- Home
- »
- Medical Devices
- »
-
Guidewires Market Size And Share, Industry Report, 2033GVR Report cover
![Guidewires Market Size, Share & Trends Report]()
Guidewires Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Coronary Guidewires, Peripheral Guidewires, Urology Guidewires, Neurovascular Guidewires), By Region (North America, Europe, APAC, Latin America, MEA), And Segment Forecasts
- Report ID: 978-1-68038-243-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
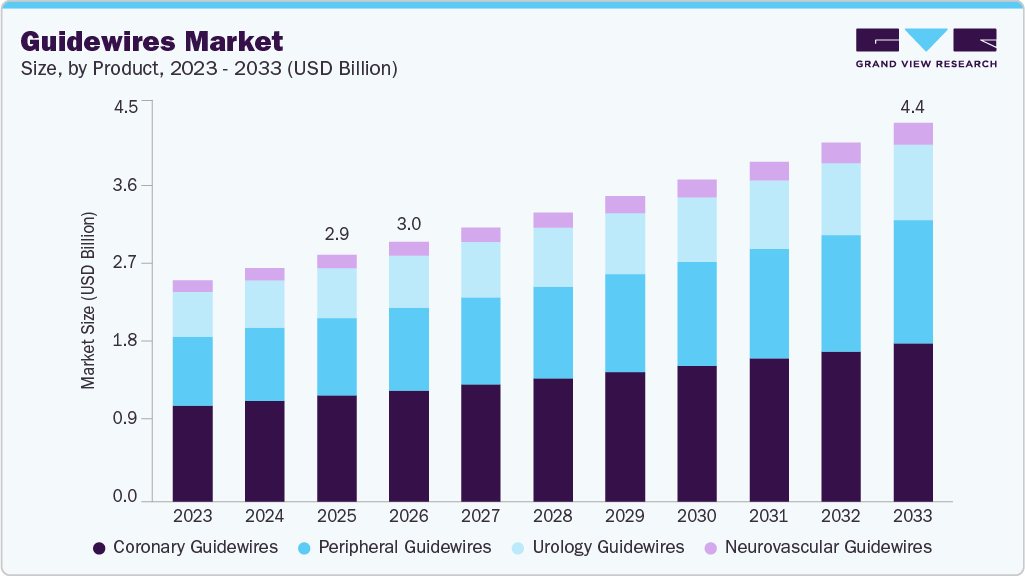
Guidewires Market Summary
The global guidewires market size was valued at USD 2.88 billion in 2025 and is projected to reach USD 4.43 billion by 2033, growing at a CAGR of 5.53% from 2026 to 2033. The growth of the guidewires market is driven by the rising volume of minimally invasive and catheter-based procedures, increasing prevalence of cardiovascular and peripheral vascular diseases, continuous advancements in guidewire design and materials to improve flexibility and torque control, and expanding adoption of interventional procedures across hospitals and ambulatory surgical centers worldwide.
Key Market Trends & Insights
- North America dominated the guidewires market with the largest revenue share of 34.80% in 2025.
- The U.S. guidewires market is growing, primarily driven by rising procedural volumes in interventional cardiology and peripheral vascular interventions, supported by an aging population and increasing prevalence of cardiovascular diseases.
- By product, the coronary guidewires segment accounted for the largest market share of 42.93% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.88 Billion
- 2033 Projected Market Size: USD 4.43 Billion
- CAGR (2026-2033): 5.53%
- North America: Largest market in 2025
The rising global burden of cardiovascular diseases, including coronary artery disease, peripheral artery disease, and structural heart disorders, is one of the significant growth drivers for the guidewires market. As the incidence of CVD increases due to ageing populations, sedentary lifestyles, obesity, diabetes, and hypertension, hospitals are witnessing a higher volume of diagnostic and interventional procedures such as angioplasty, stent placement, catheter-based imaging, and minimally invasive vascular repairs. These procedures rely heavily on guidewires for navigating complex vasculature, crossing occlusions, and enabling precise device placement. Moreover, the growing preference for minimally invasive interventions, favored for shorter hospital stays, reduced complications, and better patient outcomes, further accelerates the demand for highly specialized guidewires.

