- Home
- »
- Pharmaceuticals
- »
-
Vaccine Market Size, Share & Trends, Industry Report, 2033GVR Report cover
![Vaccine Market Size, Share & Trends Report]()
Vaccine Market (2026 - 2033) Size, Share & Trends Analysis Report By Indication (Viral Vaccines, Bacterial Vaccines), By Type (Inactivated, Live Attenuated), By Route Of Administration, By Age Group, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: 978-1-68038-990-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size, 2025$72.8BMarket Estimate, 2026$78.1BMarket Forecast, 2033$115.8BCAGR, 2026 - 20335.8%Vaccine Market Summary
The global vaccine market size was estimated at USD 72.75 billion in 2025 and is projected to reach USD 115.77 billion by 2033, growing at a CAGR of 5.78% from 2026 to 2033. The market is driven by the increasing investment in R&D and the rising focus of sponsors & investigators on reducing research costs.

Key Market Trends & Insights
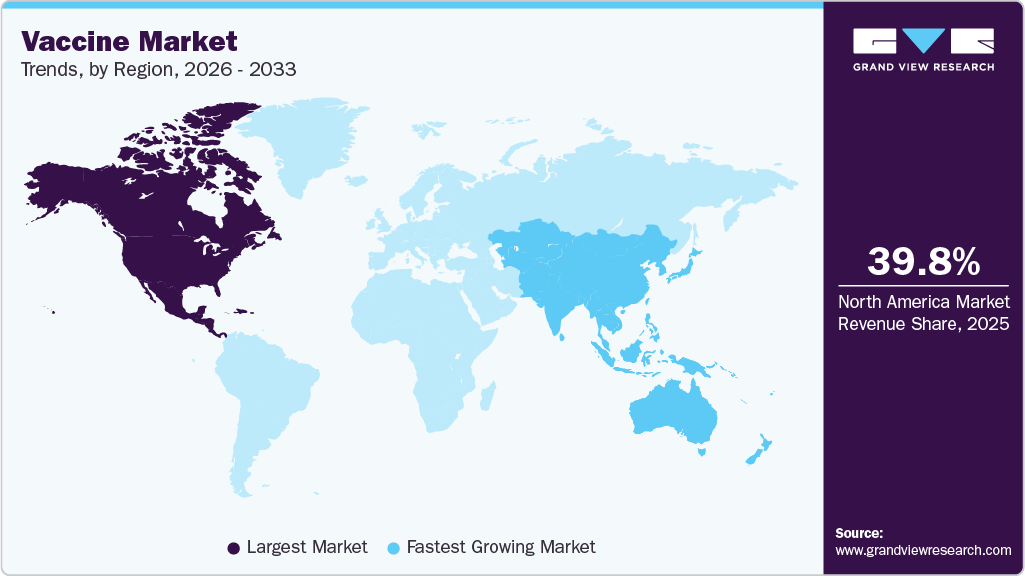
- North America dominated the global market with a share of 39.80% in 2025.
- By indication, the viral diseases segment held the largest market share of 61.17% in 2025.
- By type, the subunit vaccine segment held the largest market share of 30.72% in 2025.
- By route of administration, the parenteral segment dominated the market with a share of 84.80% in 2025.
- By age group, the adult segment dominated the market with a share of 56.65% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 72.75 Billion
- 2033 Projected Market Size: USD 115.77 Billion
- CAGR (2026-2033): 5.78%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Moreover, the increasing adoption of outsourcing laboratory services by pharmaceutical & biotechnology companies and other end users to reduce the overall cost of research activities further supports the market growth over the forecast period. The global vaccine industry is positioned as a strategic component of preventive healthcare systems and is primarily driven by public immunization programs, outbreak preparedness, and routine disease prevention initiatives across regions. Vaccines are integrated into national healthcare budgets and long-term public health planning due to their role in reducing disease burden and downstream treatment costs. According to the World Health Organization, immunization prevents an estimated 2-3 million deaths every year worldwide by protecting against vaccine-preventable diseases such as measles, diphtheria, tetanus, pertussis, and influenza. In April 2024, the WHO published a comprehensive analysis in The Lancet reporting that global immunization programs have prevented approximately 154 million deaths over the past 50 years, reinforcing vaccination as a central public health investment. In April 2025, the World Health Organization marked World Immunization Week with a focus on closing immunization gaps, strengthening immunization delivery systems, and expanding vaccine coverage across the life course, reflecting continued global policy prioritization of immunization as a core public health intervention.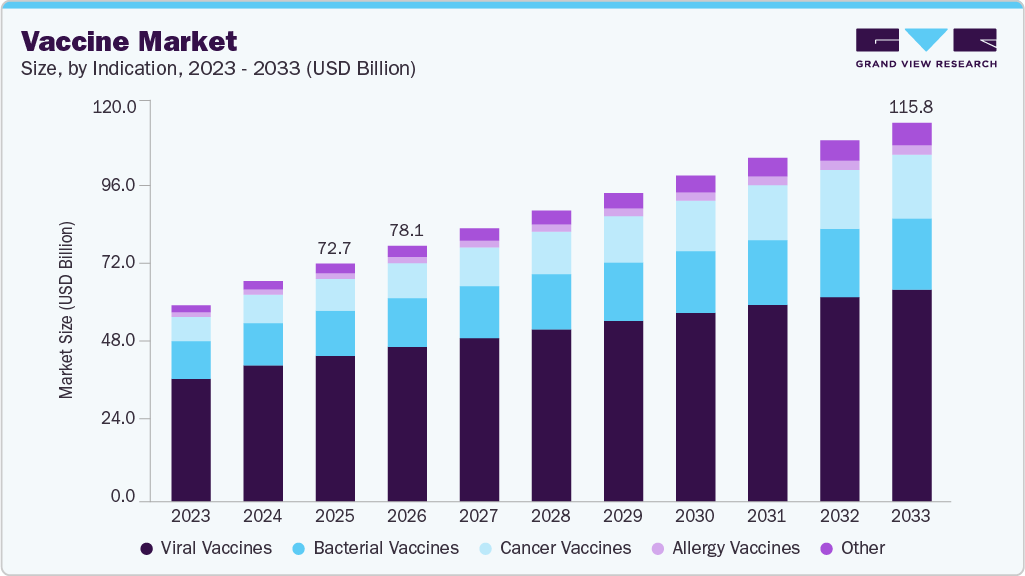
