- Home
- »
- Pharmaceuticals
- »
-
GLP-1 Receptor Agonist Market Size, Industry Report, 2033GVR Report cover
![GLP-1 Receptor Agonist Market Size, Share & Trends Report]()
GLP-1 Receptor Agonist Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Semaglutide (Ozempic, Wegovy, Rybelsus), Tirzepatide), By Application, By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68038-694-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
GLP-1 Receptor Agonist Market Summary
The global GLP-1 receptor agonist market size was estimated at USD 66.38 billion in 2025 and is projected to reach USD 185.32 billion by 2033, growing at a CAGR of 12.4% from 2026 to 2033. GLP-1 receptor agonists have become an important therapeutic class for managing type 2 diabetes and obesity through mechanisms that enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.
Key Market Trends & Insights
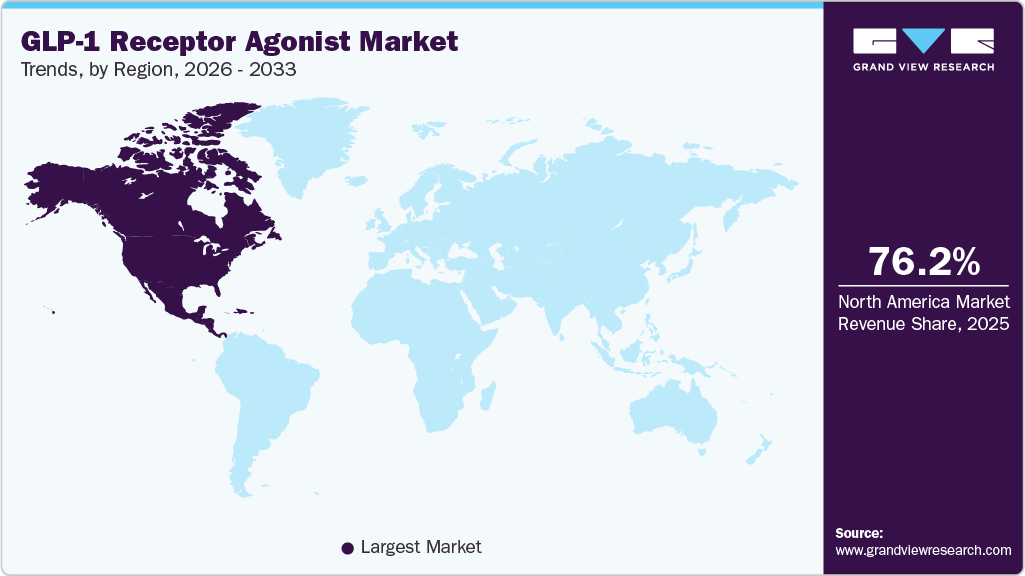
- The North America GLP-1 receptor agonist market held the largest global share of 75.46% in 2025.
- The GLP-1 receptor agonist industry in the U.S. is expected to grow significantly from 2026 to 2033.
- By product, the semaglutide (Ozempic, Wegovy, Rybelsus) segment held the highest market share of 52.83% in 2025.
- By application, the type 2 diabetes mellitus segment held the largest market share in 2025.
- By route of administration, the injectable / parenteral segment held the largest market share in 2025.
