- Home
- »
- Medical Devices
- »
-
Investigational New Drug CDMO Market Size Report, 2033GVR Report cover
![Investigational New Drug CDMO Market Size, Share & Trends Report]()
Investigational New Drug CDMO Market (2025 - 2033) Size, Share & Trends Analysis Report By Service (Contract Development, Contract Manufacturing), By End Use (Pharmaceutical Companies, Biotech Companies), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-537-3
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Investigational New Drug CDMO Market Summary
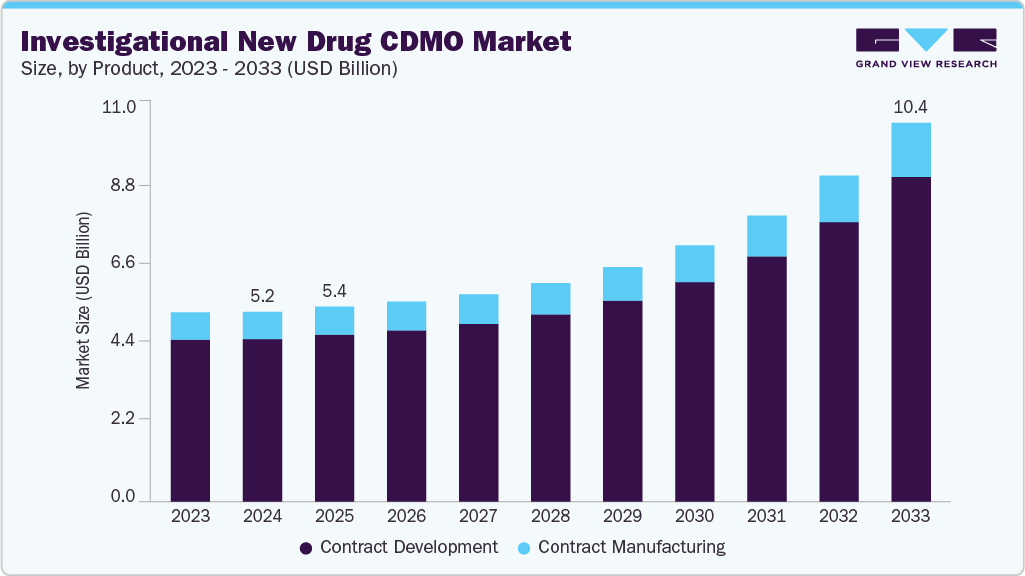
The global investigational new drug CDMO market size was estimated at USD 5.21 billion in 2024 and is projected to reach USD 10.40 billion by 2033, growing at a CAGR of 8.66% from 2025 to 2033. The investigational new drug (IND) CDMO market has been driven by rising outsourcing demand from pharmaceutical & biotechnology companies, increasing pipeline complexity, and growing need for cost efficiency among companies.
Key Market Trends & Insights
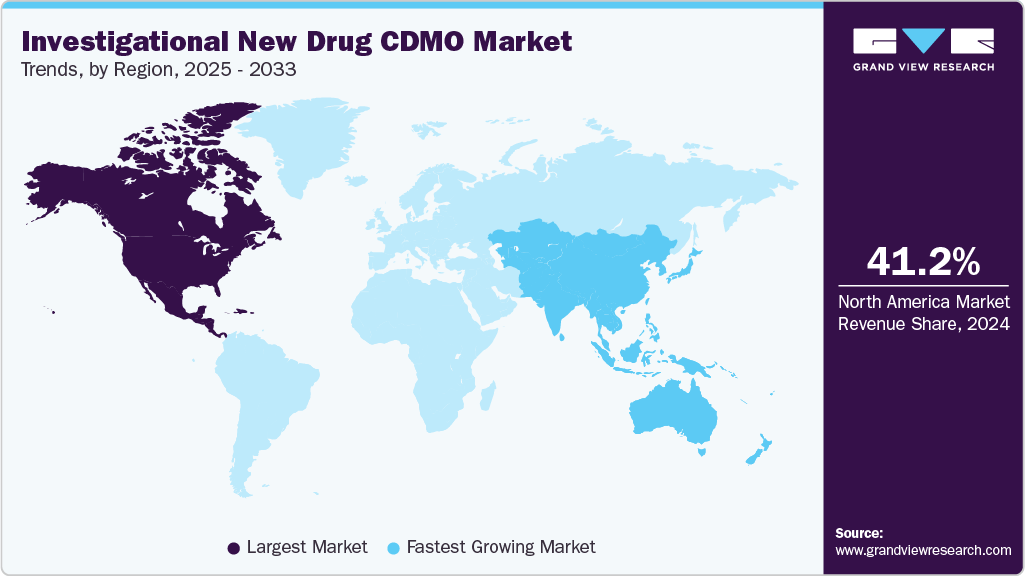
- North America investigational new drug CDMO industry held the largest share of 41.2% of the global market in 2024.