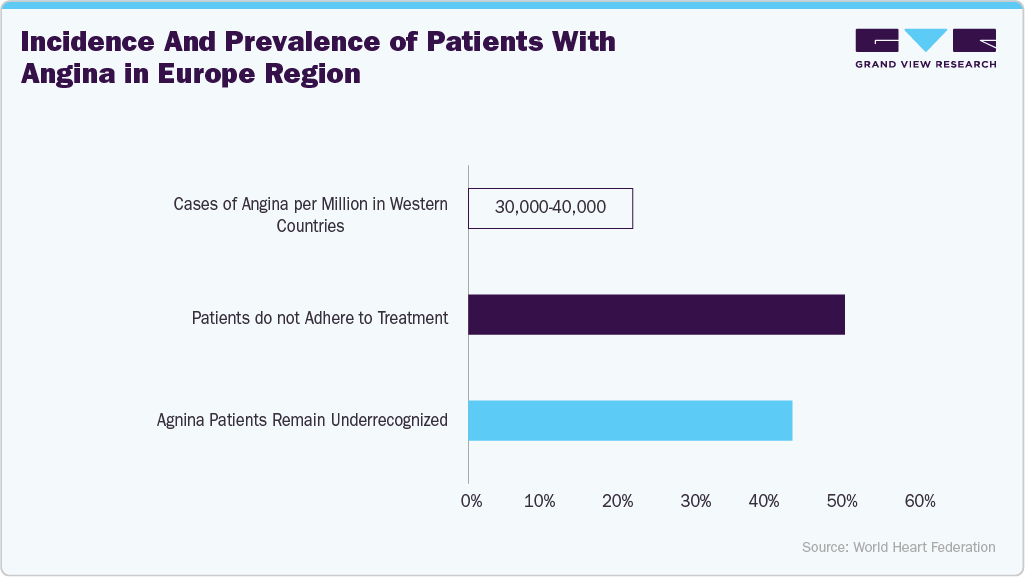
Moreover, angina pectoris, a leading cause of morbidity and mortality globally, affects a significant number of individuals. For instance, according to the National Library of Medicine, the prevalence of angina pectoris worldwide ranged from approximately 2.0% to 7.0% in 2024. In addition, in 2023, there were over 2,806,000 reported cases of angina pectoris in China. There is evidence that angina doubles the risk of serious cardiovascular events. Currently, coronary artery disease (CAD) accounts for 4.1 million deaths in Europe annually, with individuals over 65 accounting for 82.0% of these deaths.
Technological advancements are one of the key factors boosting the application of the guidewire. The advanced guidewires can be customized for specific requirements and can be operated remotely. This leads to an increased success rate of the procedures and thus the adoption rate for minimally invasive surgeries. The integration of these guidewires with imaging modalities and surgical robots has endless applications in modern surgical practices. For instance, in April 2024, Teleflex Incorporated introduced the Wattson Temporary Pacing Guidewire, designed specifically for use during Balloon Aortic Valvuloplasty (BAV) and Transcatheter Aortic Valve Replacement (TAVR). This guidewire offers supporting valve delivery, dual functionality, and ventricular bipolar pacing. It aims to improve procedural efficiencies and reduce complications associated with traditional methods.
Furthermore, rising geriatric population further fuels market growth. For instance, the WHO estimates that by 2030, 1 in 6 people worldwide will be 60 years of age or older. The same demographic will double by 2050. Moreover, diseases are far more common among older adults. These include cardiovascular disease, ischemic heart disease, vascular dementia, and multiple sclerosis, among others.
In 2023, findings from the Institute for Health Metrics and Evaluation study on the global burden of cardiovascular diseases and risks for the period 1990-2022 reveal significant insights.
-
Age-standardized cardiovascular disease (CVD) mortality rates varied regionally, ranging from 73.6 per 100,000 in High-income Asia Pacific to 432.3 per 100,000 in Eastern Europe in 2022.
-
Global CVD mortality demonstrated a significant decrease by 34.9% from 1990 to 2022.
-
Leading Causes of Disability-Adjusted Life Years (DALYs): ischemic heart disease emerged as the leading global cause with age-standardized DALYs at 2,275.9 per 100,000. Intracerebral hemorrhage and ischemic stroke followed closely as prominent CVD causes for age-standardized DALYs.
-
Age-Standardized CVD Prevalence: Varied globally, with rates ranging from 5,881.0 per 100,000 in South Asia to 11,342.6 per 100,000 in Central Asia.
- Leading Risk Factor: High systolic blood pressure accounted for the largest number of attributable age-standardized CVD DALYs at 2,564.9 per 100,000 globally.