- By distribution channel, the retail pharmacies segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 66.38 Billion
- 2033 Projected Market Size: USD 185.32 Billion
- CAGR (2026-2033): 12.4%
- North America: Largest market in 2025
- Latin America: Fastest growing market
These combined metabolic effects support glycemic control, sustained weight reduction, and improved cardiometabolic outcomes, increasing their clinical adoption. The rising global prevalence of diabetes and obesity continues to drive demand for advanced treatment options that address multiple disease pathways. The increasing integration of these therapies into clinical practice reflects growing confidence among healthcare providers in their long-term benefits and treatment effectiveness. In 2025, semaglutide received expanded clinical recognition with approval for the treatment of metabolic dysfunction-associated steatohepatitis with liver fibrosis, demonstrating the broadening therapeutic scope of GLP-1 therapies. Such developments highlight the transition of GLP-1 receptor agonists from glucose-lowering agents toward comprehensive metabolic disease management solutions. The growing burden of chronic metabolic disorders is expected to sustain strong market expansion over the coming years.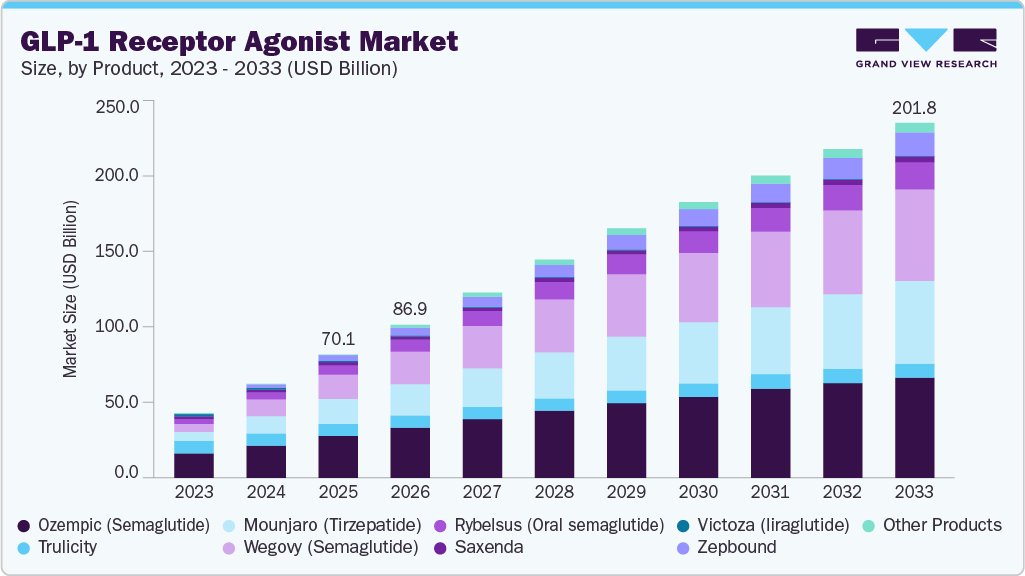
Pharmaceutical research and development activities remain a major factor supporting innovation in the GLP-1 receptor agonist market, with companies focusing on improved efficacy, convenience, and patient adherence. Advancements in oral drug delivery technologies have created new opportunities to expand patient access beyond injectable therapies. In 2024, Pfizer selected a preferred once-daily modified-release formulation of danuglipron based on pharmacokinetic study results, reflecting continued progress in oral GLP-1 development. Licensing collaborations also strengthened the pipeline, with AstraZeneca partnering for oral GLP-1 candidates targeting obesity and cardiometabolic conditions. These innovations aim to improve treatment adherence by addressing patient preference for oral medications while maintaining therapeutic effectiveness. Expanding clinical pipelines and new formulation technologies are expected to enhance competition and diversify available treatment options. Continued research investment is likely to support future product approvals and broaden the clinical applications of GLP-1 receptor agonists across metabolic disorders.
Growing awareness of chronic disease management and weight-related health risks has increased patient interest in GLP-1 receptor agonist therapies. Educational programs and clinical initiatives have improved understanding of early intervention strategies and long-term management of metabolic health. In 2024, Amylyx Pharmaceuticals advanced the development of Avexitide, a therapy targeting conditions associated with hyperinsulinemic hypoglycemia, with plans for late-stage clinical evaluation beginning in 2025. Digital health programs and disease management platforms have also integrated GLP-1 therapies into structured treatment approaches to improve patient outcomes. Rising consumer awareness of the benefits of weight management has further expanded demand beyond traditional diabetes populations. The availability of newer therapeutic indications and expanding clinical evidence continues to strengthen adoption among both patients and healthcare providers. These factors collectively support the sustained growth of the GLP-1 receptor agonist industry through 2026 and beyond.
Market Concentration & Characteristics
The GLP-1 receptor agonist market is characterized by high innovation driven by continuous advancements in peptide engineering, delivery technologies, and combination therapies. Companies are focusing on improving efficacy, reducing dosing frequency, and enhancing patient convenience through once-weekly injectables and oral formulations. The emergence of dual and triple agonists targeting multiple metabolic pathways represents a major technological leap in the segment. Innovation is also supported by robust clinical trial pipelines exploring expanded indications, including cardiovascular, renal, and liver diseases. Digital health integration and personalized treatment approaches are gradually influencing product differentiation strategies. Overall, innovation intensity remains strong due to competitive pressure among leading pharmaceutical players and significant unmet clinical needs.
