- Home
- »
- Medical Devices
- »
-
Hemodynamic Monitoring Devices Market Size Report, 2033GVR Report cover
![Hemodynamic Monitoring Devices Market Size, Share & Trends Report]()
Hemodynamic Monitoring Devices Market (2025 - 2033) Size, Share & Trends Analysis Report By System Type (Invasive, Minimally Invasive, Non-Invasive), By Product (Disposables, Monitors), By End Use, By Region, And Segment Forecasts
- Report ID: 978-1-68038-876-3
- Number of Report Pages: 190
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
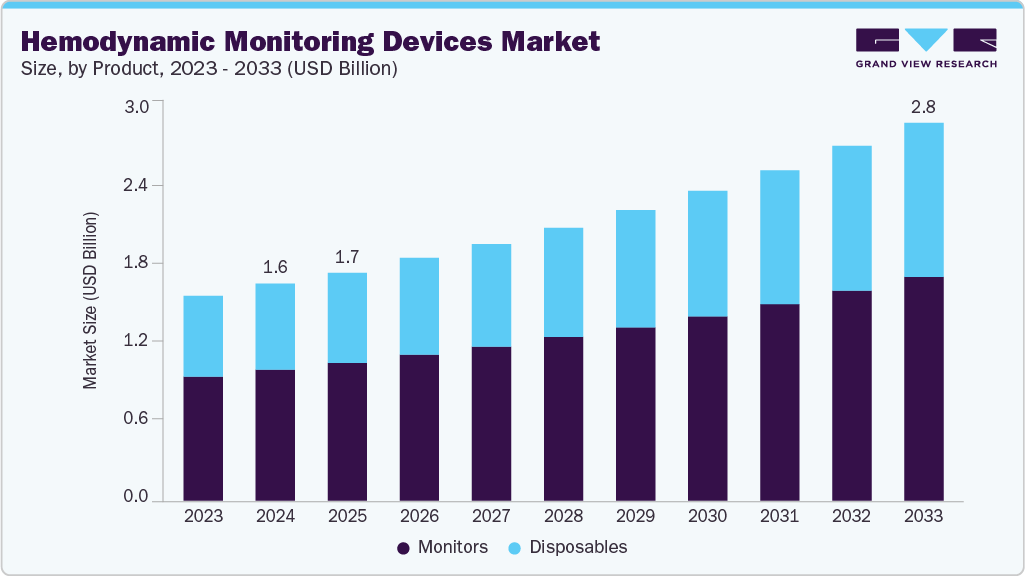
Hemodynamic Monitoring Devices Market Summary
The global hemodynamic monitoring devices market size was estimated at USD 1.58 billion in 2024 and is projected to reach USD 2.78 billion by 2033, growing at a CAGR of 6.5% from 2025 to 2033. The growth is driven by several key factors, such as the rising prevalence of cardiovascular diseases and critical illnesses, integration of AI and predictive analytics, growing demand for minimally invasive and non-invasive monitoring techniques, and technological advancements in monitoring devices, including real-time data and wireless connectivity.
Key Market Trends & Insights
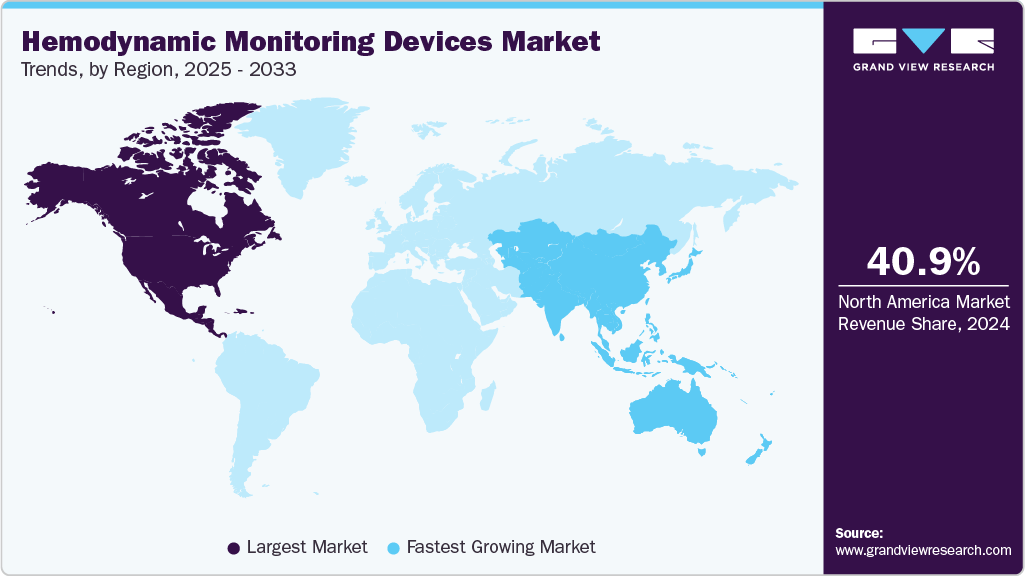
- North America dominated the hemodynamic monitoring devices market with a share of 40.99% in 2024.