Market growth is supported by the continued prevalence of infectious diseases, expansion of adult and geriatric immunization programs, and recovery of routine vaccination coverage following pandemic-related disruptions. According to the World Health Organization Global Tuberculosis Report 2024, an estimated 10.8 million people developed tuberculosis globally in 2023, with the highest burden concentrated in regions such as Southeast Asia and Africa, sustaining demand for preventive and control strategies in high-burden markets. In April 2024, the World Health Organization Global Hepatitis Report estimated that approximately 254 million people were living with chronic hepatitis B and 50 million with chronic hepatitis C worldwide, with more than 1 million new viral hepatitis infections occurring each year, underscoring the continued public health burden of hepatitis globally. In May 2025, the U.S. Food and Drug Administration approved Moderna’s next-generation COVID-19 vaccine, mNEXSPIKE, for older adults aged 65 years and above and for individuals aged 12-64 years with underlying medical conditions, indicating continued regulatory support for vaccine adoption among high-risk populations.
The market is increasingly shaped by shifts in technology platforms, product portfolios, and manufacturing strategies, reflecting a transition toward broader disease coverage and long-term supply resilience. Platform-based development approaches, particularly mRNA technologies, have enabled faster design cycles, greater flexibility in antigen selection, and the development of combination vaccines, supporting portfolio expansion beyond single-disease products. These advancements are contributing to increased focus on respiratory indications and multi-pathogen prevention strategies, particularly within adult and elderly populations. In September 2022, Pfizer and BioNTech initiated Phase III clinical trials for an mRNA-based influenza vaccine, marking the extension of mRNA platforms beyond COVID-19 applications. In May 2025, the World Health Organization reported that its mRNA Technology Transfer Programme had expanded to 15 global partners, supporting the decentralization of vaccine manufacturing and the development of regional production capacity. In parallel, expanding adult immunization frameworks and broader regulatory recommendations are increasing the addressable market for vaccines across age groups, indicating that market evolution is being driven by public-sector priorities, platform diversification, and demographic shifts rather than short-term demand fluctuations.
Market Dynamics
The rising prevalence of infectious diseases and increasing focus on pandemic preparedness are the major drivers of the global vaccine market. Frequent outbreaks of diseases such as COVID-19, influenza, dengue, RSV, measles, and other viral infections have significantly increased the demand for vaccines across both developed and developing countries. Governments, healthcare organizations, and pharmaceutical companies are investing heavily in immunization programs, vaccine research, and manufacturing infrastructure to strengthen public health systems and reduce the risk of future pandemics. In addition, advancements in vaccine technologies, including mRNA, recombinant, and viral vector platforms, are enabling faster vaccine development and broader disease protection, which further supports market expansion. The growing awareness regarding preventive healthcare and the rising adoption of adult vaccination programs are also contributing to market growth. For instance, in April 2026, the World Health Organization (WHO) and Gavi announced that over 100 million vaccine doses had been delivered to 18.3 million children across 36 countries under “The Big Catch-Up” initiative since 2023, emphasizing the global priority toward improving immunization coverage and preventing infectious disease outbreaks.