Barriers to entry in the GLP-1 receptor agonist market are substantial due to the complex biologics manufacturing processes and high research and development costs. Developing peptide-based therapeutics requires specialized expertise, advanced infrastructure, and long clinical trial timelines with significant capital investment. Regulatory approval pathways for metabolic and obesity drugs are stringent, further increasing entry complexity for new participants. Strong patent protection and established brand loyalty among existing therapies create additional competitive hurdles. Supply chain requirements, including cold-chain logistics and large-scale manufacturing capacity, also limit new entrants. As a result, the market remains dominated by a few multinational pharmaceutical companies with strong financial and technological resources.

Regulatory frameworks play a critical role in shaping the GLP-1 receptor agonist industry, as health authorities impose strict safety, efficacy, and quality requirements. Agencies such as the FDA, EMA, and other regional regulators require extensive clinical evidence, particularly for cardiovascular outcomes and long-term safety in obesity treatments. Pricing regulations, reimbursement policies, and health technology assessments significantly influence market adoption and accessibility across regions. Post-marketing surveillance obligations and pharmacovigilance requirements add to compliance costs for manufacturers. Regulatory approvals for new indications can create major growth opportunities but also involve lengthy evaluation processes. Overall, regulatory oversight both ensures patient safety and acts as a key determinant of commercialization timelines and market penetration.
The market faces competition from several therapeutic alternatives used in diabetes and weight management treatment. These include SGLT-2 inhibitors, DPP-4 inhibitors, insulin therapies, and emerging metabolic drugs with distinct mechanisms of action. Lifestyle interventions, bariatric surgery, and medical nutrition therapy also serve as non-pharmacological substitutes, particularly in obesity management. However, GLP-1 therapies often demonstrate superior weight loss and cardiometabolic benefits, strengthening their competitive positioning. Pricing differences and reimbursement availability can influence physicians' and patients' preferences toward substitute therapies. Despite the presence of substitutes, the unique efficacy profile of GLP-1 agents continues to support strong market demand.
Geographical expansion in the GLP-1 receptor agonist market is driven by rising diabetes prevalence, increasing obesity rates, and improving healthcare access in emerging economies. North America currently holds the largest market share due to strong reimbursement systems, high awareness, and early adoption of innovative therapies. Europe represents another significant market supported by an established healthcare infrastructure and regulatory approvals. Asia-Pacific is expected to witness the fastest growth due to large patient populations, urbanization, and expanding middle-class income levels. Pharmaceutical companies are actively pursuing regulatory approvals and partnerships to strengthen their presence in developing regions such as Latin America and the Middle East. Strategic expansion efforts are also focused on improving affordability and distribution networks to capture untapped market potential.
Product Insights
The semaglutide (Ozempic, Wegovy, Rybelsus) segment dominated the GLP-1 receptor agonist industry, accounting for the largest revenue share of 52.83% in 2025, driven by strong clinical efficacy in glycemic control and weight management, as well as expanding therapeutic applications. The semaglutide product segment includes GLP-1 receptor agonist formulations approved for the treatment of type 2 diabetes and obesity, administered by injection or orally to support metabolic control and sustained weight reduction. In 2025, semaglutide received expanded regulatory recognition beyond its core indications, including approval for injectable semaglutide (Wegovy) for the treatment of metabolic dysfunction-associated steatohepatitis with liver fibrosis in adults, significantly broadening its clinical utility. In February 2025, supply shortages for semaglutide products were resolved, reflecting stabilization of production and distribution capabilities. Improved product availability supported treatment continuity for patients and enhanced overall market access. These factors collectively contributed to the segment’s dominant revenue position.
