow at a significant CAGR over the forecast period. The demand is driven by complex aortic and peripheral vascular interventions and increasing product innovations. In July 2025, W.L. Gore and Associates Inc, have revealed an expanded CE Mark indication for GORE VIABAHN VBX Balloon Expandable Endoprosthesis, which is used as a bridging stent in both fenestrated and branched endovascular aneurysm repair. It has enabled broader clinical application in complex aorto-visceral aneurysmal disease. The VBX Balloon expandable endoprosthesis offers enhanced precision in delivery and expanded diameter, which supports improved procedural outcomes in challenging vascular anatomies and hence is a key solution in the balloon expandable stent category.
Anatomy Insights
By anatomy, the SFA (Superficial Femoral Artery) dominated the market with the largest revenue share of 23.3% in 2025. The strong presence of SPA in this segment is driven by the high prevalence of peripheral artery disease (PAD) and growing adoption of advanced intervention devices, which improve patient outcomes and reduce recovery time. The innovation across the SFA products also drives this growth. In 2025, according to the Medtronic REALITY Study evaluation, endovascular therapies for challenging Superficial Femoral Artery (SFA) diseases showcased the strong reliability of contemporary SFA intervention technologies in routine practice. The study comprised 100 patients with long, complex, and heavily calcified SFA lesions, and 76.7% of patients maintained primary patency at 12 months, while 92.6% freedom from clinically driven target lesion revascularization (CD-TLR).
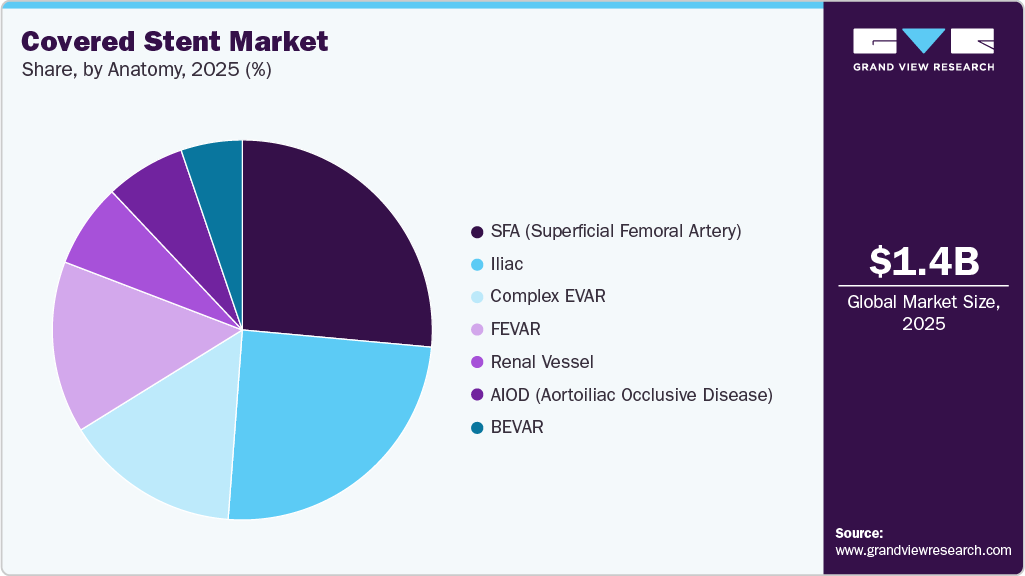
The BEVAR segment is anticipated to grow at the fastest CAGR over the forecast period. Rising CVD incidence and the technologically advanced product launch drive the growth of the market. In November 2025, Bentley InnoMed received CE Mark approval for its BeGraft Plus stent graft for use in branched endovascular aneurysm repair. The stent, available in cleared markets, offers up to 78 mm length bridge complex anatomies, providing stable support and flexibility. Early clinical experience is positive, offering physicians a reliable on-label option for complex aortic repair.
Regional Insights
North America Covered Stent Market Trends
North America dominated the covered stent market, accounting for over 35% of revenue in 2025, driven by the prevalence of peripheral artery diseases and growing adoption of minimally invasive endovascular interventions. Hospitals and outpatient vascular centers account for the majority of covered stent procedures with high potential volumes in iliac, superficial femoral, and complex EVAR/FEVAR/BEVAR interventions. In March 2025, the Journal of Invasive Cardiology published a single-center study that evaluated the off-label use of VIABAHN VBX Ballon Expandable Endoprosthesis in pediatric congenital heart disease. The report highlighted that 22 patients had no major procedural complications and significant hemodynamic improvement.
U.S. Covered Stent Market Trends
The covered stent market in the U.S. is driven by the rising prevalence of peripheral arterial disease, complex aortoiliac and femoropopliteal occlusions, and growing adoption of minimally invasive endovascular interventions. In June 2024, investigators at Washington University School of Medicine, St. Louis, Missouri, reported successful fabrication and deployment of the novel AIFEN fenestrated balloon-expandable covered stent for aortoiliac occlusive disease. The 9.7 mm × 35.7 mm device (expanded to 10 mm) incorporates a 7 mm fenestration and was implanted across the aortic bifurcation, with angiography confirming patency of both the AIFEN and contralateral iliac stent without vessel injury.