- The Asia Pacific hemodynamic monitoring devices industry is estimated to witness the fastest CAGR from 2025 to 2033.
- By product, the monitors segment held the largest market share of 60.4% in 2024.
- By system type, the non-invasive segment held the largest market share in 2024.
- By end use, the hospitals segment held the dominant market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.58 Billion
- 2033 Projected Market Size: USD 2.78 Billion
- CAGR (2025-2033): 6.5%
- North America: Largest Market in 2024
- Asia Pacific: Fastest Growing Market
The increasing prevalence of CVDs and associated risk factors is anticipated to boost the demand for hemodynamic monitoring devices. Hemodynamic monitoring devices play an increasingly important role in assessing and reducing the risk of heart failure. Hemodynamic monitoring devices, such as implantable devices, provide objective data on factors like pulmonary artery pressure. This allows doctors to identify patients at higher risk for hospitalization or even death from heart failure. Heart failure is one of the leading causes of death across the globe; for instance, according to the CDC, around 6.7 million adults of 20 years and older in the U.S. have heart failure.
In addition, the demand in the hemodynamic monitoring devices industry is constantly growing due to the rising prevalence of Sudden Cardiac Arrests (SCA). Cardiac arrests are generally severe or fatal, and the survival rate in several countries is low, mainly in cases of out-of-hospital attacks. According to the Heart and Stroke Statistics 2022 by the American Heart Association, more than 356,000 out-of-hospital cardiac arrests occur in the U.S. every year, with around 90% of them resulting in death. Hence, the increasing prevalence of cardiac arrest is anticipated to boost the demand for pacemakers.
The aging process significantly changes the cardiovascular system, making careful hemodynamic management essential for older ICU patients. As people age, the balance of the autonomic nervous system gradually shifts: parasympathetic activity weakens while resting sympathetic activity increases. This imbalance heightens the risk of cardiovascular complications in elderly individuals.
At the same time, the global population is aging rapidly. According to the WHO, the number of people aged 60 and above is projected to rise from around 1 billion in 2020 to 1.4 billion by 2030, reaching approximately 2.1 billion by 2050. Consequently, the number of elderly patients experiencing critical illnesses has grown substantially in recent years, underscoring the increasing need for advanced hemodynamic monitoring in this vulnerable population.
Integration of AI in the Hemodynamic Monitoring Devices Market
AI in hemodynamic monitoring devices enhances real-time patient assessment by analyzing continuous physiological signals such as blood pressure, cardiac output, and oxygenation to provide predictive insights and early warnings of instability. These solutions reduce clinician workload through automated data interpretation, trend analysis, and personalized treatment guidance, ultimately improving accuracy and patient outcomes.
Leading players increasingly integrate AI-driven clinical decision support into their platforms to advance these benefits. For instance, in April 2025, BD launched the HemoSphere Alta, a cutting-edge hemodynamic monitoring platform featuring AI-powered predictive algorithms that enable clinicians to anticipate and manage blood pressure instability and optimize blood flow during critical care. The system includes a unique Cerebral Autoregulation Index (CAI) for personalized cerebral blood flow insights and an advanced Hypotension Prediction Index (HPI), coupled with an enhanced touchscreen and hands-free controls that streamline clinical workflows in surgical and intensive care environments.