- The investigational new drug CDMO industry in the U.S. is expected to grow significantly over the forecast period.
- Based on service, the contract development segment held the highest market share in 2024.
- Based on end use, the pharmaceuticals companies segment held the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 5.21 Billion
- 2033 Projected Market Size: USD 10.40 Billion
- CAGR (2025-2033): 8.66%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Besides, increased emphasis on accelerating time-to-market, while early-phase development risks have been managed through partnerships with specialized CDMOs. In addition, growing therapeutic focus on oncology, rare diseases, and advanced biologics has further fueled IND new drug CDMO services.
Moreover, growing technological advancements have been adopted to strengthen CDMO service offerings across the IND space. Besides, artificial intelligence, advanced analytical platforms, organoid-based preclinical models, and digital regulatory systems have been integrated into operations to enable faster, data-rich development. Single-use bioprocessing and continuous manufacturing have provided flexibility in early-stage production, ensuring scalability and efficiency. Furthermore, investments have been directed toward expanding laboratory infrastructure, specialized bioassay capabilities, and digital quality systems to capture higher-value IND projects. In addition, strategic capital allocated to mergers, acquisitions, and alliances are enabling CDMOs to broaden service portfolios and expand global footprints. Private equity and venture capital activity has continued to support niche CDMOs, particularly those focused on biologics and cell and gene therapy.
Furthermore, the regulatory landscape has remained a critical determinant of IND CDMO performance. Increased scrutiny from the U.S. FDA, EMA, and other regional agencies has been observed, requiring adherence to evolving quality standards. Data integrity, transparency in early toxicology and bioanalytical submissions, and compliance with good laboratory and manufacturing practices have been emphasized, creating challenges and opportunities for differentiation.
Overall, the IND CDMO market has been characterized by rising demand, rapid technological adoption, and strategic expansion. In addition, market participants have been operating under competitive pressures to provide integrated, end-to-end solutions while balancing cost efficiency and compliance. This growth has been supported by globalized operations, diversified portfolios, and a focus on collaborative innovation, positioning CDMOs as indispensable partners in the early stages of drug development.
Opportunity Analysis
The investigational new drug (IND) CDMO industry is experiencing several new growth opportunities fueled by biopharma companies increasingly outsourcing early-stage development to accelerate their clinical entry. Besides, the rising complexity of biologics, cell and gene therapies, and oncology pipelines fuels the demand for specialized IND-enabling services such as toxicology, formulation, analytical validation, and GMP production. In addition, emerging biotech start-ups often lack in-house capabilities, leading to increased reliance on CDMOs, contributing to market growth. Moreover, regulatory pressures for high-quality submissions enhance the value of CDMOs with strong compliance expertise and global alignment.