Europe Covered Stent Market Trends
The covered stent market in Europe is expected to grow significantly over the forecast period. The European covered stent market is driven by the increasing burden of peripheral arterial disease, aortic aneurysms, and complex vascular occlusions across aging populations in Western and Central Europe. High adoption of endovascular procedures over open surgery in Germany, France, Italy, Spain, and the UK supports demand for covered stents in iliac, femoropopliteal, and complex EVAR or FEVAR interventions. In September 2022, an article published in the Journal of Clinical Medicine analyzed 279 patients treated with 683 balloon expandable covered bridging stent during fenestrated endovascular aneurysm repair, the study showed freedom from stent related complications of 97 percent at 1 year and 94 percent at 3 years, with overall reintervention free survival of 98 percent and 97 percent at the same time points, respectively.
The UK covered stent industry is influenced by structured vascular service delivery models and consolidation of complex procedures into designated arterial centers. Concentration of expertise within regional vascular hubs supports higher volumes of iliac and femoropopliteal interventions. Increasing detection of vascular disease through screening initiatives, including abdominal aortic aneurysm screening programs, also contributes to procedural demand. In September 2024, UK researchers from the University of Surrey helped publish ESC guidelines recommending regular PAD screenings, early detection via the estimated ankle brachial pressure index, personalized treatment plans, and lipid-lowering therapies to prevent serious complications
The covered stent industry in Germany is expected to grow significantly during the forecast period, supported by the rising demand due to its high procedural volumes in peripheral and complex aortic interventions. Large tertiary hospitals and certified vascular centers perform substantial numbers of iliac, femoropopliteal, and complex EVAR procedures each year. In July 2022, Bentley InnoMed completed enrollment of 100 patients in its BGP study evaluating the BeGraft peripheral balloon expandable covered Stent as a bridging stent in fenestrated endovascular aortic aneurysm repair for complex aortic aneurysms.
Asia Pacific Covered Stent Market Trends
The covered stent industry in Asia Pacific is expected to register the fastest growth rate over the forecast period, driven by increasing healthcare access, peripheral arterial disease, aortic aneurysms, and complex vascular conditions. Growing awareness and affordability of it are also driving the growth. In June 2025, an article published in Frontiers in Cardiovascular Medicine discussed the rising burden of lower extremity peripheral arterial disease (PAD) in China. The study highlighted that new PAD cases reached 2.45 million in 2021 and are projected to rise to 4.04 million by 2046, emphasizing the need for enhanced prevention and management strategies.
China's covered stent market is anticipated to register considerable growth during the forecast period. The covered stent market in China is expanding rapidly due to the rising prevalence of peripheral arterial disease, aortic aneurysms, and complex vascular conditions. The procedural efficiency, shorter hospital stays, and the growth of tertiary and specialized vascular centers support adoption. Partnerships between manufacturers and hospitals for training, real-world evidence generation, and device optimization further drive market uptake. In May 2022, researchers at West China Hospital of Sichuan University, Chengdu, China, published the protocol of the NEONATAL randomized controlled trial comparing the BARD LIFESTREAM Covered Stent and the GORE VIABAHN Self-Expandable Covered Stent for aortoiliac obstructive disease.
The covered stent market in India is witnessing significant growth during the forecast period. The increasing burden of cardiovascular diseases, including peripheral arterial disease, aortic aneurysms, and complex coronary and peripheral vascular conditions, drives the market. In September 2025, AIIMS Bhopal completed Sinus Venosus Atrial Septal defect (ASD) closure using an advanced covered stent. The case involved complex cardiac anatomy, with the patient having an additional pulmonary vein that required protection.
Latin America Covered Stent Market Trends
The covered stent industry in Latin America is expected to witness considerable growth over the forecast period. The key markets, such as Brazil and Argentina, are seeing growth due to increasing cardiovascular disease prevalence and expansion of tertiary care hospitals and specialized endovascular centers. Adoption of advanced covered stents is aided by clinician training programs, technology transfer from international manufacturers, and a focus on reducing procedural complications and hospitalization time.
The Brazil covered stents market is the largest in Latin America, driven by rising cardiovascular disease burden, increasing healthcare expenditure, and government-led initiatives to strengthen cardiac care infrastructure. Expanding clinical research collaborations and improving access to advanced vascular interventions are further supporting adoption, although procurement complexities and pricing sensitivity continue to influence market dynamics.
Middle East And Africa Covered Stent Market Trends
The covered stent industry in the Middle East and Africa is anticipated to grow significantly. Covered stent uptake in this region is boosting due to government healthcare initiatives, partnerships with global device companies, and increased availability of balloon-expandable and self-expanding stents for complex interventions, particularly in iliac, renal, and femoral vessels.