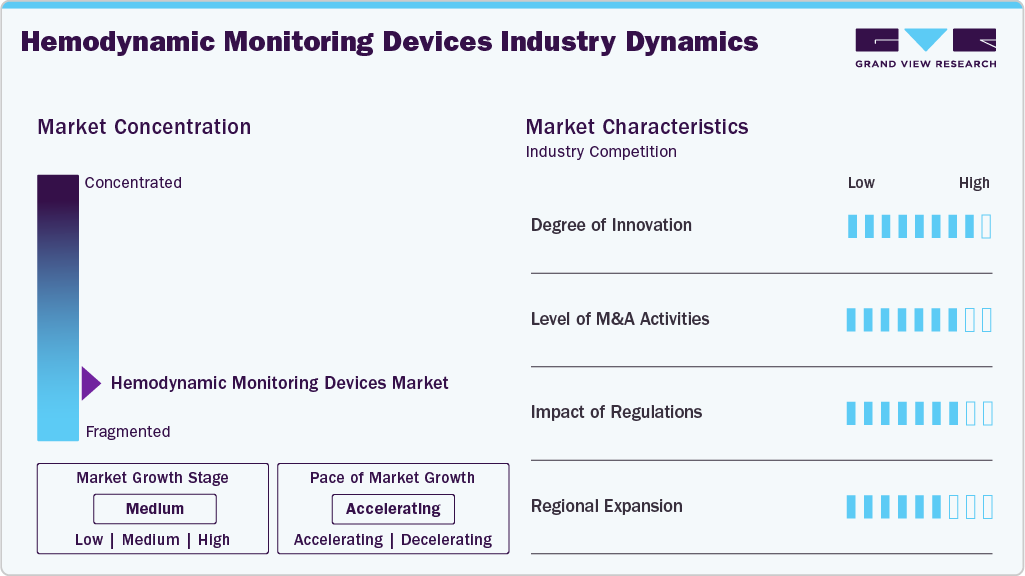
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, characteristics, and participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion. For instance, the hemodynamic monitoring devices market is fragmented, with many providers entering the market. The degree of innovation, the level of partnerships & collaboration activities, and the impact of regulations on the industry is high. However, the regional expansion observes moderate growth.
The degree of innovation in the hemodynamic monitoring devices industry is high, with players like Edwards Lifesciences, Philips, and GE HealthCare advancing minimally invasive and non-invasive technologies to improve accuracy, ease of use, and patient safety. The field is further evolving with BD’s integration of AI in its HemoSphere Alta platform, which marks a step toward predictive and intelligent clinical decision support in hemodynamic monitoring.
M&A activities support geographic expansion and market entry for companies. For instance, in September 2024, BD completed the USD 4.2 billion acquisition of Edwards Lifesciences' Critical Care product group, renaming it BD Advanced Patient Monitoring to expand its portfolio of smart connected care solutions, including advanced monitoring technologies and AI-enabled clinical decision tools. This strategic step boosts BD's capabilities in closed-loop hemodynamic monitoring and integrated infusion platforms, with the new unit located in Irvine, California.
“The health care industry is being redefined by AI, robotics, and autonomous solutions, and our team continues to accelerate the application of these new technologies to improve the quality and cost of patient care around the world. Today, BD is proud to welcome BD Advanced Patient Monitoring, whose leading technologies, AI-enabled solutions, and strong innovation pipeline expand our portfolio of smart, connected care solutions, create significant value for customers, and put BD at the forefront of enabling the future of health care."
-Tom Polen, chairman, chief executive officer, and president of BD.
Regulations significantly impact the market, as manufacturers must meet strict safety, efficacy, and quality standards set by agencies like the U.S. FDA and European Medicines Agency (EMA). These requirements ensure reliable patient outcomes, raise product development costs, and extend approval timelines. Simultaneously, compliance with international standards such as ISO 13485 and MDR in Europe promotes innovation in device accuracy, data security, and interoperability, ultimately shaping market competitiveness and adoption.
The level of regional expansion in the industry is moderate. Expanding into new regions has become a key growth strategy for companies in the hemodynamic monitoring devices market, allowing them to tap into underserved areas and increase device adoption. For instance, in June 2023, Mindray launched an advanced hemodynamic monitoring solution integrating Edwards Lifesciences' FloTrac sensor into its BeneVision N series patient monitors, enhancing clinical decision support for critically ill patients in Europe and China.
System Type Insights
The non-invasive segment dominated the market with a 55.6% share in 2024. Several factors, such as increasing demand for patient-friendly monitoring methods, lower risk of infections and complications, advances in non-invasive technologies, and greater adoption in ICUs and surgical settings, support the segment's growth. In addition, features such as patient comfort, ease of use, reduced risk of complications compared to invasive systems, and increasing adoption of home care therapies offered by non-invasive systems are further contributing to the segment's growth.
The minimally invasive segment of the hemodynamic monitoring devices industry is expected to grow significantly during the forecast period. This growth can be attributed to the ability of these systems to provide accurate hemodynamic data, reduce patient risk, and increase adoption in critical care and perioperative settings. Rising preference for safer alternatives to invasive monitoring and technological advancements in sensor-based and catheter-less systems are contributing to the growth.