Furthermore, the expansion of drug-device combination products and personalized therapies is creating new growth opportunities for CDMOs that offer integrated development and manufacturing solutions. Besides, expansion across regions such as North America, Europe, Asia Pacific, and Latin America provides both cost advantages and access to diverse patient populations for early-phase trials. Thus, these dynamics highlight the importance of CDMOs as critical partners, presenting opportunities for innovation, flexible capacity, and strategic collaborations with pharmaceutical and biotechnology companies.
Impact of U.S. Tariffs on the Global Investigational New Drug CDMO Market
The imposition of U.S. tariffs has significantly impacted the investigational new drug CDMO industry, as many of these organizations depend on international sources for active pharmaceutical ingredients (APIs), biologic materials, and essential laboratory supplies. Besides, increasing costs due to tariff measures have led to operational challenges, including delays in IND-enabling studies and strained profit margins, especially for smaller CDMOs with limited ability to absorb these financial pressures. In addition, supply chain disruptions have hindered GMP manufacturing and the timely production of clinical trial materials, ultimately impacting the timelines for IND submissions and causing reallocation of resources among sponsors seeking cost-effective outsourcing solutions.
The overall IND CDMO market demonstrates resilience against these challenges, fueled by a robust demand for biologics, cell and gene therapies, and oncological research. The effect of tariffs has spurred CDMOs to localize operations, reshape their supply chains, and pursue diversified network strategies, opening new avenues for U.S.-based firms to meet emerging demands. While larger CDMOs have managed disruptions through multi-national sourcing, smaller companies are exploring partnerships to mitigate risks. Thus, growing demand for innovative therapies, combined with regulatory acceleration programs & continued outsourcing trends, strengthens the essential role of CDMOs in advancing IND initiatives, proving their adaptability amidst geopolitical hurdles.
Technological Advancements

Technological advancements are reshaping the Investigational New Drug (IND) CDMO industry, enabling faster, more reliable, and cost-efficient development pathways. Artificial intelligence and machine learning are now central to optimizing drug discovery, predicting toxicity, and accelerating candidate selection, thereby shortening timelines for IND submissions. Besides, advanced analytical and bioassay technologies provide precise pharmacokinetic, biomarker, and efficacy data, strengthening regulatory packages and reducing the risk of clinical trial failures. In addition, on the manufacturing side, single-use bioprocessing and continuous manufacturing systems offer flexibility, faster scale-up, and contamination control, which are vital for small-batch IND material production, especially in biologics and advanced therapies. Moreover, in preclinical research, organoids and 3D cell culture models transform drug screening by mimicking human tissue more accurately, improving translational predictability, and reducing reliance on animal studies. Furthermore, digital quality and regulatory platforms, including eQMS and blockchain-enabled traceability, ensure compliance, streamline audit readiness, and enhance data integrity, positioning CDMOs as indispensable, technology-driven partners in early-stage drug development.
Pricing Model Analysis

In the market, pricing models are designed to balance sponsor needs with service provider sustainability. Milestone-based pricing links payments to key development achievements, such as toxicology completion or IND submission, reducing risk for sponsors while ensuring CDMOs are rewarded for progress. The fixed-fee model provides predictable costs for clearly defined tasks like formulation or analytical testing, which is ideal for projects with limited scope changes. In addition, value-based pricing aligns compensation with the impact delivered, such as risk reduction or accelerated regulatory approval, making it attractive for high-value biologics and cell therapies. Moreover, the subscription/retainer model offers sponsors ongoing access to expertise and infrastructure through recurring fees, ensuring capacity availability and predictable revenue for CDMOs. Thus, these models support flexibility, strengthen partnerships, and cater to diverse sponsor needs across the IND development lifecycle.
Market Concentration & Characteristics
The investigational new drug CDMO industry growth stage is moderate, at an accelerating pace. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
IND CDMOs demonstrate high innovation by integrating AI-driven analytics, organoid models, and continuous manufacturing platforms. These advancements enhance predictive accuracy, streamline IND-enabling studies, and reduce clinical risks. Innovation is concentrated in biologics, cell, and gene therapy pipelines requiring specialized, early-stage expertise.

