- Home
- »
- Pharmaceuticals
- »
-
Food Allergy Diagnostics & Therapeutics Market Report 2030GVR Report cover
![Food Allergy Diagnostics And Therapeutics Market Size, Share & Trends Report]()
Food Allergy Diagnostics And Therapeutics Market (2024 - 2030) Size, Share & Trends Analysis Report By Product (Diagnostics, Therapeutics), By Allergen Source, By End-use, By Region, And Segment Forecasts
- Report ID: GVR-3-68038-746-9
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2022
- Forecast Period: 2024 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
“2030 Food allergy diagnostics & therapeutics market value to reach USD 13.28 billion”
The global food allergy diagnostics and therapeutics market size was estimated at USD 7.91 billion in 2023 and is projected to grow at a CAGR of 8.0% from 2024 to 2030. The increasing incidence of food allergies is expected to fuel market growth. Allergic reactions can result in various symptoms, including itchiness, tongue swelling, hives, low blood pressure, diarrhea, and vomiting.
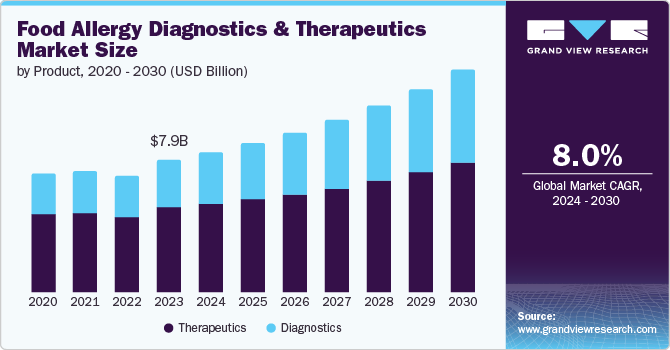
The World Allergy Organization has reported a global increase in the prevalence of food allergies. Eggs, milk, wheat, fish, and tree nuts are among the most commonly known allergens. Interestingly, fish allergies are more widespread in Asia than in Western countries. Furthermore, the market growth is attributed to increased awareness, unmet medical needs, and the introduction of innovative products. Innovative diagnostic methods, including enhanced blood tests, Skin Prick Tests, and molecular diagnostics, improve allergy detection by increasing accuracy and speed. This, in turn, drives market growth.
The global market for food allergy diagnostics and therapeutics presents promising prospects for growth. These opportunities stem from the increasing need for affordable treatment options, advancements in diagnostic tools, the emergence of innovative products like epinephrine auto-injectors, and the growing prevalence of food allergies on a global scale. Nevertheless, it is imperative to address obstacles, such as high medication costs, to ensure sustainable market expansion.
Product Insights And Trends
"The therapeutics segment is expected to witness growth at 6.5% CAGR."
The market is segmented into diagnostics and therapeutics. The therapeutics segment dominated the market in 2023 with a revenue share of 64.0%. This segment is further divided into antihistamines and epinephrine auto-injectors. Pharmaceutical corporations and research organizations are progressively allocating resources toward advancing innovative remedies for food allergies. This emphasis on therapeutic research and progress has introduced more effective treatments.
Diagnostic products encompass a range of instruments, consumables, and services. The segment is expected to grow at the fastest CAGR during the forecast period owing to the availability of several advanced and rapid test kits. An example of such a kit is the IgG food intolerance screening test kit, which detects food-specific IgG antibodies. The increasing need for blood tests in the population has significantly expanded in vitro testing. Skin prick tests are commonly utilized to detect food allergies, which boosts the utilization of in vivo testing within the market.
Allergen Source Insights And Trends
"Peanuts segment is expected to witness growth at 5.9% CAGR"
The peanuts segment dominated the market, accounting for a share of 23.1% in 2023. The segment growth is attributed to the high prevalence of peanut allergies. Peanuts top the list of allergens because they possess highly allergenic proteins specific to a population. The proteins Ara h 1, Ara h 2, and Ara h 3 are recognized for inducing severe allergic responses. These proteins exhibit resistance to both heat and digestion, thereby elevating the probability of an allergic reaction.
The eggs segment is expected to grow at the fastest CAGR of 21.3% during the forecast period. Egg allergies develop when the immune system incorrectly recognizes specific proteins in eggs as dangerous. Typical signs may involve skin irritation, nasal blockage, gastrointestinal problems, and, in some instances, breathing difficulties. In extreme situations, an egg allergy can trigger anaphylaxis, a critical allergic response.
End-use Insights
“Hospitals and clinics are projected to witness a growth rate of 8.1% CAGR”
In 2023, the hospitals and clinics segment dominated the market due to a surge in individuals seeking medical assistance for allergies. The rise in hospitalization for allergic cases has also contributed to the segment growth. Furthermore, the increasing risk of severe allergic reactions has led to a rise in early therapeutic interventions in hospitals and clinics.