The expansion of hospital infrastructure, particularly the increase in the number of advanced catheterization laboratories (cath labs), is a key driver for the guidewires market. As hospitals and specialized cardiac care centers invest in modern facilities and equipment to meet the increasing demand for minimally invasive procedures, the need for high-quality guidewires grows in proportion. Catheterization labs are essential for diagnostic and interventional procedures such as angioplasty, stent placement, and peripheral vascular interventions, all of which require precise and reliable guidewires to navigate blood vessels safely. Moreover, government initiatives and private investments aimed at upgrading healthcare infrastructure in emerging and developed markets have led to a surge in the number of operational cath labs, enabling more complex procedures and increasing procedural volumes.
Recent Expansions and Upgrades in Catheterization Laboratories (2025)
Month & Year
Facility Name
Country
Description
Dec-25
Jabil - Advanced Catheter Development Lab (St. Petersburg, Florida)
U.S.
Jabil opened a next-generation catheter development lab supporting rapid prototyping, device engineering, and clinical innovation. Works with University of Florida and USF to accelerate minimally invasive catheter technologies.
Sep-25
SSM Health Good Samaritan Hospital - Upgraded Cardiac Cath Lab (Mt. Vernon, Illinois)
U.S.
Hospital unveiled its newly upgraded cath lab following a USD 1 million investment. Features advanced imaging and interventional systems enabling faster diagnosis and safer treatment for cardiac emergencies. It is the only cath lab serving nine counties, improving access to critical cardiovascular care.
Sep-25
Samuel Fineman Cardiac Catheterization AI Research Lab - Mount Sinai Hospital (New York)
U.S.
New AI-powered cath lab aimed at integrating advanced algorithms for risk stratification, workflow optimization, and outcome prediction in interventional cardiology. Led by Dr. Annapoorna Kini.
Feb-25
Surrey Memorial Hospital - Two New Cardiac Cath Labs
Canada
Construction began for two advanced cath labs featuring full imaging suites (FPDs, angioplasty systems, fluoroscopy). Part of a USD 10M regional cardiovascular upgrade, with USD 9M raised. Designed to address rising cardiac cases in the Fraser Health region.
14-Jan-25
Texas Heart Institute - New Research-Dedicated Catheterization Laboratory
U.S.
THI launched a new research-focused cath lab equipped with the Philips Allura Xper FD10 system. Offers reduced radiation exposure, digital flat detector imaging, and compact design to enhance procedural precision for minimally invasive cardiovascular research.
Source: Press release, GVR analysis
Market Concentration & Characteristics
The guidewires market is characterized by a growing concentration driven by growing technological advancements and increasing healthcare demand for continuous medicinal solutions. The market is marked by significant R&D investments, collaborations, and product innovations, ensuring robust growth. Companies focus on developing user-friendly, non-invasive, and highly accurate platforms for various online pharmacies.
The guidewire market is characterized by continuous technological advancement aimed at improving performance, safety, and clinical outcomes. Recent innovations focus on enhancing materials and design, such as the use of Nitinol and other alloys for superior flexibility, kink resistance, torque control, and surface coatings (hydrophilic or polymer) that reduce friction and improve navigability through complex vascular anatomy. There is a growing trend toward smart guidewires that integrate with advanced imaging modalities and sensor technologies to provide real-time feedback on position, pressure, and vessel characteristics. These innovations enable clinicians to make more informed decisions during complex procedures and enhance procedural accuracy. In October 2022, researchers at the Hong Kong University of Science and Technology (HKUST) developed an AI-based guidewire recognition and counting technology to reduce the risk of guidewire retention in patients’ bodies after a clinical procedure known as central venous catheter insertion.