M&A activity in the IND CDMO sector remains strong, with large players acquiring niche firms specializing in early-phase development, biologics, and advanced therapies. Consolidation strengthens service portfolios, expands geographic reach, and enables CDMOs to capture greater market share in IND-focused outsourcing.
Regulations significantly shape the IND CDMO market, with stringent FDA, EMA, and NMPA requirements driving demand for high-quality, compliant services. CDMOs with regulatory expertise gain a competitive advantage, helping sponsors navigate complex submissions and improve the likelihood of IND approval success.
CDMOs are expanding service portfolios beyond traditional chemistry and manufacturing into integrated IND-enabling solutions, including toxicology, bioanalysis, regulatory consulting, and clinical trial material supply. This diversification strengthens client partnerships, reduces outsourcing fragmentation, and positions CDMOs as one-stop providers for early development.
Regional expansion is accelerating, with North America and Europe as innovative hubs, while Asia Pacific and Latin America attract attention for cost efficiency and patient diversity. CDMOs invest in global footprints to support cross-border IND submissions and sponsor needs.
Service Insights
Based on service, in 2024, the contract development segment held the largest market share in the market, accounting for a revenue share of 85.60%. The segment is gaining momentum due to increasing pharmaceutical & biotech companies outsourcing and growing early-stage R&D to accelerate drug pipelines. Besides, contract development provides comprehensive IND-enabling support, including preclinical studies, bioanalysis, toxicology, formulation, and GMP manufacturing. In addition, by integrating regulatory expertise with advanced technologies, CDMOs ideally support sponsors in navigating complex submission requirements while reducing cost and development risks. Moreover, growing demand for biologics, cell, and gene therapies is expanding outsourcing opportunities with tailored solutions enabling faster progression from discovery to clinical trials. Thus, these factors position contract development as a critical partner for IND advancement.
The contract manufacturing segment is expected to grow significantly during the forecast period. Most pharmaceutical and biotechnology companies, such as CDMOs, depend on external expertise for drug development. CDMOs offer end-to-end services including formulation, process optimization, analytical testing, and production of clinical trial materials. This enables faster time-to-market and lowers R&D costs. Besides, the rising demand for biologics, gene & cell therapies, and complex molecules is fueling the CDMOs' requirements in the industry. Moreover, contract manufacturing regulatory compliance & quality assurance support market growth. Furthermore, strategic collaborations, technological innovations, and capacity expansion are enhancing the role of CDMOs, making contract development an essential part of accelerating healthcare innovation, further providing scalable, high-quality therapies across the globe. Such factors are expected to drive the market growth over the estimated period.
End Use Insights
In terms of end use, in 2024, the pharmaceutical companies segment accounted for the largest revenue share in the market. The segment is driven by increasing collaboration between pharmaceutical companies & IND-focused CDMOs and rising focus on drug development pipelines. Besides, CDMOs ideally provide essential services across preclinical research, formulation, toxicology, analytical testing, and GMP manufacturing, enabling pharmaceutical firms to advance investigational new drug (IND) applications efficiently, further contributing to segment growth. In addition, outsourcing to specialized partners helps companies reduce operational costs, accelerate timelines, and gain access to innovative technologies. Moreover, pharmaceutical innovators ideally benefit from CDMOs’ regulatory expertise and flexible capacity, particularly in biologics, oncology, and advanced therapies. This strategic initiative further strengthens clinical readiness while allowing pharmaceutical companies to focus resources on core innovation and late-stage commercialization.