The high costs associated with vaccine development and distribution are one of the major restraints affecting the growth of the global vaccine market. Developing vaccines requires substantial investment in research, clinical trials, regulatory approvals, and large-scale manufacturing, which significantly increases overall production costs. In addition, maintaining cold-chain storage and transportation systems for temperature-sensitive vaccines further raises operational expenses, especially in developing regions with limited healthcare infrastructure. Strict regulatory standards and quality control requirements also add complexity and increase the financial burden on manufacturers. Smaller biotechnology companies often face challenges in securing sufficient funding to commercialize vaccine candidates, limiting innovation and market entry. Furthermore, declining demand for certain vaccines after pandemic periods can create financial pressure on manufacturers and reduce profitability. For instance, in July 2025, Moderna announced plans to reduce approximately 10% of its global workforce as part of a broader cost-cutting initiative aimed at lowering operating expenses by nearly USD 1.5 billion by 2027 amid declining COVID-19 vaccine sales and rising financial pressures.
Advancements in mRNA and next-generation vaccine technologies presented a significant opportunity for the global vaccine market. The successful commercialization of mRNA-based COVID-19 vaccines accelerated global investment in innovative vaccine platforms capable of faster development, higher efficacy, and broader disease protection. Pharmaceutical and biotechnology companies increasingly focused on developing mRNA vaccines for influenza, RSV, HIV, cancer immunotherapy, and other infectious diseases. These technologies also enabled rapid responses to emerging health threats and future pandemics, creating strong long-term growth opportunities for the market. In addition, increasing government funding, strategic collaborations, and advancements in genomic research supported the development of personalized and therapeutic vaccines. The growing demand for combination vaccines and improved immunization coverage across developing countries further strengthened market potential. For instance, in May 2026, Clinical Trials Arena reported that Moderna’s Phase III trial for its mRNA-based seasonal influenza vaccine, mRNA-1010, demonstrated superior efficacy compared to the current standard-of-care influenza vaccine. The study enrolled more than 40,000 adults aged 50 years and older, with influenza-like illness reported in 411 of 20,179 participants receiving mRNA-1010, compared to 557 of 20,124 participants receiving GSK’s Fluarix or Alpharix vaccine. The vaccine showed 26.6% greater effectiveness than the comparator vaccine, highlighting the growing commercial and clinical potential of next-generation mRNA vaccine technologies in the global vaccine market.
Market Concentration & Characteristics
The market shows high levels of innovation driven by ongoing need to respond to emerging pathogens, improve efficacy and enhance safety. Breakthroughs in messenger RNA and viral vector technologies have transformed development timelines. Research efforts focus on broad spectrum and single-dose vaccines to improve coverage. Public and private funding fuels early stage discovery and clinical validation. Collaboration between biotech startups and large pharmaceutical firms accelerates technology transfer. Continuous innovation shapes competitive positioning and future revenue streams.
Entering the market requires substantial financial investment in research, clinical trials and manufacturing capacity. Complex regulatory approvals demand extensive safety and efficacy evidence before products reach market. Intellectual property rights create competitive fences that protect incumbent firms. High technical expertise and specialized facilities are essential for development and production. New entrants must establish distribution networks and cold chain logistics to compete effectively. These barriers concentrate market power among established global players.
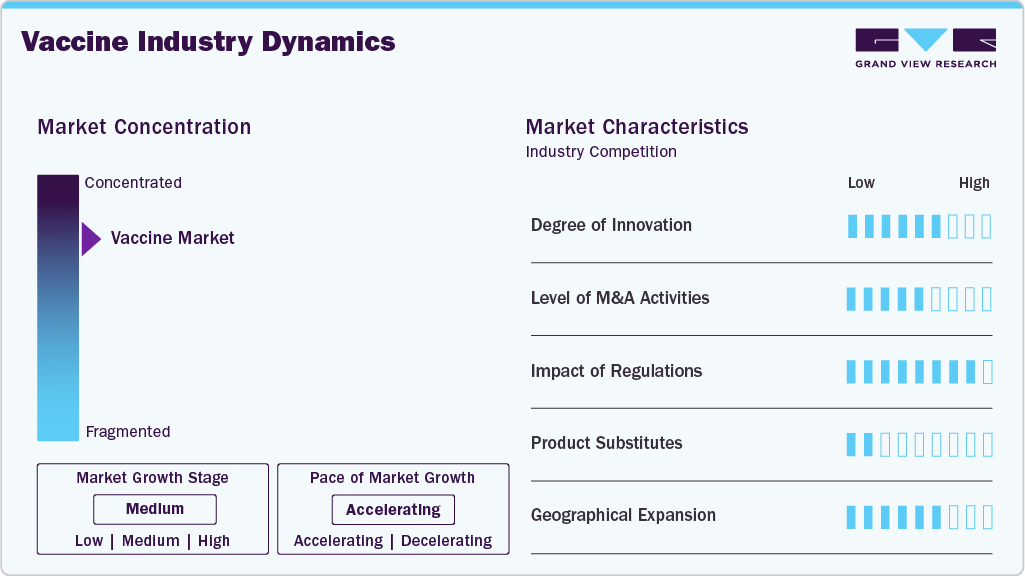
Regulations heavily influence market dynamics by ensuring product safety and public trust. Licensing and approval processes vary by country but typically involve multi-phase clinical trials with stringent oversight. Regulatory standards drive investment in quality management systems throughout the value chain. Post-market surveillance obligations require ongoing monitoring and reporting of adverse events. Compliance increases development time and cost, shaping strategic planning. Harmonization efforts across regions can reduce duplication and accelerate access.