The tirzepatide (mounjaro, zepbound) segment is projected to grow at the fastest CAGR of 13.9% over the forecast period, fueled by strong clinical outcomes in both glycemic control and weight reduction, as well as increasing adoption across metabolic treatment pathways. The tirzepatide product segment comprises dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist formulations indicated for the treatment of type 2 diabetes mellitus and chronic weight management, administered via subcutaneous injection to support metabolic improvement. Tirzepatide received regulatory approval in May 2022 for improving glycemic control in adults with type 2 diabetes, following evaluation of clinical safety and efficacy data. In November 2023, regulatory approval was expanded to include chronic weight management in adults with obesity or overweight and at least one weight-related comorbidity, based on clinical trial outcomes demonstrating significant body weight reduction. These expanding indications and strong therapeutic performance are supporting rapid market growth. Increasing physician confidence and patient demand are expected to further accelerate segment expansion.
Application Insights
The type 2 diabetes mellitus segment dominated the GLP-1 receptor agonist market, accounting for the largest revenue share of 81.22% in 2025, driven by the high global disease burden and strong clinical adoption of GLP-1 receptor agonists for glycemic management and cardiometabolic risk reduction. Over the past five years, the American Diabetes Association has revised treatment algorithms based on improved understanding of disease mechanisms and therapeutic profiles, resulting in broader recommendations for GLP-1 receptor agonists across multiple clinical scenarios. This class of therapies has gained prominence due to its effectiveness in glucose control, along with favorable weight and cardiovascular outcomes. In July 2024, Amylyx Pharmaceuticals announced the acquisition of a Phase 3-ready GLP-1 receptor antagonist, Avexitide, which holds FDA Breakthrough Therapy Designation for conditions associated with hyperinsulinemic hypoglycemia. The therapy has received Rare Pediatric Disease and Orphan Drug designations for congenital hyperinsulinism and related conditions, with Phase 3 development planned for 2025. These ongoing clinical advancements continue to strengthen innovation within the broader diabetes treatment landscape.
The obesity / chronic weight management segment is projected to grow at the fastest CAGR of 25.3% over the forecast period, owing to the rapidly increasing global prevalence of overweight and obesity and rising demand for effective pharmacological interventions. In 2022, more than 2.5 billion adults were classified as overweight, including 890 million living with obesity, while childhood obesity affected hundreds of millions globally, highlighting the scale of the health challenge. Adult obesity has more than doubled, and adolescent obesity has increased nearly fourfold since 1990, creating significant clinical demand for long-term weight management therapies. Pharmaceutical companies are intensifying development efforts to address this need, with multiple pipeline candidates targeting metabolic pathways involved in weight regulation. Pfizer is advancing several obesity-focused therapies, including danuglipron, which has demonstrated promising efficacy in clinical evaluation. A potential once-daily oral formulation could enhance patient convenience and strengthen competition within the oral GLP-1 receptor agonist market.
Route of Administration Insights
The injectable / parenteral segment dominated the market with the largest revenue share of 69.19% in 2025 due to strong clinical efficacy, established physician familiarity, and widespread use in both diabetes and obesity management. Injectable GLP-1 receptor agonists are administered by injection and mimic the endogenous incretin hormone GLP-1, stimulating insulin secretion in response to meals, suppressing glucagon release, slowing gastric emptying, and promoting satiety, thereby improving glycemic control and supporting weight reduction. Common therapies within this segment include liraglutide, exenatide, semaglutide, and dulaglutide, each offering different dosing schedules. Treatment options range from daily injections to once-weekly formulations, enhancing patient convenience. The introduction of long-acting formulations has significantly improved adherence and treatment persistence. These clinical and convenience advantages
The oral segment is projected to grow at a CAGR of 5.5% over the forecast period, driven by increasing patient preference for non-injectable treatment options and advancements in drug-delivery technologies. Oral GLP-1 receptor agonists represent a significant advancement in type 2 diabetes management by providing a convenient alternative to injectable therapies while maintaining similar mechanisms of action, including enhanced insulin secretion, reduced glucagon release, delayed gastric emptying, and improved satiety. These effects contribute to improved glycemic control and support weight management outcomes. The first approved oral GLP-1 receptor agonist, semaglutide (Rybelsus), received regulatory approval in 2019 as a once-daily tablet formulation. The availability of an oral option has increased acceptance among patients hesitant to initiate injectable therapy. Positive clinical outcomes demonstrating effective glucose reduction and weight benefits are expected to support continued segment growth.