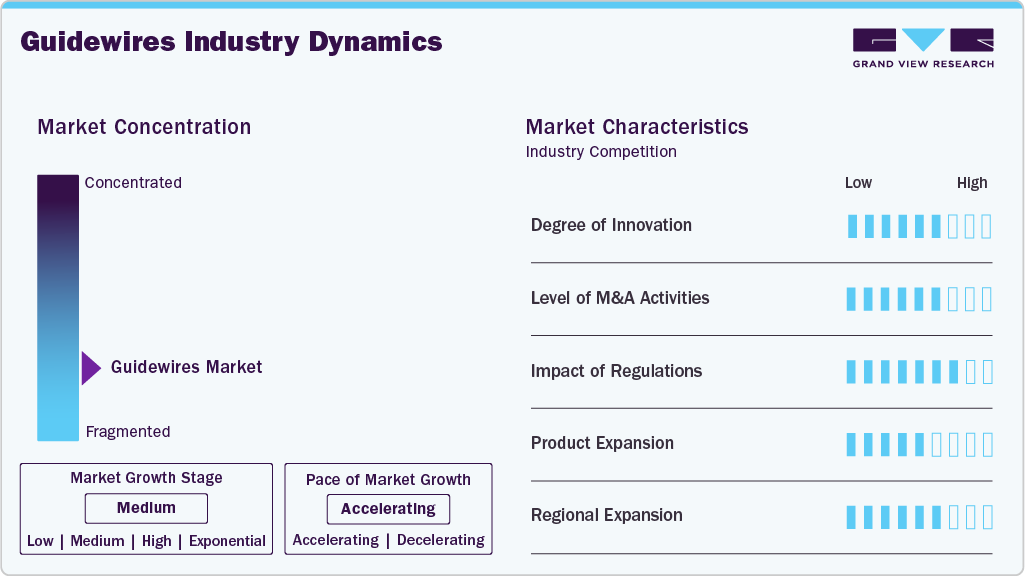
Regulations have a significant impact on the guidewire market by ensuring the safety, performance, and clinical reliability of products. Regulatory authorities require strict compliance with biocompatibility standards, manufacturing quality systems, and clinical validation, which increases trust among clinicians and patients. However, these requirements also raise development costs and extend approval timelines, particularly for highly innovative or novel guidewire designs.
Mergers and acquisitions in the guidewires industry are rising due to the need for R&D, reflecting the industry's dynamic nature. Companies are leveraging M&A activities to innovate and offer advanced solutions that meet the evolving needs of healthcare professionals. For instance, in June 2024, Silk Road Medical announced a definitive agreement to be acquired by Boston Scientific. This acquisition marks a significant development in the medical technology industry and is expected to have various implication for both companies involved.This strategic move is likely to expand Boston Scientific’s portfolio of minimally invasive treatment options for cardiovascular diseases and enable Silk Road Medical to leverage Boston Scientific’s commercial infrastructure to accelerate the adoption of its TCAR technology on a larger scale.
Guidewires market is leveraging technology to enhance the overall patient care services. In June 2023, Sensome announced a partnership with Asahi Intecc, a Japanese medical device company, to work on the development of the next-generation Clotild Smart Guidewire for stroke treatment. This partnership aims to combine Sensome’s expertise in sensor technology with Asahi Intecc’s experience in medical devices to create an innovative solution for better stroke management.
The companies are leveraging their combined expertise, resources, and technologies to further improve patient outcomes and advance the field of guidewires. For instance, in November 2022, Guidewire expanded its technology distribution into the London market. This move is driven by the belief that automation can bring significant efficiency to the London Market’s unique underwriting processes. Sheridon Glenn, Guidewire’s global vice president for strategic markets and initiatives, highlighted the complexity of risks underwritten in the London Market, emphasizing the need for specialists and multiple insurers throughout the process.
Product Insights
The coronary guidewires segment accounted for the largest market share of 42.93% in 2025. This dominance is driven by the increase in demand for minimally invasive coronary surgery and the increasing prevalence of cardiovascular disease. For instance, according to the FDA, in January 2023, heart disease is responsible for almost one in four deaths in the U.S. Moreover, according to the CDC, it was reported in May 2024 that approximately 25.0 million adults in the U.S. have total cholesterol levels above 240 mg/dL, which is considered high. High cholesterol and uncontrolled high blood pressure are both significant risk factors for cardiovascular disease, heart disease, stroke, and other detrimental conditions.