In the vaccine industry, traditional substitutes include preventive therapeutics such as antiviral drugs and monoclonal antibodies. Public health measures like sanitation, masking and social distancing reduce disease transmission without pharmaceutical intervention. Diagnostic testing paired with early treatment can lessen disease impact, reducing demand for some vaccines. Alternative preventive technologies like immune modulators may shift investment priorities. Consumer preferences for non-invasive options influence market dynamics. Substitutes challenge pricing and allocation of research resources.
Geographical expansion in the vaccine industry is driven by global demand for immunization against endemic and pandemic diseases. Manufacturers pursue entry into emerging markets with large populations and growing healthcare access. Strategies include licensing agreements, local partnerships and technology transfer to navigate regional regulatory landscapes. Distribution infrastructure investments support cold chain requirements in remote areas. Market growth is shaped by government procurement programs and international aid initiatives. Global presence diversifies revenue and spreads development risks.
Indication Insights
The viral vaccines segment dominated the market with the largest revenue share of 61.17% in 2025, supported by structurally embedded demand from national immunization programs, regulatory prioritization, and continued platform investments. For instance, in 2024, public health authorities across major markets reinforced routine and adult immunization schedules for viral diseases, including influenza, HPV, and COVID-19 boosters. Regulatory bodies supported ongoing strain updates, label extensions, and lifecycle management of existing viral vaccines, ensuring continuity of procurement and manufacturing demand beyond pandemic-driven volumes.
The cancer vaccines segment is projected to grow at the fastest CAGR of 9.10% over the forecast period, supported by formal integration of therapeutic cancer vaccines into national and international oncology innovation agendas. For instance, in 2025, global health and regulatory institutions increasingly positioned cancer vaccines within broader cancer control and precision medicine frameworks, emphasizing their role in addressing high-burden cancers with unmet clinical need. This shift elevated cancer vaccines from experimental immunotherapies to strategically prioritized oncology interventions, encouraging sustained funding, structured clinical pathways, and manufacturing preparedness. As a result, the segment is benefiting from improved policy visibility and long-term development support, reinforcing its accelerated growth trajectory relative to other vaccine categories.
Type Insights
The Subunit Vaccines segment dominated the market with the largest revenue share of 30.72% in 2025 and is expected to grow at the fastest CAGR of 7.67% over the forecast period, due to continued validation of protein-based platforms by regulators and public health buyers, supporting reliability, safety, and scalable manufacturing. For instance, in 2024-2025, Novavax advanced and sustained the use of its recombinant protein (subunit) vaccine platform for COVID-19, with regulatory authorizations supporting ongoing use and updates. The platform’s favorable tolerability profile, compatibility with existing cold-chain infrastructure, and suitability for booster strategies reinforced institutional confidence in subunit technologies for repeated and large-scale immunization, underpinning the segment’s continued leadership.
Route of Administration Insights
The parenteral segment dominated the market with the largest revenue share of 84.80% in 2025, supported by the injectable-centric vaccine portfolios of long-established manufacturers serving national immunization programs. For instance, in 2025, Sanofi continued to anchor its global vaccines business around parenteral formulations, with injectable vaccines forming the backbone of its pediatric and adult immunization portfolio.Sanofi’s sustained investment in injectable manufacturing capacity, fill-finish infrastructure, and supply reliability reflects the continued preference of public health authorities for parenteral administration due to its consistent immunogenicity, standardized dosing, and well-established regulatory pathways. This reinforces the parenteral route as the default and most trusted mode of vaccine delivery globally.