Product Insights
The monitors segment led the hemodynamic monitoring devices market, with the largest revenue share of 60.44% in 2024. Monitors are the most vital part of the monitoring system as they display all the parameters assessed by healthcare professionals. In addition, the increased investments by leading manufacturers to develop patient-centered care and introduce technologically advanced products are expected to boost demand for hemodynamic monitors. For instance, in September 2023, iRhythm Technologies, Inc. launched its latest product, the next-generation Zio monitor, in the U.S. This new monitor is designed to be the smallest, lightest, and thinnest cardiac monitor offered by iRhythm.
The disposables segment of the hemodynamic monitoring devices industry is anticipated to witness the fastest CAGR over the forecast period. This segment growth can be attributed to the increasing preference for single-use devices to reduce infection risks, rising adoption in critical care settings, and the convenience and cost-effectiveness of disposable components in hemodynamic monitoring procedures.
End Use Insights
The hospitals dominated the hemodynamic monitoring devices market in 2024. This can be attributed to the increasing prevalence of chronic diseases, such as heart failure, respiratory disorders, and sepsis, which require close monitoring of hemodynamic parameters. For instance, according to the CDC, around 1.7 million patients in the U.S. suffer from sepsis annually. This rising burden of critical and chronic illnesses is prompting hospitals and ICUs to adopt advanced hemodynamic monitoring devices to ensure timely interventions, improve patient outcomes, and reduce mortality rates.
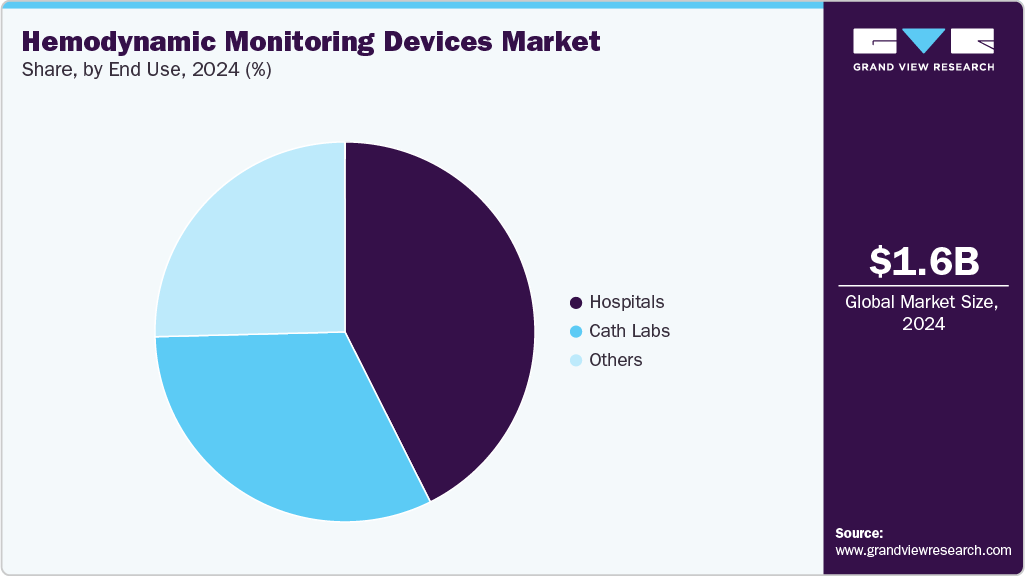
The cath labs segment is expected to witness significant growth opportunities during the forecast period. These labs provide a range of invasive surgical procedures, such as coronary angiography, percutaneous coronary interventions (angioplasty and stent placement), and others that necessitate the demand for hemodynamic monitoring devices. Moreover, strategic initiatives such as expansion are expected to boost the segment growth. For instance, in July 2023, Mount Sinai Queens announced the opening of a new catheterization lab to provide comprehensive care to patients suffering from heart ailments.
Regional Insights
North America dominated the global hemodynamic monitoring devices market with the largest revenue share of 40.99% in 2024. This can be attributed to several key factors, including technological advancements, the increasing prevalence of chronic diseases, and the availability of skilled professionals. The strong presence of industry players, such as Edwards Lifesciences Corporation, GE HealthCare, Baxter, and ICU Medical, has significantly shaped the market landscape. These companies have been at the forefront of manufacturing and distributing, contributing to the technology's adoption & the market's growth in North America.