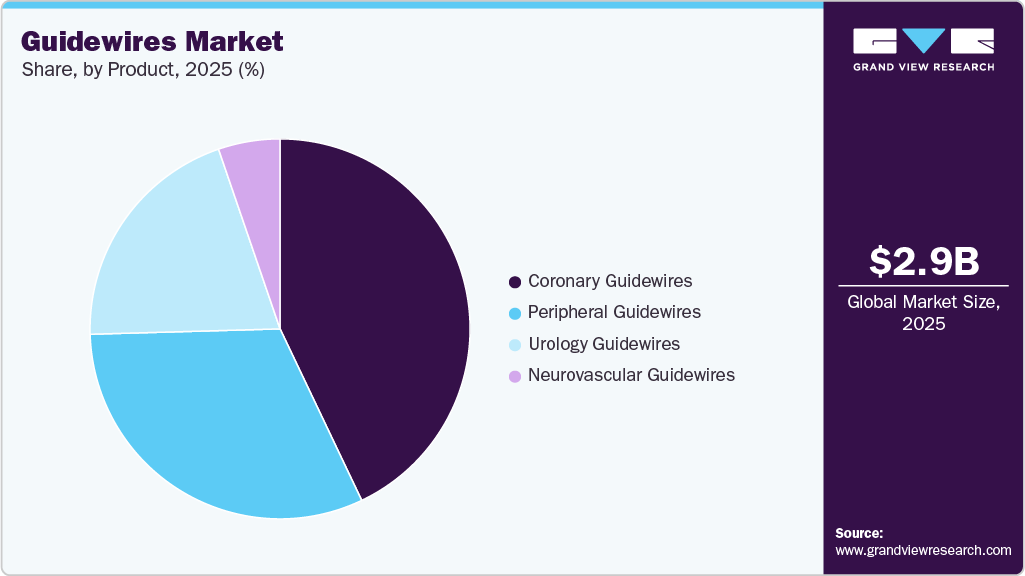
In addition, high blood pressure is among one of the heart disease leading causes of premature death on a global scale. For instance, as reported by WHO in March 2023, it is estimated that around 1.28 billion adults between the ages of 30 and 79 worldwide are affected by hypertension. Throughout the projection period, the segment's dominance is anticipated to be aided by the introduction of new technologies.
The neurovascular guidewires segment is expected to grow at the fastest CAGR over the forecast period. This rapid growth is majorly owed to an elevated risk of neurovascular conditions that impact the cerebellum, spinal cord, and brain vascular system, including aneurysms, cavernoma, arteriovenous malformations, dural arteriovenous fistula, cerebral hemorrhage, and stroke. The existence of vital blood vessels makes the treatment of such illnesses difficult.
Regional Insights
North America guidewires market accounted for the largest share of 34.80% in 2025. The growing preference for minimally invasive surgeries is driving regional demand. This is mainly attributed to the high prevalence of heart diseases and rise in the number of coronary and peripheral interventions. In January 2024, the world’s first partial heart transplant was conducted by a team at Duke Health. The transplant successfully achieved functioning valves and arteries that grow along with the young patient. This groundbreaking procedure marks a significant advancement in the field of cardiac surgery and offers hope for patients with complex heart conditions.
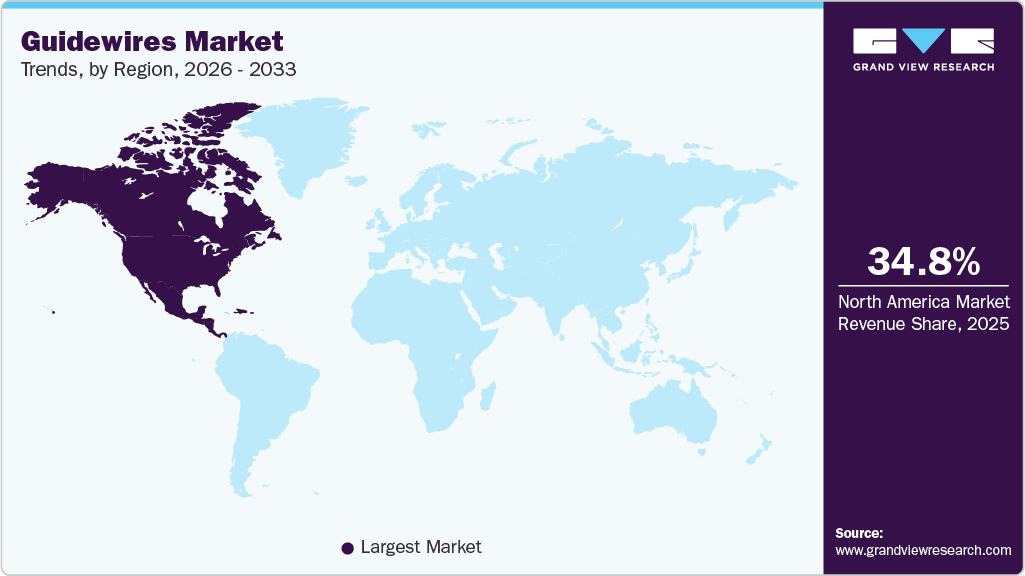
In addition, the presence of key players such as Abbott; Terumo Medical Corporation; B. Braun Melsungen AG; and Boston Scientific Corporation is also one of the indicators that depicts its current huge market potential. The growing prevalence of cardiovascular diseases and rising preference for minimally invasive surgeries in the U.S. are key drivers supporting the regional market's expansion.