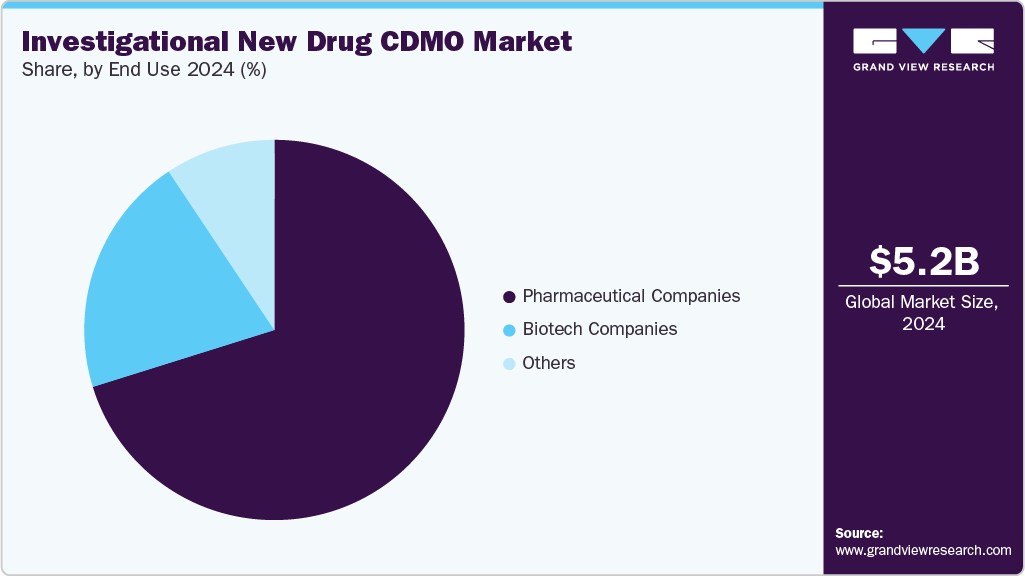
The biotech companies segment is anticipated to grow at the second fastest CAGR during the forecast period. Biotechnology companies have increasingly relied on Investigational New Drug (IND) CDMOs to accelerate early-stage development and mitigate operational risks. As biotech pipelines expand in oncology, rare diseases, and advanced biologics, CDMOs are engaged to provide specialized expertise in bioanalytics, formulation, toxicology, and manufacturing. Partnerships have been structured to reduce capital expenditure while ensuring regulatory compliance and scalability. By leveraging advanced technologies such as AI-driven analytics, single-use systems, and digital regulatory tools, CDMOs have supported biotech firms in navigating complex IND submissions. This collaboration has enabled faster timelines, cost efficiency, and greater innovation, making CDMOs strategic allies in biotech growth. Such factors are expected to drive the market growth.
Regional Insights
North America investigational new drug CDMO industry dominated globally in 2024, holding a revenue share of 41.16%.The market growth is attributed to strong regulatory frameworks, robust R&D pipelines, and demand for accelerated drug approvals. Besides, CDMOs in the region support biotech and pharmaceutical innovators with preclinical formulation, toxicology, and GMP manufacturing solutions. In addition, strategic collaborations, advanced analytics, and flexible capacity drive growth, while high investment in biologics, cell, and gene therapies enhances outsourcing needs. North America’s established infrastructure, venture funding, and government support for clinical development position it as a leading hub for IND-related services. Such factors are expected to drive the market over the estimated time period.
U.S. Investigational New Drug CDMO Market Trends
The investigational new drug CDMO industry in the U.S. accounted for the largest market share in the North America market, owing to the rising number of biotechnology start-ups, an innovation-driven ecosystem, and supportive FDA pathways. Companies increasingly rely on CDMOs for early-phase development, IND-enabling studies, and scalable manufacturing to meet complex regulatory requirements. Besides, established market players and niche players enhance service diversity across small molecules, biologics, and advanced therapies, which are expected to drive market growth over the estimated time period. Rising costs of in-house development, coupled with accelerated FDA programs, are driving partnerships between CDMOs and drug developers, strengthening the U.S. as the central IND outsourcing destination.
