- Home
- »
- Healthcare IT
- »
-
Indian Clinical Trials Market Size And Share Report, 2030GVR Report cover
![Indian Clinical Trials Market Size, Share & Trends Report]()
Indian Clinical Trials Market Size, Share & Trends Analysis Report, By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Interventional Trials, Observational Trials), By Indication, By Region, And Segment Forecasts, 2023 - 2030
- Report ID: GVR-2-68038-676-9
- Number of Pages: 113
- Format: Electronic (PDF)
- Historical Range: 2018 - 2021
- Industry: Healthcare
Report Overview
The Indian clinical trials market size was valued at USD 2.1 billion in 2022 and is anticipated to grow at a CAGR of 8.3% over the forecast period. The key factors estimated to drive the market are the globalization of clinical trials, adoption of new technology in clinical research, growing disease variation and prevalence, and increasing research and development promoting outsourcing. The government in this country is taking active initiatives to improve R&D activities, likely to boost the market's growth. In October 2021, the Department of Pharmaceuticals (India) proposed a new policy to reduce the time required to approve innovative products by at least 50% within the next two years, to improve the R&D activities in the country. Such initiatives are likely to propel the growth of the market further.
The digitalization of clinical trials is expected to impact market growth positively. Digitalization has enabled streamlining several trial processes, such as data capture, regulatory compliance, logistics, and supplies management. Furthermore, with the introduction of digital therapeutics, real-time data acquisition related to safety and toxicity is becoming increasingly easy, thereby promoting timely rectification in trial design and facilitating market growth. The cost of clinical trials in India is nearly half that incurred in the U.S. and Europe. Thus, the cost efficiency and a large treatment-naïve patient pool offered by the county are anticipated to drive the market.
The outbreak of the COVID-19 pandemic has created a catastrophic situation across the world, and India faced no different scenario. The initial period of the pandemic has created a halt in research and development activities across the country due to factors such as disruption in the supply chain and the shutdown of manufacturing plants for a certain timeframe, thereby creating obstacles in the path of clinical research being conducted. Moreover, the implemented lockdown has reduced the number of nationwide patient trials.
However, the COVID-19 pandemic had created an urgent need for treatments and vaccines, owing to which a significant number of clinical trials were performed in the country post-2021, thus reflecting a rebound in the total number of trials conducted across India. For instance, in November 2021, U.S.-based Akston Biosciences announced that it would start the clinical trial for its second-generation COVID-19 vaccine 'AKS-452' in India. Similarly, in April 2021, the Ministry of Ayush and Council of Scientific & Industrial Research in India announced that it completed a multi-center clinical trial of AYUSH 64 drug used to treat mild-to-moderate COVID-19 infections in the country. The rise in the number of clinical trials for COVID-19 post-2020 is likely to impact the market growth from 2021 positively.
Phase Insights
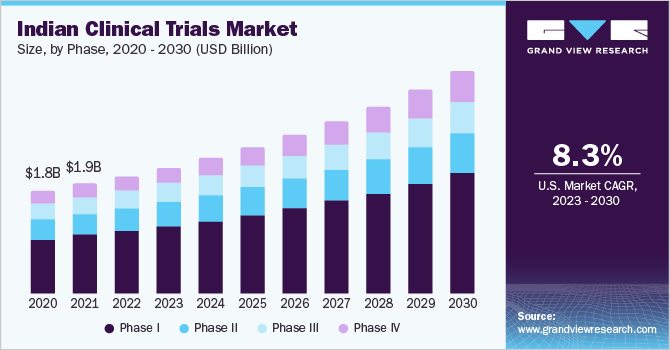
Based on phase, the market is segmented into phases I, II, III, and IV. The phase III segment dominated the market with the largest revenue share of 53.3% in 2022, owing to the cost-intensive nature of this segment. Phase III trials require a large patient population, which is one of the prime reasons for the high cost of this trial. Moreover, the studies in the phase last for a longer time, compared to phase I and II, which further improves the trial's cost and the market's segment share.