U.S. Guidewires Market Trends
The guidewires market in the U.S. held a significant share of North America's guidewires market in 2025. The market’s growth is expected to be boosted by the increasing infertility rates in the country. For instance, as per the data published by CDC in May 2024, it is projected that in the U.S., approximately 19.0% of heterosexual women aged 15 to 49 years with no prior births are unable to conceive after one year of trying, while around 26.0% experience challenges in getting pregnant or maintaining a pregnancy. This suggests that the market for fertility treatments and services is poised for continued growth.
Europe Guidewires Market Trends
The Europe guidewires marketheld a significant share in 2025 due to increasing production of guidewires in the region. For instance, in November 2023, Integer emphasized its European expansion plans at the Medica/CompaMed event, particularly focusing on investments in its facilities in Ireland. The company has expanded its New Ross facility for guidewire manufacturing to meet the increasing demand for OEM and custom-developed guidewire products. This expansion includes specialized capabilities in complex delivery wire grinding and assembly to cater to markets such as neurovascular, structural heart, electrophysiology, and peripheral vascular. Integer is also enhancing its offerings in medical textiles, complex braiding, implant covering, and coatings like ePTFE to serve its European customer base better.
The UK guidewires market is experiencing growth, driven due to the presence of stringent regulations and guidelines in the country. Coronary heart disease is a significant driver of the UK guidewire market, primarily due to its high prevalence and the substantial volume of interventional cardiology procedures performed annually. CHD management frequently involves coronary angiography, percutaneous coronary intervention, and stent placement, all of which require guidewires as a core consumable.
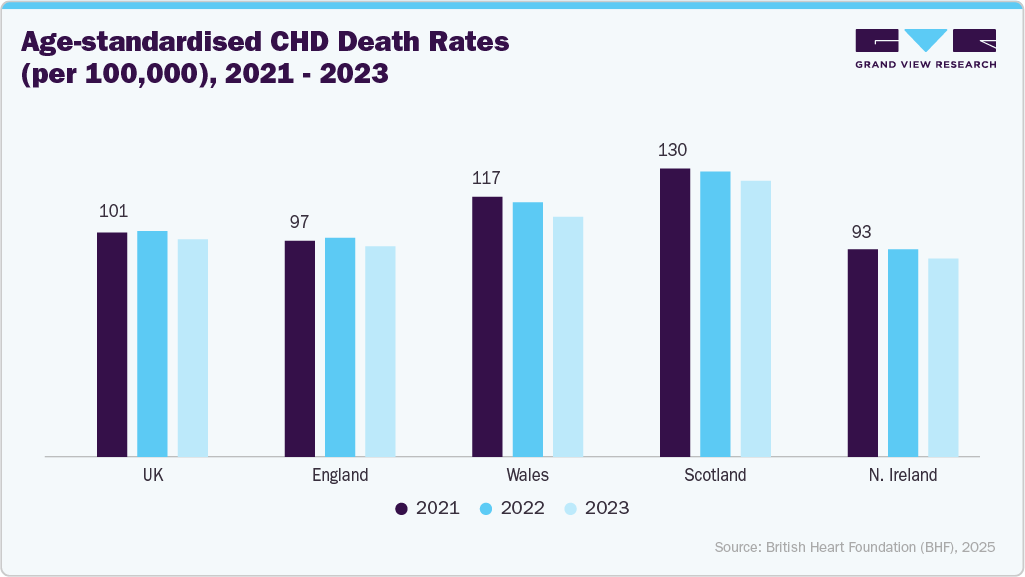
The guidewires market in Germany is evolving due to the rising aging population, growing prevalence of cardiovascular issues, presence of a sophisticated healthcare system, a highly qualified workforce, and high healthcare spending. According to the data published by Robert Koch Institute 2025, Coronary heart disease was the most common cause of death in Germany in 2023, with 119,800 deaths
Asia Pacific Guidewires Market Trends
The Asia Pacific Guidewires market is experiencing significant growth which is driven by the increasing prevalence of chronic diseases, rising geriatric population, and increasing healthcare expenditure. For instance, as per the JACC Journals report published in December 2023, the age-standardized cardiovascular disease mortality rates among countries in High-income Asia Pacific ranged from 72.7 to 252.6 per 100,000, representing a 3.5-fold difference in 2022. Furthermore, investments in healthcare infrastructure, favorable government initiatives, and untapped opportunities in emerging countries such as China and India also remain promising drivers of growth. Additionally, the market growth is also driven by a large geriatric population, high frequency of heart disease, and demand for advanced minimally invasive surgery techniques with faster results and higher accuracy.