The Canada investigational new drug CDMO industry is expected to grow at a significant CAGR during the forecast period. The market is driven by the strong clinical research networks, government incentives, and a growing biotechnology ecosystem. Besides, local CDMOs supporting early-stage development with formulation, toxicology, bioanalysis, and GMP capabilities are expected to cater to domestic and cross-border clients. In addition, increasing emphasis on biologics, vaccines, and advanced therapies boosts demand for specialized IND services, further contributing to the market. The Canadian regulatory landscape, aligned with U.S. and European standards, provides a favorable environment for outsourcing. Collaboration with academic research institutions and incentives for innovation further position the country as a competitive contributor in IND-focused CDMO services in the market.
Europe Investigational New Drug CDMO Market Trends
The Europe investigational new drug CDMO industry is driven by diverse biotech clusters, strong regulatory compliance under EMA oversight, and rising demand for early-phase outsourcing. CDMOs in the region offer specialized expertise in small molecule development, biologics, and cell/gene therapies, helping sponsors navigate IND-equivalent clinical trial authorizations. Strategic investments in flexible capacity, sustainability initiatives, and advanced analytical platforms are strengthening competitiveness. Countries such as Germany, the UK, France, and Switzerland also fuel innovation, supporting biotech start-ups and pharmaceutical companies in accelerating IND submissions.
The investigational new drug CDMO industry in Germany held the highest share in 2024. This growth can be attributed to a robust pharmaceutical industry, advanced infrastructure, and high R&D expenditure. Besides, CDMOs provide comprehensive support for IND-enabling studies, ranging from preclinical services to GMP manufacturing. In addition, the country has a strong demand for biologics and biosimilar development with emphasis on quality, compliance, and technological innovation. Besides, partnerships with academic research centers & global pharma companies support enhancing capabilities in early-stage programs. Moreover, its strong regulatory alignment with EMA standards and role as a European innovation hub make it a preferred destination for IND outsourcing. Such factors are expected to drive the market over the estimated time period.
The UK investigational new drug CDMO industryis expected to grow significantly over the forecast period. The country's growth is fueled by strong biotech innovation, favorable regulatory pathways, and leading clinical research capabilities. Besides, CDMOs in the country provide tailored solutions in formulation, toxicology, and early-phase manufacturing to support sponsors in achieving IND or CTA approvals. In addition, growing cell and gene therapy development, with specialized infrastructure, is expected to drive the market over the estimated period. Moreover, government supports and venture capital funding fuel innovation in the market. Furthermore, Brexit mentioned that increased investment in domestic R&D capacity enhances opportunities for CDMOs in IND development, further contributing to market growth.
Asia Pacific Investigational New Drug CDMO Market Trends
Asia Pacific investigational new drug CDMO industry is expected to grow at a significant CAGR over the forecast period.The market is driven by rising cost advantages, the developing biotechnology industry, and supportive government initiatives. Besides, market players operating in the dynamic outsourcing environment include China, Japan, India, South Korea, and Australia, which are known for their expertise in small molecule development, biologics, and biosimilars, with a growing emphasis on cell & gene therapy pipelines. Besides, efforts toward regulatory harmonization & strategic partnerships with Western sponsors enhance market credibility & foster cross-border collaboration. In addition, rising venture funding and clinical trial activities boost demand for early-phase services. Thus, expanding capacity and efficiency in this region positions it as a competitive hub for IND CDMO growth.
The investigational new drug CDMO industry in China is witnessing new growth opportunities due to substantial government investment, a large domestic patient population, and evolving regulatory reforms. CDMOs in China offer various IND-enabling solutions, including preclinical studies, formulation development, and GMP manufacturing. There is a rising demand for biologics, biosimilars, and innovative therapies, which enhances outsourcing partnerships, particularly from global sponsors looking for cost-effective services. In addition, expanding infrastructure in the country, advanced manufacturing technologies, and a skilled workforce make it a favored destination for early-phase development. In addition, ongoing efforts to align with international regulatory standards and foster cross-border collaborations are propelling China's integration into the global IND CDMO landscape.