U.S. Hemodynamic Monitoring Devices Market Trends
The hemodynamic monitoring devices industry in the U.S. is driven by the growing prevalence of chronic conditions such as COPD and heart disease, which require continuous cardiovascular and respiratory monitoring. For instance, according to data from the American Lung Association in 2022, 11.7 million adults (4.6%) were diagnosed with COPD, highlighting the rising need for effective hemodynamic monitoring solutions. Increasing adoption of advanced monitoring technologies in ICUs and critical care settings, along with a focus on improving patient outcomes, is further contributing to the market growth.
Europe Hemodynamic Monitoring Devices Market Trends
The Europe hemodynamic monitoring devices industry’s growth is driven by factors such as the increasing adoption of connected and integrated monitoring systems, rising demand for real-time hemodynamic data in ICUs, the need to improve patient outcomes, the focus on minimally invasive yet comprehensive monitoring solutions in critical care, and strategic initiatives by key industry players. For instance, in September 2021, Masimo received the EU CE Mark for its LiDCO board-in-cable (BIC) module, which integrates with the Masimo Root Patient Monitoring platform to provide clinicians with advanced hemodynamic monitoring capabilities.
The hemodynamic monitoring devices market in the UK is witnessing steady growth, with a notable increase in critical care records from 2020-21 to 2021-22, reaching 246,286 records, marking a 4.6% rise. This growth can be attributed to the escalating prevalence of chronic diseases and the growing elderly demographic. According to a December 2021 update from the Health and Family Records (HFR), the UK is undergoing a demographic shift, with the population above 85 years projected to double to 2.6 million within the next 25 years. This aging trend in the population highlights the increasing prevalence of conditions such as cardiovascular and neurological diseases commonly associated with aging. Thus, the expanding geriatric population is anticipated to fuel the demand for hemodynamic monitoring devices.
The hemodynamic monitoring devices market in Germany held a significant share of the Europe market in 2024. This can be attributed to the rising prevalence of chronic conditions such as cardiovascular ailments, cancer, diabetes, and neurodegenerative disorders among the population is anticipated to drive up the necessity for patient monitoring systems, consequently fueling market expansion
Asia Pacific Hemodynamic Monitoring Devices Market Trends
The hemodynamic monitoring devices industry in the Asia Pacific is witnessing strong growth, driven by the rising prevalence of cardiovascular and chronic respiratory diseases, increasing ICU admissions, and expanding healthcare infrastructure. Adoption of advanced monitoring technologies, growing awareness of early detection and patient safety, and the demand for minimally invasive, real-time hemodynamic monitoring solutions are key factors propelling market expansion. In April 2022, a research article titled Cardiovascular Disease Mortality and Potential Risk Factors in China: A Multi-Dimensional Assessment by a Grey Relational Approach reported that approximately 290 million people in China suffer from cardiac disorders, including stroke, coronary heart disease, heart failure, and hypertension, underscoring the region’s growing need for effective hemodynamic monitoring.
The Japan hemodynamic monitoring devices market is expected to witness significant growth over the forecast period. The expanding geriatric population in the country primarily drives the rise in demand. In September 2022, as per the World Bank, Japan’s population declined by 820,000, while the number of seniors aged 65 years and older increased by 60,000, reaching a record total of 36.27 million individuals. This surge in the senior population represents the highest growth rate recorded thus far. This demographic group is at a higher risk of chronic diseases, which is anticipated to fuel the expansion of the market in Japan.
The hemodynamic monitoring devices market in China has witnessed significant growth recently, primarily driven by favorable conditions for innovation & progress. The country’s aging population & the increasing prevalence of chronic diseases have created a pressing demand for cutting-edge medical technologies. As per a UN report, the elderly population is expected to double from 10% in 2017 to 20% by 2037. The rising geriatric population, escalating per capita healthcare expenditure, and the growing incidence of diseases like cancer and cardiovascular disease are among the major factors anticipated to fuel market growth in China
Latin America Hemodynamic Monitoring Devices Market Trends
The hemodynamic monitoring devices industry in Latin America is experiencing significant growth and evolution driven by various factors such as increasing awareness about cardiovascular disease and the rising prevalence of chronic conditions, such as hypertension & diabetes, across the region. Moreover, the aging population in Latin America is contributing to the growing demand for hemodynamic monitoring devices, as elderly individuals are more susceptible to cardiovascular issues.