Distribution Channel Insights
The retail pharmacies segment dominated the GLP-1 receptor agonist market, accounting for the largest revenue share of 54.74% in 2025, driven by wide accessibility, established patient purchasing behavior, and strong prescription volumes for diabetes and obesity treatments. Retail pharmacies remain primary dispensing points for GLP-1 receptor agonists, supporting convenient medication access and ongoing therapy management. As of July 2024, major pharmacy chains experienced lower-than-expected profitability amid high demand and challenges in maintaining adequate stock levels for GLP-1 medications. The surge in prescriptions, particularly for semaglutide products used for glycemic control and weight management, put pressure on supply across retail channels. European regulatory authorities identified ongoing shortages as a major public health concern, highlighting a continued supply-demand imbalance. Despite these challenges, retail pharmacies maintained dominance due to their broad reach and patient reliance on outpatient medication access.

The online pharmacies segment is projected to grow at the fastest CAGR of 21.2% over the forecast period due to increasing digital healthcare adoption, the convenience of home delivery, and the rising demand for chronic disease medications. The market for GLP-1 receptor agonists through online pharmacies has expanded significantly as the popularity of semaglutide therapies for diabetes and weight management grows. By December 2023, more than 2.5 million prescriptions for semaglutide were filled in the United States, reflecting strong patient demand and treatment adoption. High out-of-pocket costs for GLP-1 therapies have prompted patients to explore alternative purchasing channels that may offer lower prices. Online platforms also reduce barriers related to prescription access and pharmacy visits. These factors collectively support the rapid growth of the online distribution segment.
Regional Insights
North America held the largest share of the GLP-1 receptor agonist market at 75.46% in 2025, driven by the high prevalence of obesity and type 2 diabetes, as well as strong adoption of innovative therapeutics. Advanced healthcare infrastructure enables early diagnosis and treatment initiation across patient populations. High awareness among physicians regarding cardiometabolic benefits supports prescription growth. The presence of major pharmaceutical manufacturers strengthens product availability and commercialization efficiency. Favorable insurance coverage for metabolic disorders contributes to treatment accessibility. Continuous product launches and expanding indications further reinforce regional market leadership.
U.S. GLP-1 Receptor Agonist Market Trends
The U.S. GLP-1 receptor agonist industry dominates North America in 2025, driven by the significant demand for obesity management therapies. High healthcare spending allows broad access to premium-priced biologic medications. Strong clinical research activity accelerates the adoption of next-generation incretin-based drugs. Physicians' preference for therapies with proven cardiovascular outcomes increases prescribing rates. Direct-to-consumer marketing enhances patient awareness and treatment demand. The expanding population with metabolic syndrome continues to create sustained growth opportunities.
Europe GLP-1 Receptor Agonist Market Trends
Europe holds a substantial share of the GLP-1 receptor agonist industry, supported by the rising incidence of metabolic disorders and an aging population. Well-established healthcare systems promote structured diabetes management and long-term adherence to treatment. Clinical guidelines recommend the use of GLP-1 therapies for high-risk patients. The rising focus on weight management as part of chronic disease care is driving market expansion. Pharmaceutical companies are actively launching new products across multiple European countries. Demand for therapies with improved safety and efficacy profiles continues to rise across the region.
The GLP-1 receptor agonist market in the UK is expanding due to increasing obesity prevalence and demand for advanced diabetes therapies. Healthcare providers emphasize treatments with proven cardiovascular and weight reduction benefits. Growing patient awareness regarding metabolic health drives consultation rates and therapy uptake. Adoption of once-weekly formulations improves adherence among patients with chronic diseases. Clinical recommendations supporting incretin-based therapies contribute to the expansion of prescriptions. The availability of newer treatment options enhances overall market development.
The Germany GLP-1 receptor agonist market represents a key market in Europe, driven by a large diabetic population and strong healthcare spending capacity. Physicians increasingly prescribe GLP-1 receptor agonists for patients who require improved glycemic control and weight management benefits. The country’s advanced pharmaceutical distribution networks support efficient product availability. Demand for innovative injectable therapies continues to increase across specialized care centers. Aging demographics contribute to the rising incidence of metabolic disorders requiring long-term treatment. The continuous introduction of advanced formulations further supports market growth.