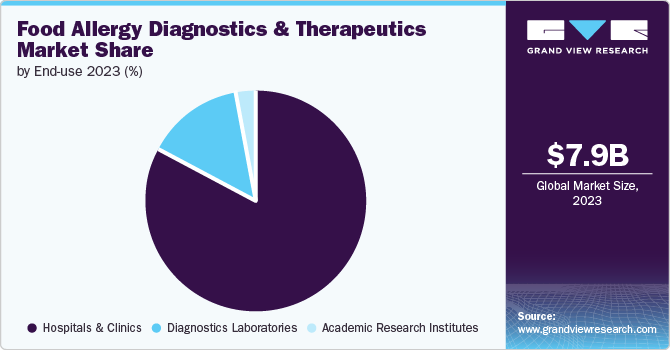
The diagnostic laboratories segment is projected to experience substantial growth during the forecast period, primarily driven by the widespread use of allergy diagnostic kits in disease diagnosis. Allergists frequently advise patients to undergo blood or skin tests to diagnose food allergies and determine the presence of food-specific immunoglobulin E antibodies in their bodies. In addition, a rise in the number of diagnostic centers contributes to segment growth.
Regional Insights & Trends
“North America is a leading region to witness growth rate at 7.3% CAGR”
The food allergy diagnostics and therapeutics market in North Americadominated the global industry in 2023. North America is an advanced region with significant healthcare spending. It dominates the global market due to the growing awareness of the opportunities in allergy medications. As a result, American companies are investing in R&D of anti-allergenic drugs. In addition, new competitors are introducing innovative products, leading to increased competition between manufacturers.
U.S. Food Allergy Diagnostics And Therapeutics Market Trends
The U.S. food allergy diagnostics and therapeutics market accounted for a share of 84.9% in 2023. The country's high prevalence of food allergies triggers the demand for advanced diagnostics and effective treatment options. The U.S. healthcare system is a market leader, known for its advanced infrastructure, widespread access to medical services, and comprehensive allergy testing and treatments.
Treatment advancements have played a key role in market growth and development. Oral immunotherapy offers promising growth to the market. For instance, in January 2020, Palforzia was approved by the U.S. Food and Drug Administration (FDA) for treating peanut allergy in children. This oral immunotherapy aims to reduce allergic reactions, such as anaphylaxis, that can happen due to accidental peanut exposure. It is suitable for children aged four to seventeen years with a confirmed peanut allergy diagnosis.
Europe Food Allergy Diagnostics And Therapeutics Market Trends
The food allergy diagnostics and therapeutics market in Europe offers potential growth and is identified as a lucrative region in this landscape. The number of cases of allergies is increasing in Europe, particularly in urban communities, due to exposure to environmental contaminants.
The UK food allergy diagnostics and therapeutics market is expected to grow rapidly due to the increasing prevalence of allergic conditions, including hay fever, asthma, and food allergies, fueling the need for diagnostic examinations and efficient therapeutic options. The rising occurrence of food allergies has stimulated the need for diagnostic examinations and treatment alternatives. The awareness among individuals regarding allergy management has also played a key role in expanding the market. Furthermore, the progress in diagnostic technologies and therapeutic methods has facilitated more efficient management and treatment of food allergies.
The food allergy diagnostics and therapeutics market in Germany has witnessed tremendous growth in recent years due to a rise in the prevalence of food allergies. Moreover, high awareness among individuals regarding various therapies available for food allergies has significantly contributed to the market's growth. Germany's exceptional medical infrastructure further acts as a catalyst in propelling the advancement of this sector.
Asia Pacific Food Allergy Diagnostics And Therapeutics Market Trends
The Asia Pacific food allergy diagnostics and therapeutics market is anticipated to grow significantly. One of the key factors influencing market growth is the increasing incidence of allergy disorders among both children and the elderly in Asian countries. Moreover, the utilization of in-vitro diagnostic (IVD) methods has risen, leading to improved availability of diagnostic tools.
The food allergy diagnostics and therapeutics market in India is expected to grow rapidly as R&D efforts intensify. Researchers and healthcare experts are dedicated to investigating food allergies, resulting in the emergence of novel diagnostic techniques and treatment alternatives. This continuous exploration plays a vital role in broadening the market by introducing cutting-edge solutions for addressing food allergies.
The China food allergy diagnostics and therapeutics market witnessed significant growth in 2023 as a result of the rise of telemedicine and digital health solutions. Patients now can consult allergists virtually and obtain personalized guidance and diagnostic services without the need for in-person appointments. The convenience and ease of access provided by telehealth play a significant role in enhancing patient outcomes and revolutionizing the field of allergy management.
Key Food Allergy Diagnostics And Therapeutics Company Insights
Some of the key companies in the food allergy diagnostics and therapeutics market include Allergy Therapeutics; Cambridge Allergy Ltd; Immunomic Therapeutics, Inc.; and Prota Therapeutics Pty Ltd. Vendors in the market are prioritizing the expansion of their customer base to establish a strong competitive advantage within the industry. Consequently, prominent industry players are implementing various strategic measures, including mergers and acquisitions, as well as forming partnerships with other leading companies.