The guidewires market in Japan is growing, driven by the increasing strategic initiatives by the market players. For instance, in July 2024, Sensome signed an exclusive commercial distribution agreement with Cosmotec, a prominent distributor in Japan. The agreement pertains to the distribution of Sensome’s innovative clot-sensing guidewire in the Japanese market. This partnership signifies a significant step forward in the availability and accessibility of advanced medical devices in Japan. By leveraging Cosmotec’s established distribution network and expertise in the region, Sensome aims to introduce its cutting-edge clot-sensing guidewire to healthcare providers and patients across Japan.
The China guidewires market is gaining traction for healthcare monitoring due to the increasing technological advancements in the country. For instance, in June 2024, MicroPort Coronary achieved a significant milestone with its Intravascular Piezoelectric Guidewire System gaining access to the “Green Path” in China. This regulatory pathway accelerates the approval process for innovative medical devices that address unmet clinical needs and offer significant advancements in patient care. The “Green Path” designation signifies that the Intravascular Piezoelectric Guidewire System meets the stringent criteria set forth by Chinese regulatory authorities for fast-track approval. This recognition underscores the system’s clinical value, safety profile, and potential to address critical needs within the healthcare landscape.
Latin America Guidewires Market Trends
The Latin American Guidewires market is experiencing lucrative growth rate due to the rising awareness regarding cardiovascular issues and treatments by conducting programs. For instance, according to the Society of Thoracic Surgeons, STS/EACTS/LACES Cardiovascular Surgery Conference, Latin America, is planned to be held in December 2024. The 2024 program is to showcase the latest advancements and best practices in coronary artery disease, congenital heart disease, thoracic aortic disease, atrial fibrillation, and the surgical treatment of heart failure.
The guidewires market in Brazil is poised for rapid growth owing to the increasing strategic initiatives by the market players in the country and growing application of guidewires in the country. For instance, in September 2023, the Reewarm PTX Drug Coated Balloon (DCB) Catheter, developed by Endovastec was introduced in Brazil with a successful implantation procedure. This milestone marks the continued expansion of the product into international markets, aiming to provide advanced treatment options for patients globally. The procedure was carried out by the medical team at Paulo Sacramento Hospital in Sao Paulo, Brazil. The positive outcomes observed from this initial implantation pave the way for future applications of the technology across different regions, contributing to enhanced patient care and treatment outcomes worldwide.
Middle East & Africa Guidewires Market Trends
The MEA guidewires market is fueled by the growing focus on improving healthcare infrastructure and access to advanced medical technologies in the MEA. Government initiatives in several countries are playing a crucial role. For instance, Saudi Arabia's Vision 2030 healthcare transformation plan emphasizes the adoption of innovative medical technologies. This, coupled with increasing investments in healthcare facilities, is creating a more receptive environment for guidewires, hence driving the Saudi Arabia Guidewires market.
Key Guidewires Company Insights
The guidewires market is highly competitive, with key players holding significant positions. The major companies are undertaking various organic as well as inorganic strategies, such as new product development, collaborations, acquisitions, mergers, and regional expansion, to serve the unmet needs of their customers.
Key Guidewires Companies:
The following are the leading companies in the guidewires market. These companies collectively hold the largest market share and dictate industry trends.
- Boston Scientific Corporation
- Abbott
- Cordis (Cardinal Health)
- AngioDynamics
- Medtronic
- Stryker
- Cook
- BD
- Codman Neuro (Integer Holdings Corporation)
- Terumo Medical Corporation
- Merit Medical Systems
- Olympus Corporation
- Teleflex Incorporated
- B. Braun
- Asahi Intecc Co., Ltd.
- ConMed Corporation
- Argon Medical Devices
- SP Medical A/S
- TE Connectivity (formerly Creganna Medical)
- Galt Medical Corp
- Balton
- Manish Medi Innovation
- ST. Stone Medical Devices Private Limited
- Custom Wire Technologies, Inc.