The GLP-1 receptor agonist market in France demonstrates consistent demand due to the rising prevalence of obesity and chronic metabolic conditions. Healthcare professionals are increasingly focusing on therapies that provide multi-system benefits, including cardiovascular risk reduction. Awareness regarding lifestyle-related diseases contributes to earlier treatment adoption. Expanding access to advanced incretin therapies strengthens physicians' confidence in prescribing. Long-term disease management strategies encourage the use of effective glucose-lowering agents. Continuous clinical evidence supporting therapeutic outcomes drives market expansion.
Asia Pacific GLP-1 Receptor Agonist Market Trends
Asia Pacific is experiencing rapid growth in the GLP-1 receptor agonist industry, driven by large patient populations and increasing urbanization. Rising disposable income levels improve access to advanced metabolic treatments across developing economies. Growing awareness of obesity-related complications contributes to higher diagnosis and treatment rates. Healthcare providers are adopting innovative therapies to better manage complex metabolic disorders. Pharmaceutical companies are expanding distribution channels to capture emerging opportunities. The increasing prevalence of sedentary lifestyles continues to fuel long-term demand.
The Japan GLP-1 receptor agonist market holds a significant position, supported by a well-established healthcare system and a strong focus on diabetes management. Physicians emphasize therapies that provide glycemic control with minimal risk of hypoglycemia. Aging population trends contribute to increasing metabolic disease burden requiring pharmacological intervention. High acceptance of injectable therapies supports patient adherence to treatment. Pharmaceutical innovation and clinical research activity remain strong in the country. Demand for therapies with proven safety profiles continues to expand.
The GLP-1 receptor agonist market in China is emerging as a major growth region, driven by a rapidly increasing diabetic population and lifestyle changes. Urbanization and dietary transitions contribute to a higher incidence of obesity-related disorders. Expanding healthcare infrastructure improves access to diagnosis and treatment in urban regions. Rising middle-class income levels support the adoption of advanced therapeutic options. Domestic pharmaceutical companies are investing in the development and commercialization of metabolic drugs. Increasing awareness regarding chronic disease management continues to accelerate market growth.
Latin America GLP-1 Receptor Agonist Market Trends
Latin America is projected to register the fastest CAGR of 14.6% over the forecast period, driven by rising diabetes prevalence and improved healthcare access. Growing urban populations contribute to a higher incidence of obesity and metabolic syndrome. Physicians are adopting newer incretin therapies to address unmet treatment needs across the region. Expansion of private healthcare services improves patient access to innovative medications. Pharmaceutical companies are strengthening regional distribution networks to capture emerging demand. Rising awareness regarding long-term complications of diabetes supports therapy adoption.
The Brazil GLP-1 receptor agonist market is the largest within Latin America, supported by a significant diabetic population. Rising obesity prevalence increases demand for effective weight management therapies. Urban healthcare centers are increasingly adopting innovative injectable treatments. Private healthcare utilization contributes to higher access to advanced medications. Physician familiarity with GLP-1 clinical benefits supports steady prescription growth. The country continues to expand as awareness of treatment improves and patient demand grows.
Middle East & Africa GLP-1 Receptor Agonist Market Trends
The Middle East and Africa GLP-1 receptor agonist industry is growing gradually, driven by the rising prevalence of metabolic diseases among urban populations. Rising awareness regarding diabetes complications encourages demand for advanced therapeutic options. Expanding healthcare infrastructure in key countries improves access to treatment. Pharmaceutical companies are entering regional markets to capture long-term growth opportunities. The growing focus on lifestyle-related health risks is contributing to therapy adoption. Market expansion remains moderate with improving healthcare access.
The Saudi Arabia GLP-1 receptor agonist market is significant within the Middle East, due to the high prevalence of obesity and diabetes among the population. The growing demand for advanced metabolic therapies supports the adoption of GLP-1 therapies in clinical settings. Healthcare providers emphasize treatments that improve glycemic control and weight outcomes. Rising patient awareness of chronic disease management contributes to increased prescription volume. Availability of innovative injectable formulations improves treatment adherence. The market continues to expand amid rising healthcare utilization and disease burden.