-
Immunomic Therapeutics, Inc. is a biotechnology company in the development stage. It focuses on creating vaccines using lysosomal-associated membrane proteins to address various health targets, including allergies
-
Prota Therapeutics, founded in 2016, is an Australian biotech company specializing in developing and commercializing innovative combination probiotic allergen oral immunotherapy treatments. As a privately held entity, Prota Therapeutics is dedicated to advancing biotechnology
Key Food Allergy Diagnostics and Therapeutics Companies:
The following are the leading companies in the food allergy diagnostics and therapeutics market. These companies collectively hold the largest market share and dictate industry trends.
- Astellas Pharma, Inc.
- Aimmune Therapeutics
- Meridian Medical Technologies
- ALK-Abelló Ltd
- Medeca Pharma AB
- bioMeriux SA
- Omega Diagnostics Group PLC
- HYCOR Biomedical
- HOB Biotech Group Corp Ltd.
- Cambridge Allergy Ltd
- Medeca Pharma AB
Recent Developments
-
In February 2024, the U.S. Food and Drug Administration (FDA) approved the use of Xolair injection for the treatment of immunoglobulin E (IgE)-mediated food allergies in certain adult and pediatric populations aged one year and older. This approval is aimed at reducing the risk of Type 1 allergic reactions, including the potential for anaphylaxis, that can occur due to accidental exposure to allergenic foods
Food Allergy Diagnostics And Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 8.38 billion
Revenue forecast in 2030
USD 13.28 billion
Growth rate
CAGR of 8.0% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Quantitative units
Revenue in USD million/billion, and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, allergen source, end-use, region
Regional scope
North America, Europe, Asia Pacific, Latin America, MEA
Country scope
U.S., Canada, Mexico, Germany, UK, France, Italy, Spain, Denmark, Sweden, Norway, China, Japan, India, South Korea, Australia, Thailand, Brazil, Argentina, Saudi Arabia, Kuwait, UAE, South Africa
Key companies profiled
Astellas Pharma, Inc.; Aimmune Therapeutics; Meridian Medical Technologies; Medeca Pharma AB; bioMeriux SA; Omega Diagnostics Group PLC; Cambridge Allergy Ltd; Immunomic Therapeutics, Inc; Prota Therapeutics Pty Ltd; ALK-Abello Ltd; HYCOR Biomedical; HOB Biotech Group Corp. Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Food Allergy Diagnostics And Therapeutics Market Report Segmentation
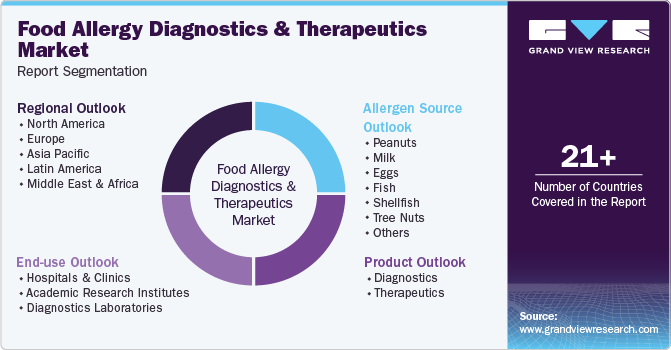
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global artificial food allergy diagnostics and therapeutics market report based on product, allergen source, end-use, and region.
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Diagnostics
-
Instruments
-
Consumables
-
Services
-
Tests
-
In Vivo/Skin
-
Skin Prick Test
-
Patch Test
-
-
In Vitro
-
-
-
Therapeutics
-
Antihistamines
-
Epinephrine Auto-injector
-
-
-
Allergen Source Outlook (Revenue, USD Billion, 2018 - 2030)
-
Peanuts
-
Milk
-
Eggs
-
Fish
-
Shellfish
-
Tree Nuts
-
Wheat
-
Soy
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals And Clinics
-
Academic Research Institutes
-
Diagnostics Laboratories
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
-
Middle East and Africa (MEA)
-
KSA
-
UAE
-
South Africa
-
-
Frequently Asked Questions About This Report
b. The global food allergy diagnostics and therapeutics market size was estimated at USD 7.91 billion in 2023 and is expected to reach USD 8.38 billion in 2024.
b. The global food allergy diagnostics and therapeutics market is expected to grow at a compound annual growth rate of 8.0% from 2024 to 2030 to reach USD 13.28 billion by 2030.
b. North America dominated the food allergy diagnostics and therapeutics market in 2023. This is attributable to increase in incidence of food allergies and high awareness about various available treatments for food allergies.
b. Some key players operating in the food allergy diagnostics and therapeutics market include Allergy Therapeutics; Cambridge Allergy Ltd; Astellas Pharma, Inc.; Immunomic Therapeutics, Inc.; Prota Therapeutics Pty Ltd; Aimmune Therapeutics; Meridian Medical Technologies; ALK-Abelló Ltd; Medeca Pharma AB; bioMérieux SA; Omega Diagnostics Group PLC; HYCOR Biomedical; and HOB Biotech Group Corp., Ltd.
b. Key factors that are driving the market growth include rising prevalence of food allergy, increasing incidence of allergic conditions among both pediatric and adult population, technological advancements and increase in number of R&D initiatives being undertaken for development of drugs with higher efficacy.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