The nasal segment is projected to grow at the fastest CAGR of 6.44% over the forecast period. This growth is primarily driven by increasing interest in needle-free vaccine delivery, mucosal immunity benefits, and improved patient compliance. For instance, Bharat Biotech advanced and commercialized an intranasal COVID-19 vaccine platform, positioning nasal delivery as a viable alternative to injectable administration for respiratory pathogens. The company’s progress demonstrated the potential of nasal vaccines to induce localized mucosal immunity at the site of infection while simplifying administration and reducing dependence on trained healthcare personnel. Such real-world advancement by manufacturers has strengthened regulatory and institutional interest in nasal delivery platforms, supporting their accelerated growth outlook compared with conventional routes.
Age Group Insights
The adult segment held the largest revenue share of 56.65% in the market in 2025 and is expected to grow at the fastest CAGR of 6.63% over the forecast period. Factors supporting growth are the rising prevalence of vaccine-preventable diseases and age-related conditions in adult populations. For instance, in 2024 - 2025, public health authorities continued to highlight the high and growing burden of influenza, shingles (herpes zoster), pneumococcal disease, and COVID-19 among adults, particularly in older and immunocompromised populations. This disease prevalence has reinforced the expansion of adult immunization recommendations and booster-based vaccination strategies, positioning adult vaccination as a critical component of preventive healthcare. As a result, sustained epidemiological need, rather than episodic outbreaks, has structurally supported both the dominance and faster growth of the adult vaccine segment.
Distribution Channel Insights
The Hospital & Retail Pharmacies segment dominated the market with the largest revenue share of 56.36% in 2025 and is expected to grow at the fastest CAGR of 6.76% over the forecast period, driven by expanded vaccination access points, integration of immunization services into routine care, and rising adult vaccination uptake. For instance, in 2025, public health authorities across major markets continued to expand authorization for retail pharmacies and hospital outpatient settings to administer a broad range of vaccines, including adult and booster vaccinations.

This policy-driven expansion positioned hospitals and retail pharmacies as primary points of care for vaccination delivery, benefiting from extended operating hours, established cold-chain infrastructure, and direct patient access. As vaccination increasingly shifts toward convenient, community-based settings, hospitals and retail pharmacies have strengthened their role as the dominant and fastest-growing distribution channels in the vaccine industry.
Regional Insights
North America vaccine industry held the largest share of the global market for vaccines in 2025, accounting for 39.80% of global revenue, strong public funding, advanced healthcare infrastructure, and high adult immunization uptake. The region continues to prioritize respiratory vaccines, booster programs, and preparedness for emerging infectious diseases. Policy focus has shifted toward long term vaccination strategies, including adult and elderly populations, supported by pharmacy-based administration and hospital outpatient settings. Manufacturers continue to invest in advanced vaccine platforms and domestic manufacturing capacity to improve supply security. Regulatory clarity, stable reimbursement frameworks, and strong participation from large multinational vaccine producers underpin sustained market leadership.
U.S. Vaccine Market Trends
The United States vaccine industry continues to serve as the global anchor for vaccine innovation, procurement, and administration. Adult immunization has gained increasing emphasis, supported by expanded recommendations for influenza, COVID boosters, shingles, and pneumococcal vaccines. Retail pharmacies and hospital systems have become primary vaccination access points, improving convenience and coverage. Federal agencies continue to prioritize domestic vaccine manufacturing and supply chain resilience. Regulatory agencies maintain strong support for platform-based vaccine development and lifecycle management. Ongoing public health initiatives and employer supported vaccination programs continue to reinforce long term demand stability.
Europe Vaccine Market Trends
Europe vaccine industry represents a policy driven market with strong emphasis on public health coordination and equitable access. National immunization programs remain the primary demand driver, supported by centralized procurement and strict regulatory oversight. The region has increased focus on adult and elderly vaccination, particularly for respiratory and age-related infectious diseases. Governments continue to emphasize supply security, regional manufacturing capacity, and preparedness planning. Regulatory harmonization across European markets supports consistent product adoption. Hospitals and public healthcare systems remain the dominant vaccination delivery channels, ensuring predictable long-term demand.
The United Kingdom vaccine industry is characterized by centralized procurement and strong public health governance. The National Health Service continues to play a central role in vaccine distribution, with growing use of pharmacies and community clinics for adult vaccination. The country has emphasized seasonal and booster vaccination strategies, particularly for respiratory infections. Investments in domestic vaccine research and manufacturing continue to support supply resilience. Policy focus remains on prevention driven healthcare and improved coverage across adult and vulnerable populations, supporting steady market expansion.