Middle East & Africa Hemodynamic Monitoring Devices Market Trends
The hemodynamic monitoring devices industry in the Middle East & Africa is expected to grow rapidly over the forecast period. This growth of the regional market can be attributed to the high prevalence of cardiac diseases. With more people suffering from cardiovascular conditions, the demand for effective management options like hemodynamic monitoring devices is growing rapidly.
Key Hemodynamic Monitoring Devices Company Insights
The market is fragmented, with many small and large players operating in this space. This leads to intense competition between smaller players to sustain their position. Strategies such as new product launches and partnerships are playing a key role in propelling the market growth.
Key Hemodynamic Monitoring Devices Companies:
The following are the leading companies in the hemodynamic monitoring devices market. These companies collectively hold the largest market share and dictate industry trends.
- BD
- GE HealthCare
- Baxter
- ICU Medical, Inc.
- Koninklijke Philips N.V.
- Masimo Corporation
- Deltex Medical Limited
- Getinge
- Nihon Kohden Corporation
- OSYPKA MEDICAL
- Schwarzer Cardiotek GmbH
- Caretaker Medical
- Medtronic
- Retia Medical Systems, Inc.
- Sramek BioDynamics, Inc.
Recent Developments
-
In July 2025, Terumo Health Outcomes announced a U.S. distribution agreement with Caretaker Medical to offer the VitalStream wearable, non-invasive hemodynamic monitoring platform, providing real-time, continuous cardiovascular data to improve early detection and treatment in high-acuity settings. This partnership supports clinicians with safer, wire-free monitoring and aims to enhance clinical workflows and patient outcomes.
-
In April 2025, Medtronic expanded its Acute Care & Monitoring portfolio by entering a U.S. distribution agreement with Retia Medical for the Argos cardiac output monitor, which uses an advanced Multi-Beat Analysis algorithm to provide accurate, real-time hemodynamic data for high-risk surgical and critically ill patients. This monitor enhances perioperative and critical care by offering comprehensive insights without the need for calibration or costly disposables.
-
In February 2021, Masimo completed the acquisition of LiDCO Group for USD 40.1 million, expanding its portfolio with advanced hemodynamic monitoring technology. This strategic move would enhance Masimo's capabilities in critical care and high-risk surgical patient monitoring, with LiDCO's primary markets being in the U.S. & the UK, and key markets in Europe, China, & Japan.
-
In September 2023, Caretaker Medical partnered with Red One Medical to provide ICU-level, continuous hemodynamic monitoring using the VitalStream wearable device to U.S. military units and Veterans Affairs, improving early detection of cardiovascular changes and enabling safer care for soldiers and veterans in field and hospital settings.
Hemodynamic Monitoring Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.67 billion
Revenue forecast in 2033
USD 2.78 billion
Growth Rate
CAGR of 6.5% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company market share, competitive landscape, growth factors, and trends
Segments covered
System type, product, end use, region
Regional scope
North America; Europe; Asia Pacific; Central & South America; MEA
Country Scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Sweden; Denmark; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
BD; GE HealthCare; Baxter; ICU Medical, Inc.; Koninklijke Philips N.V.; Masimo Corporation; Deltex Medical Limited; Getinge; Nihon Kohden Corporation; OSYPKA Medical; Schwarzer Cardiotek GmbH; Caretaker Medical; Medtronic; Sramek BioDynamics, Inc.; Retia Medical Systems, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Hemodynamic Monitoring Devices Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global hemodynamic monitoring devices market report based on system type, product, end use, and region:
-
System Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Invasive
-
Minimally Invasive
-
Non-Invasive
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Disposables
-
Monitors
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Catheterization Labs
-
Other
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global hemodynamic monitoring devices market size was estimated at USD 1.58 billion in 2024 and is expected to reach USD 1.67 billion billion in 2025.
b. The global hemodynamic monitoring devices market is expected to witness moderate growth from 2025 to 2033 to reach USD 2.78 billion by 2033
b. North America dominated the hemodynamic monitoring devices market with a share of about 40.99% in 2024. This is attributable to increasing demand for minimally invasive technologies and new product launches
b. Some key players operating in the hemodynamic monitoring devices market include Edward Lifesciences, Masimo Deltex Medical, Philips Medical, GE Healthcare, ICU Medical, Sramek BioDynamics, Inc., OsypkaCardiotek GmbH
b. Key factors that are driving the market growth include increasing demand for non-invasive monitoring techniques coupled with the rising prevalence of chronic illness
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