The Saudi Arabia covered stents market is a smaller but steadily growing segment, supported by increasing prevalence of peripheral artery disease, rising healthcare investments, and expansion of advanced cardiovascular centers. Adoption is primarily concentrated in high-end hospitals for complex vascular procedures, with growth further driven by the gradual uptake of minimally invasive technologies and the ongoing modernization of the healthcare system.
Key Covered Stent Company Insights
Key players operating in the covered stent market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Covered Stent Companies:
The following key companies have been profiled for this study on the covered stent market.
- Cook
- Bentley InnoMed GmbH
- Medtronic
- Getinge AB
- iVascular.
- W. L. Gore & Associates, Inc.
- Solaris Endovascular
- LeMaitre Vascular, Inc.
- Endologix LLC
- Artivion, Inc
- Merit Medical Systems
- BD (Angiomed GmbH & Co. Medizintechnik KG)
Recent Developments
-
In July 2025, Getinge’s iCast covered stent was approved by the FDA PMA. It received approval as a bridging stent for use in aneurysmal disease, further broadening clinical use in complex cases.
-
In November 2025, Bentley got CE approval for the BeGraft Plus covered stent graft system. It is a bridging stent in branched endovascular aneurysm repair (BEVAR) and is expanding its portfolio in complex aortic procedures. It will be used in patients requiring endovascular abdominal aortic aneurysm repair with a branched aortic endovascular graft to keep visceral arteries perfused. The system is indicated for permanent intraluminal placement in iliac and renal arteries and will restore or improve vessel patency and seal aneurysms, acute perforations, ruptures, and fistulas.
-
In October 2025, Getting AB’s Advanta V12 balloon-expandable covered stent received updated CE approval in Europe for use as a bridging stent. It is used in complex endovascular aneurysm repair procedures such as fenestrated endovascular aneurysm repair (FEVAR), branched endovascular aneurysm repair (BEVAR), and Iliac branch device (IBD) aneurysmal repair. The system helps to treat a wide spectrum of occlusive and aneurysmal diseases.
Covered Stent Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.5 billion
Revenue forecast in 2033
USD 2.1 billion
Growth rate
CAGR of 4.6% from 2026 to 2033
Actual data
2021 - 2025
Forecast data
2026 - 2033
Quantitative units
Unit volume and procedure volume in thousand units; revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Volume & revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Device type, anatomy, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Cook; Bentley InnoMed GmbH; Medtronic; Getinge AB; iVascular.; W. L. Gore & Associates, Inc.; Solaris Endovascular; LeMaitre Vascular, Inc.; Endologix LLC; Artivion, Inc; Merit Medical Systems; BD (Angiomed GmbH & Co. Medizintechnik KG)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Covered Stent Market Report Segmentation
This report forecasts volume & revenue growth at the global, regional, and country levels,and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global covered stent market report based on device type, anatomy, and region:

Total Addressable Market (TAM)
-
Anatomy Outlook (Unit Volume and Procedure Volume in Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Iliac
-
Common Iliac
-
External Iliac
-
-
AIOD (Aortoiliac Occlusive Disease)
-
SFA (Superficial Femoral Artery)
-
Complex EVAR
-
FEVAR
-
BEVAR
-
Renal Vessel
-
Serviceable Obtainable Market (SOM)
-
Device Type Outlook (Unit Volume and Procedure Volume in Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Balloon Expandable
-
Iliac
-
Common Iliac
-
External Iliac
-
-
AIOD (Aortoiliac Occlusive Disease)
-
Complex EVAR
-
FEVAR
-
BEVAR
-
Renal Vessel AIOD (Aortoiliac Occlusive Disease)
-
-
Self-expanding Covered Stent (SX-CS)
-
External Iliac
-
SFA (Superficial Femoral Artery)
-
Complex EVAR
-
FEVAR
-
BEVAR
-
-
-
Anatomy Outlook (Unit Volume and Procedure Volume in Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Iliac
-
Common Iliac
-
External Iliac
-
-
AIOD (Aortoiliac Occlusive Disease)
-
SFA (Superficial Femoral Artery)
-
Complex EVAR
-
FEVAR
-
BEVAR
-
Renal Vessel
-
-
Regional Outlook at both TAM and SOM (Unit Volume and Procedure Volume in Thousand Units; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some of the key players operating in the market are Boston Scientific Corporation, Medtronic, W. L. Gore & Associates, Inc., Cook, Terumo Corporation, B. Braun Interventional Systems, Inc., BD, Getinge, Biotronik.
b. The key factors driving the covered stents market include the rising prevalence of vascular diseases, technological advancements in stent design, and increasing awareness and early diagnosis of conditions requiring vascular intervention.
b. The global covered stent market size was estimated at USD 1.4 billion in 2025 and is expected to reach USD 1.5 billion in 2026.
b. The global covered stent market is expected to grow at a compound annual growth rate of 4.6% from 2026 to 2033 to reach USD 2.1 billion by 2033.
b. North America dominated the covered stent market with a share of over 35% in 2025. This is attributable to the growth in R&D investments by the market participants and FDA approvals for covered stents.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