- ACE Medical Devices
- BrosMed Medical Co., Ltd.
- Cathwide Medical Co. Ltd.
- Creo Medical
- Foshan Dihua Technology Co., Ltd.
Recent Developments
-
In December 2025, Atraverse Medical announced that it had secured U.S. FDA 510(k) clearance for its fully integrated HOTWIRE Transseptal Access System, marking a significant milestone in left-heart access technology. The system combines the HOTWIRE RF Generator, which features impedance-guided shutoff to automatically stop energy delivery after transseptal crossing, with the HOTWIRE RF Guidewire, a zero-exchange guidewire offering universal sheath compatibility.
-
In October 2025, Medtronic plc announced the launch of the Stedi Extra Support Guidewire, a purpose-built guidewire designed to enhance the performance of the Evolut transcatheter aortic valve replacement (TAVR) system and compatible with all commercially available TAVR platforms.
-
In April 2025, Boston Scientific Corporation announced the launch of its Kinetix Guidewire, a next-generation coronary guidewire designed for use in percutaneous coronary intervention (PCI) procedures. The Kinetix Guidewire features a micro-cut nitinol sleeve and a nitinol distal core, enabling significantly improved torque control, flexibility, and maneuverability compared with conventional spring-coil guidewires.
-
In June 2024, Medtronic launched the Steerant Aortic Guidewire for EVAR and TEVAR procedures. The device is designed to facilitate catheter placement and exchange during diagnostic or interventional procedures in the aorta, providing support for endovascular aneurysm repair (EVAR) and thoracic EVAR (TEVAR) of abdominal aortic aneurysms.
Guidewires Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.04 billion
Revenue forecast in 2033
USD 4.43 billion
Growth rate
CAGR of 5.53% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Boston Scientific Corporation; Abbott; Cordis (Cardinal Health); AngioDynamics; Medtronic; Stryker; Cook; BD; Codman Neuro (Integer Holdings Corporation); Terumo Medical Corporation; Merit Medical Systems; Olympus Corporation; Teleflex Incorporated; B. Braun; Asahi Intecc Co., Ltd.; ConMed Corporation; Argon Medical Devices; SP Medical A/S; TE Connectivity (formerly Creganna Medical); Galt Medical Corp; Balton; Manish Medi Innovation; ST. Stone Medical Devices Private Limited; Custom Wire Technologies, Inc.; ACE Medical Devices; BrosMed Medical Co., Ltd.; Cathwide Medical Co. Ltd.; Creo Medical; Foshan Dihua Technology Co., Ltd
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Guidewires Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis on the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the guidewires market report on the basis of product, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Coronary Guidewires
-
Peripheral Guidewires
-
Urology Guidewires
-
Neurovascular Guidewires
-
-
Region Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
The global guidewires market size was estimated at USD 2.88 billion in 2025 and is expected to reach USD 3.04 billion in 2026.
The global guidewires market is expected to grow at a compound annual growth rate of 5.53% from 2026 to 2033 to reach USD 4.43 billion by 2033.
North America dominated the guidewires market with a share of 34.80% in 2025. This is attributable to the high prevalence of cardiovascular diseases and the rise in the number of coronary and peripheral interventions.
Some key players operating in the guidewires market include Boston Scientific Corporation, Abbott, Cordis (Cardinal Health), AngioDynamics, Medtronic, Stryker, Cook, BD, Codman Neuro (Integer Holdings Corporation), Terumo Medical Corporation, Merit Medical Systems, Olympus Corporation, Teleflex Incorporated, B. Braun, Asahi Intecc Co., Ltd., ConMed Corporation, Argon Medical Devices, SP Medical A/S, TE Connectivity (formerly Creganna Medical), Galt Medical Corp, Balton, Manish Medi Innovation, ST. Stone Medical Devices Private Limited, Custom Wire Technologies, Inc., ACE Medical Devices, BrosMed Medical Co., Ltd., Cathwide Medical Co., Ltd., Creo Medical, Foshan Dihua Technology Co., Ltd.
Key factors that are driving the market growth include a rising preference for minimally invasive surgical procedures owing to advantages such as smaller incisions, faster recovery time, and reduced pain.
About the Author(s)
Medical Devices Research Team
Healthcare · Medical DevicesThis report was authored by the medical devices Research Team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the medical devices segment of the healthcare industry. All findings are based on proprietary healthcare databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