Key GLP-1 Receptor Agonist Company Insights
Novo Nordisk A/S and Eli Lilly and Company hold significant positions in the GLP-1 receptor agonist market, supported by strong incretin portfolios and continuous product innovation. Sanofi focuses on diabetes therapies and combination treatment approaches to strengthen its presence. AstraZeneca advances cardiometabolic research through strategic development initiatives. Teva Pharmaceutical Industries Ltd. explores opportunities in complex generics and peptide therapeutics. Glenmark Pharmaceuticals contributes through biosimilar development and expansion into emerging markets. The market reflects intense competition driven by innovation, expanding indications, and rising global demand for obesity and diabetes treatments.
Key GLP-1 Receptor Agonist Companies:
The following key companies have been profiled for this study on the GLP-1 receptor agonist market
- Eli Lilly and Company
- Sanofi
- Novo Nordisk A/S
- AstraZeneca
- Teva Pharmaceutical Industries Ltd.
- Glenmark Pharmaceuticals
Recent Developments
-
In June 2024, Teva Pharmaceutical Industries Ltd. launched an authorized generic of Victoza (liraglutide injection 1.8mg) in the U.S., marking the first generic GLP-1 product available in this market. This move enhances Teva’s complex generics portfolio and addresses the growing demand for GLP-1 therapies.
-
In March 2024, Novo Nordisk’s Wegovy (semaglutide) received FDA approval to reduce the risk of serious cardiovascular events, including heart attack, stroke, and cardiovascular death, in adults with cardiovascular disease and obesity or overweight. This makes Wegovy the first weight loss medication approved for this purpose. The approval is based on clinical trial results showing that Wegovy significantly lowers the risk of major cardiovascular events compared to placebo.
-
In January 2024, Glenmark Pharmaceuticals launched the Lirafita, a biosimilar of liraglutide, in India. Priced at around USD 1.19, it offers a 70% cost reduction for type 2 diabetes treatment. Lirafit improves glycemic control and is supported by clinical trials demonstrating its efficacy, safety, & benefits for weight management and cardiovascular health.
GLP-1 Receptor Agonist Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 82.01 billion
Revenue forecast in 2033
USD 185.32 billion
Growth rate
CAGR of 12.4% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, route of administration, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Eli Lilly and Company; Sanofi; Novo Nordisk A/S; AstraZeneca; Teva Pharmaceutical Industries Ltd.; Glenmark Pharmaceuticals.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global GLP-1 Receptor Agonist Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global GLP-1 receptor agonist market report based on product, application, route of administration, distribution channel, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Semaglutide (Ozempic, Wegovy, Rybelsus)
-
Tirzepatide (Mounjaro, Zepbound)
-
Dulaglutide (Trulicity)
-
Liraglutide (Victoza, Saxenda)
-
Others (Byetta, Bydureon, SOLIQUA)
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Type 2 Diabetes Mellitus
-
Obesity / Chronic Weight Management
-
Cardiovascular Risk
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Injectable / Parenteral
-
Oral
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global GLP-1 receptor agonist market size was estimated at USD 66.38 billion in 2025 and is projected to reach USD 82.01 billion in 2026.
b. The global GLP-1 receptor agonist market is expected to grow at a compound annual growth rate (CAGR) of 12.4% from 2026 to 2033, reaching approximately USD 185.32 billion by 2033.
b. Based on product, the semaglutide (Ozempic, Wegovy, Rybelsus) segment dominated the market with the largest revenue share of 52.83% in 2025. This growth is driven by strong clinical efficacy in glycemic control and weight management, expanding therapeutic applications, and increasing adoption for metabolic disorders including type 2 diabetes and obesity.
b. Key players operating in the market include Eli Lilly and Company, Sanofi, Novo Nordisk A/S, AstraZeneca, Teva Pharmaceutical Industries Ltd., and Glenmark Pharmaceuticals. These companies focus on product innovation, expanding indications, and strategic collaborations to strengthen their presence in the global GLP-1 receptor agonist market.
b. The market growth is driven by the rising global prevalence of type 2 diabetes and obesity, increasing demand for effective weight management therapies, and growing awareness regarding metabolic disease management. Advancements in peptide engineering, oral drug delivery technologies, and the development of dual and triple agonist therapies are further supporting innovation and market expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