Germany vaccine industry remains one of the most influential markets in Europe, driven by high healthcare spending and strong institutional trust in immunization programs. Adult vaccination uptake continues to improve, supported by physician led administration and pharmacy participation. The country emphasizes evidence-based recommendations and long-term disease prevention strategies. Domestic and multinational manufacturers benefit from a stable regulatory environment and well-defined reimbursement mechanisms. Germany also plays a strategic role in European vaccine manufacturing and research, supporting supply stability and innovation focused development across therapeutic areas.
France vaccine industry is a robust market supported by national immunization schedules and strong hospital-based delivery systems. Adult and elderly vaccination coverage has become an increasing priority, particularly for influenza and respiratory diseases. Government initiatives continue to address vaccine hesitancy through education and public engagement. France also benefits from a strong domestic pharmaceutical and vaccine manufacturing base, supporting innovation and export capacity. Regulatory stability and public reimbursement continue to provide predictable demand and long-term market visibility.
Asia-Pacific Vaccine Market Trends
Asia Pacific vaccine industry is expected to register a significant CAGR of 7.5% over the forecast period. The market is driven by population growth, expanding healthcare access, and rising public health investment. Governments across the region continue to strengthen immunization infrastructure and domestic manufacturing capabilities. Adult vaccination programs are expanding alongside traditional pediatric coverage. Regional players are increasingly active in vaccine development and supply for both domestic and export markets. Policies focuses on preparedness and self-reliance have increased following recent public health challenges, supporting long term growth momentum.
Japan vaccine industry is a highly regulated and quality focused market with strong government oversight. The country continues to emphasize adult and elderly immunization as part of its aging population strategy. Vaccine adoption is supported by physician led administration and hospital-based care. Regulatory authorities maintain rigorous approval standards while supporting innovation through structured pathways. Japan also continues to invest in domestic vaccine research and manufacturing, reinforcing supply security and technological advancement. Demand remains stable and policy driven.
China vaccine industry has emerged as a major vaccine producer and consumer, supported by large scale public immunization programs and growing domestic innovation. Government initiatives continue to strengthen national vaccine supply chains and manufacturing quality standards. Adult vaccination coverage is expanding gradually alongside established childhood programs. China is also increasing its role in global vaccine supply through exports and international cooperation. Regulatory reforms have improved transparency and oversight, supporting broader adoption and long-term market confidence.
Latin America Vaccine Market Trends
Latin America vaccine industry remains a public sector driven market, with national immunization programs serving as the primary demand channel. Governments continue to prioritize disease prevention and equitable access, supported by international collaboration. Adult vaccination programs are gradually expanding, particularly for influenza and respiratory diseases. Local manufacturing capabilities are improving, although the region continues to rely on imports for advanced vaccines. Budget sensitivity and procurement cycles influence market dynamics, but long-term demand remains stable due to public health commitments.
Brazil vaccine industry represents the largest market in Latin America, driven by strong government led immunization programs and extensive public healthcare coverage. The country has a long-standing focus on mass vaccination campaigns and disease prevention. Domestic public sector manufacturers play a key role in supply, supported by technology transfer partnerships. Adult and booster vaccination has gained prominence in recent years. Brazil continues to strengthen regulatory oversight and manufacturing capabilities, supporting both domestic use and regional distribution.
Middle East & Africa Vaccine Market Trends
The Middle East and Africa vaccine industry shows uneven but improving market development, driven by government initiatives and international support. Countries are expanding immunization coverage and strengthening cold chain infrastructure. Adult vaccination remains underpenetrated but is gaining policy attention, particularly in higher income markets. Regional manufacturing and full-finish investments are increasing to reduce import dependence. Public sector procurement and donor funded programs remain central to demand, with gradual shifts toward broader preventive healthcare strategies.
Saudi Arabia vaccine industry represents one of the most advanced markets in the Middle East, supported by strong government funding and centralized healthcare planning. The country has emphasized adult vaccination and preparedness strategies aligned with public health modernization goals. Hospitals and government healthcare facilities serve as the primary vaccination channels. Investments in local pharmaceutical and vaccine manufacturing continue to improve supply security. Regulatory authorities support adoption of advanced vaccine technologies, reinforcing long term market stability and growth.
Key Vaccine Company Insights
Serum Institute of India Pvt. Ltd. holds a prominent position in the market through large scale manufacturing capabilities, cost efficient production, and a broad portfolio spanning pediatric, influenza, and COVID-19 vaccines. Seqirus and Sanofi maintain notable market presence supported by specialized expertise in influenza vaccines, established global distribution networks, and continued investments in vaccine development. Merck & Co., Inc. and Pfizer Inc. maintain substantial market shares driven by extensive vaccine pipelines, advanced R&D capabilities, and long-standing experience in complex and combination vaccines. Moderna Inc. and BioNTech SE expanded their market footprint by leveraging mRNA platform technologies and rapid clinical development capabilities. Sinovac and AstraZeneca contribute to market expansion through inactivated and viral vector vaccine platforms, broad international reach, and collaborations that improve vaccine access across diverse regions.