The phase I segment is expected to grow at the fastest CAGR of 10.0% over the forecast period from 2023 to 2030 owing to significant R&D investments by global and local pharmaceutical and biotechnological firms. The country's demand for new treatments and biologics further contributes to the segment's growth.
Study Design Insights
The interventional trials segment dominated the market with the largest revenue share of 40.8% in 2022 due to the large number of trials adopting this study design. This type of study design includes large randomized clinical trials and is considered one of the best study designs for detecting small to moderate effects of drugs. These studies also provide the best means of minimizing the confounding effect, thus improving their adoption compared to other clinical study designs.
The observational trials segment is expected to grow at the fastest CAGR of 8.5% during the forecast period due to its ease of conduct and level of control, making it a good option for countries like India, where the infrastructure for clinical trials may not be as well-developed. The increasing prevalence of chronic diseases, regulatory support from the government, and availability of the largest datasets for observational studies are driving the segment's growth.
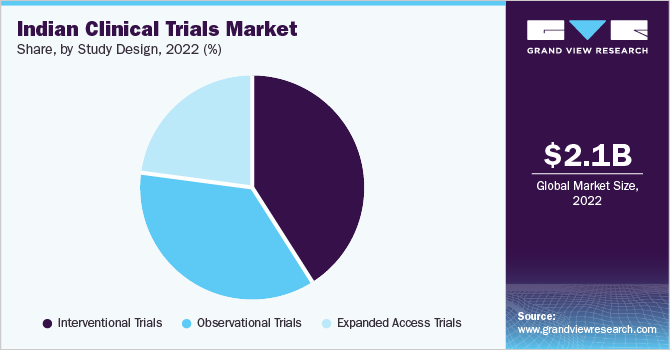
The expanded access trials segment is anticipated to grow at a lucrative CAGR during the forecast period. Factors such as the growing incidence of serious diseases, demand for new innovative treatment options, and the country's high burden of chronic diseases are anticipated to fuel the segment’s growth. Moreover, owing to the COVID-19 pandemic, an expanded access study with convalescent plasma was used in India to treat patients with COVID-19. Such actions are further contributing to the segment growth.
Indication Insights
The oncology segment dominated the Indian clinical trials market with the largest revenue share of 24.9% in 2022 and is expected to grow at the fastest CAGR of 10.5% over the forecast period, owing to the increasing prevalence of cancer in the country and the growing demand for advanced medical devices and cancer treatments. Cancer is considered one of the major causes of death globally and is expected to affect approximately 2 million people in India by 2040, according to the Cancer Tomorrow estimates. Most treatments for cancer are ineffective in the last stages of the disease, which is a key reason for the growing demand for new treatment options for cancer in the country.
The various measures and launches are driving the oncology market growth. For instance, in November 2023, the International Association of Oncology (AIO) in partnership with BioLEAGUES announced the 5th Indo Oncology Summit, taking place in Bhubaneshwar, India, on the subject “Cancer Care: Era of Precision.” In January 2023, the Indian government announced the launch of its first made-in-India CERVAVAC vaccine, a “Quadrivalent Human Papillomavirus” (qHPV) vaccine that helps prevent cervical cancer. In March 2023, Dr. AV Cancer Institute of Personalized Cancer Therapy and Research, Gurugram, launched India’s first Precision Oncology treatment center to make precision oncology treatment affordable and easily accessible nationwide.
CNS conditions held a significant revenue share in 2022. Many people in India are affected by neurological disorders like stroke, epilepsy, tremors, and Parkinson’s disease; the trend is similar globally. This is expected to impact R&D investments for clinical trials in the country positively.