Key Vaccine Companies:
The following key companies have been profiled for this study on the vaccine market.
- Serum Institute of India Pvt. Ltd.
- Seqirus
- Sanofi
- Merck & Co., Inc.
- Pfizer Inc.
- Moderna Inc.
- Sinovac
- BioNTech SE
- AstraZeneca
Competitive Benchmarking
Operating Strategies
Competitive Edge
Weaknesses
Mature Players: Pfizer Inc.
- Mature players focus on expanding vaccine portfolios, strengthening global manufacturing capacity, securing regulatory approvals, and forming partnerships with governments and healthcare agencies.
- They also invest heavily in research and development for advanced vaccines, including RSV, influenza, pneumococcal, and combination vaccines to maintain long-term market leadership and competitive positioning.
- Mature vaccine companies maintain competitive advantage through strong global distribution networks, large-scale manufacturing infrastructure, diversified product portfolios, financial strength, and long-standing relationships with public health organizations.
- Their ability to support large immunization programs and invest continuously in vaccine innovation helps sustain their dominant market position globally.
- Mature players face challenges including high operational and research costs, strict regulatory compliance requirements, pricing pressures from government-funded immunization programs, and fluctuating demand for pandemic-related vaccines.
- Large organizational structures may also reduce flexibility and slow response times to rapidly changing healthcare and market conditions.
Emerging Players: BioNTech SE
- Emerging players prioritize innovation-driven strategies focused on advanced vaccine technologies such as mRNA and recombinant platforms.
- They emphasize rapid clinical development, strategic collaborations, licensing agreements, and targeted investments in next-generation vaccines to accelerate commercialization and strengthen their market presence against established pharmaceutical companies.
- Emerging players gain competitive advantage through technological innovation, agility, and rapid response capabilities for emerging infectious diseases.
- Their expertise in mRNA and next-generation vaccine platforms enables faster vaccine development timelines, improved adaptability, and stronger positioning in pandemic preparedness and personalized vaccine development markets.
- Emerging players often face limited manufacturing capacity, lower brand recognition, funding dependency, and restricted global distribution capabilities.
- They may also experience difficulties in scaling production, achieving widespread regulatory approvals, and competing with established pharmaceutical companies that possess stronger financial resources and broader commercial infrastructure.
Recent Developments
-
In December 2025, University of Oxford launched the world’s first Phase II clinical trial of a Nipah virus vaccine candidate, the ChAdOx1 NipahB vaccine, in Bangladesh in partnership with the International Centre for Diarrhoeal Disease Research, Bangladesh and funded by the Coalition for Epidemic Preparedness Innovations, and that the trial had started earlier that month and was set to enrol 306 healthy participants aged 18 to 55, noting that the disease could be fatal in up to 75% of cases and that since 1998 there had been 750 recorded cases with 415 deaths
-
In October 2025, Australia’s CSL Ltd, through its vaccine arm CSL Seqirus, signed a collaboration with Vaccine Industrial Company and Saudi Arabia’s Health Ministry to localize manufacturing of cell-based seasonal and pandemic influenza vaccines at a facility in Sudair City, following a period when CSL abandoned plans to complete the spin-off of Seqirus in fiscal 2026, and noting that influenza causes nearly 4,440 deaths annually in Saudi Arabia with 96 percent of intensive care patients in 2024 having not been vaccinated, with the agreement intended to help the country prepare for a pandemic in 2026 and supply vaccines for the 2026/27 season.
-
In December 2025, Sanofi agreed to buy U.S. biotech Dynavax Technologies for about USD 2.2 billion (1.9 billion euros) to strengthen its adult vaccine portfolio, with the deal valuing Dynavax at roughly USD 15.50 per share in cash, representing a 39 percent premium over its closing price of USD 11.13 on December 23, 2025, adding Dynavax’s marketed adult hepatitis B vaccine that had generated USD 90 million in third-quarter 2025 sales and is forecast to reach peak annual sales of USD 609 million in the U.S., along with an experimental shingles vaccine, and with the acquisition expected to close in the first quarter of 2026 without changing Sanofi’s 2025 financial guidance
Vaccine Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 78.13 billion
Revenue forecast in 2033
USD 115.77 billion
Growth rate
CAGR of 5.78% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Indication, type, route of administration, age group, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Serum Institute of India Pvt. Ltd.; Seqirus; Sanofi; Merck & Co., Inc.; Pfizer Inc.; Moderna Inc.; Sinovac; BioNTech SE; AstraZeneca
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Vaccine Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global vaccine market report based on indication, type, route of administration, age group, distribution channel, and region:
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Viral Vaccines
-
Hepatitis
-
Pediatric (Children)
-
Adult
-
-
Influenza
-
Pediatric (Children)
-
Adult
-
-
HPV
-
Pediatric (Children)
-
Adult
-
-
MMR
-
Pediatric (Children)
-
Adult
-
-
Rotavirus
-
Pediatric (Children)
-
Adult
-
-
Herpes Zoster
-
Pediatric (Children)
-
Adult
-
-
Japanese Encephalitis
-
Pediatric (Children)
-
Adult
-
-
RSV
-
Pediatric (Children)
-
Adult
-
-
Others
-
Pediatric (Children)
-
Adult
-
-
-
Bacterial Vaccines
-
Meningococcal Diseases
-
Pediatric (Children)
-
Adult
-
-
Pneumococcal diseases
-
Pediatric (Children)
-
Adult
-
-
DPT
-
Pediatric (Children)
-
Adult
-
-
Others
-
Pediatric (Children)
-
Adult
-
-
-
Cancer Vaccines
-
Pediatric (Children)
-
Adult
-
-
Allergy Vaccines
-
Pediatric (Children)
-
Adult
-
-
Other
-
Pediatric (Children)
-
Adult
-
-
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Subunit Vaccines
-
Recombinant Vaccines
-
Conjugate Vaccines
-
Toxoid Vaccines
-
-
Inactivated
-
Live Attenuated
-
mRNA Vaccines
-
Viral Vector Vaccines
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Oral
-
Parenteral
-
Nasal
-
-
Age Group Outlook (Revenue, USD Million, 2021 - 2033)
-
Pediatric
-
Adult
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital & Retail Pharmacies
-
Government Suppliers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Delivered Customizations
This report has been delivered with the following In-depth customizations
Client Request
Customization Delivered
Value Adds
RSV Adult Vaccine Commercial Opportunity Assessment (U.S. & Europe)
Conducted a targeted assessment of the adult RSV vaccine opportunity across the U.S., Germany, France, Italy, Spain, and the U.K. The study evaluated age-wise target populations, hospitalization burden, ACIP and EMA recommendation impact, pricing benchmarks of GSK and Pfizer vaccines, pharmacy-based vaccination trends, and payer reimbursement scenarios. The analysis also included physician adoption behavior and retail channel penetration forecasts.
Helped the client identify priority launch markets, estimate peak revenue potential, optimize pricing strategy, and understand competitive positioning against existing RSV vaccine manufacturers across high-value adult immunization markets.
mRNA Oncology Vaccine Partnership & Pipeline Benchmarking
Delivered a detailed benchmarking study on mRNA cancer vaccine developers focusing on BioNTech, Moderna, and personalized neoantigen vaccine startups. The assessment covered Phase I–III oncology pipelines, partnership structures, manufacturing scalability, lipid nanoparticle technology access, intellectual property positioning, and clinical success probability across melanoma, lung cancer, and colorectal cancer indications.
Enabled the client to identify acquisition and licensing targets, evaluate white-space opportunities in therapeutic vaccines, and prioritize investment decisions in high-potential oncology vaccine platforms with strong commercialization pathways.
Vaccine Cold Chain & Local Manufacturing Feasibility Study for Africa
Conducted a country-level feasibility assessment for vaccine manufacturing and cold-chain expansion across Kenya, South Africa, Nigeria, and Egypt. The study analyzed fill-finish infrastructure, import dependency, WHO prequalification readiness, storage bottlenecks, donor funding participation, and regional procurement dynamics involving Gavi and UNICEF vaccine programs.
Supported infrastructure investment planning by identifying high-demand vaccine categories, local production incentives, supply security risks, and partnership opportunities to strengthen regional vaccine accessibility and long-term procurement stability.
Frequently Asked Questions About This Report
Key factors that are driving the market growth include rising demand for better healthcare infrastructure and high awareness levels of the benefits of immunization.
The global vaccine market size was estimated at USD 72.75 billion in 2025 and is expected to reach USD 78.13 billion in 2026.
The global vaccine market is expected to grow at a compound annual growth rate of 5.78% from 2026 to 2033 to reach USD 115.77 billion by 2033.
The Viral Vaccines segment dominated the market with the largest revenue share of 61.17% in 2025, supported by structurally embedded demand from national immunization programs, regulatory prioritization, and continued platform investments.
Some key players operating in the vaccine market include Serum Institute of India Pvt. Ltd., Seqirus, Sanofi, Merck & Co., Inc., Pfizer Inc., Moderna Inc., Sinovac, BioNTech SE and AstraZeneca
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