Key Companies & Market Share Insights
The key market players adopt various strategies such as mergers, acquisitions, and collaborations to maintain their market share. For instance, in February 2023, Syneos Health collaborated with Haystack Health to improve and enhance the enrollment and identification of patients for clinical trials. Also, in January 2023, Syneos Health announced its collaboration with Fosun Pharma U.S. intending to deliver full-service commercial support through its novel anti-PD-1 antibody. In December 2021, Aragen Life Sciences, an India-based CRO, acquired Intox Pvt. Ltd. in Maharashtra, India, to expand Aragen’s end-to-end integrated discovery and development platform for biotechnology, pharmaceuticals, and other industries. In December 2021, Thermo Fisher Scientific Inc. acquired PPD Inc., which globally delivers clinical research services to the biotech and biopharma industry.
In November 2021, EQT and Goldman Sachs acquired Parexel to provide innovative and advanced solutions to their customers and patients. Similarly, in February 2021, ICON plc, a global provider of outsourced drug and device development companies, acquired PRA Health Sciences, Inc. This acquisition is aimed to provide hybrid and decentralized solutions to satisfy increasing customer needs by offering new medicines and devices to market.
In February 2020, India-based clinical-stage company, Aurigene collaborated with a biotechnology companyCuris, Inc. As per the terms of the collaboration, Aurigene provided funding for conducting Phase 2b/3 randomized study on evaluating the effect of CA-170 in patients with non-squamous non-small cell lung cancer (nsNSCLC). The adoption of such strategies by the market players is likely to impact market growth positively. Some prominent players in the Indian clinical trials market include:
-
IQVIA HOLDINGS INC.
-
PAREXEL International Corporation
-
Pharmaceutical Product Development (PPD) LLC
-
Charles River Laboratory
-
ICON PLC
-
PRA Health Inc.
-
Chiltern International LTD.
-
Syneos Health Inc.
-
SGS SA
-
Syngene International Limited
-
Aurigene Discovery Technologies Limited
-
Aragen Life Sciences
-
Abiogenesis Clinpharm Pvt Ltd
-
Accelsiors
-
Accutest Global.
-
Clario
-
Cliantha Research
-
Cliniminds
-
CliniRx
-
JSS Medical Research
Indian Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 2.21 billion
Revenue forecast in 2030
USD 3.87 billion
Growth Rate
CAGR of 8.3% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
September 2023
Quantitative units
Revenue in USD million/billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication
Key companies profiled
IQVIA HOLDINGS INC.; PAREXEL International Corporation; Pharmaceutical Product Development (PPD), LLC; Charles River Laboratory; ICON PLC; PRA Health Inc.; Chiltern International LTD.; Syneos Health Inc.; SGS SA; Syngene International Limited; Aurigene Discovery Technologies Limited; Aragen Life Sciences; Abiogenesis Clinpharm Pvt Ltd; Accelsiors; Accutest Global; Clario; Cliantha Research; Cliniminds; CliniRx; JSS Medical Research
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Indian Clinical Trials Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2018 to 2030. For the purpose of this study, Grand View Research has segmented the Indian clinical trials market report on the basis of phase, study design, and indication:
-
Phase Outlook (Revenue in USD Million, 2018 - 2030)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue in USD Million, 2018 - 2030)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue in USD Million, 2018 - 2030)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Other
-
-
CNS Condition
-
Epilepsy
-
Parkinson's Disease (PD)
-
Huntington's Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular
-
Others
-
Frequently Asked Questions About This Report
b. The Indian clinical trials market size was estimated at USD 2.1 billion in 2022 and is expected to reach USD 2.21 billion in 2023.
b. The Indian clinical trials market is expected to grow at a compound annual growth rate of 8.3% from 2023 to 2030 to reach USD 3.87 billion by 2030.
b. Phase III dominated the Indian clinical trials market with a share of 53.3% in 2022. This is attributable to the cost-intensive nature of this segment.
b. Some key players operating in the Indian clinical trials market include IQVIA, Inc.; ICON PLC; PRA Health Sciences; GVK Biosciences; PAREXEL International Corporation; Syneos Health; Charles River Laboratories; and Syngene International Ltd.
b. Key factors that are driving the Indian clinical trials market growth include globalization of clinical trials, rising prevalence of chronic disease, and digitalization of clinical trials.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