The investigational new drug CDMO market in Japan is driven by a well-established pharmaceutical ecosystem, advanced manufacturing capabilities, and a strong focus on biologics & regenerative medicine innovation. Besides, CDMOs ideally support sponsors by facilitating IND-enabling studies, toxicology assessments, and the production of early-phase clinical materials, all under the oversight of the PMDA. In addition, robust government support for cell & gene therapies establishes Japan as a frontrunner in developing advanced therapies. Moreover, collaborations among domestic pharmaceutical companies, academic institutions, and international partners further enrich the service offerings. With an emphasis on precision medicine and high-quality standards, Japan provides a highly competitive landscape for IND outsourcing.
India investigational new drug CDMO industry is experiencing rapid expansion attributed to cost efficiency, skilled scientific talent, and increasing integration with global pharmaceutical supply chains. Besides, domestic CDMOs in the country ideally support preclinical development, toxicology, formulation, and GMP production for IND submissions across small molecules and biologics. In addition, rising demand from global biotech and pharma companies has increased investments in the country for infrastructure, digital platforms, and technological innovations. Moreover, the country has increased focus on innovative generics therapy pipelines, contributing to the early-phase outsourcing market. Furthermore, with favorable government policies and growing clinical trial activity, the country is emerging as a competitive IND CDMO destination, which is expected to drive the market over the estimated period.
Latin America Investigational New Drug CDMO Market Trends
The investigational new drug CDMO industry in the Latin America region is expected to witness significant growth over the estimated time period. In Latin America, the market is developing steadily, supported by an increased clinical trial activity, supportive government initiatives, and strategic partnerships with multinational pharmaceutical companies. Besides, countries such as Brazil and Mexico have emerged as regional hubs offering services in early-phase development, formulation, and GMP compliance. In addition, CDMOs in the region are enhancing their capabilities to meet international standards, making them more attractive to global sponsors looking for cost-effective solutions. Moreover, rising demand for biologics and vaccines further fuels the investment in specialized services. However, the regulatory landscape in the region is likely to be challenging due to its diversity. Collaboration with U.S. and European companies is fueling the credibility & positioning of Latin America as an increasingly important player in the IND CDMO services sector.
Brazil investigational new drug CDMO industryis driven by a robust local pharmaceutical industry, supportive government policies, and expanding clinical trial activities. CDMOs offer various services, including toxicology, formulation, and GMP manufacturing, essential for early-stage drug development. Besides, the market in the country is fueled by rising demand for biologics, biosimilars, and vaccines, as well as collaborations with multinational sponsors. In addition, regulatory reforms are enhancing transparency and aligning with global standards, making the country an attractive outsourcing hub for IND services. Moreover, domestic innovation and increasing healthcare demand further fuel the market's potential.
Middle East & Africa Investigational New Drug CDMO Market Trends
The investigational new drug CDMO industry in the MEA region is expected to experience steady growth due to rising government healthcare investments, growing clinical research activities, and the developing pharmaceutical industry. Countries such as the UAE, Saudi Arabia, and South Africa are also emerging as hotspots in the region for formulation, toxicology, and early-phase manufacturing capabilities. In addition, collaborations among multinational sponsors and regional CDMOs are becoming more common, allowing access to cost advantages and expanding patient pools. Also, the presence of ongoing harmonization efforts for regulatory scenarios is improving in the region with global standards. Furthermore, rising demand for biologics & vaccines further enhances opportunities, positioning the region as a strategic growth area for IND CDMO services.
UAE investigational new drug CDMO industry is experiencing growth driven by strong government support, innovative healthcare policies, and substantial investments in pharmaceutical R&D. Besides, local CDMOs offer essential early-phase services such as formulation, bioanalysis, and the supply of clinical trial materials, contributing to market growth. In addition, strategic collaborations with multinational drug developers are enhancing service capabilities, especially in biologics and vaccines. Moreover, the UAE’s regulatory alignment with global standards and its strategic geographic location position it as a competitive outsourcing destination. Furthermore, rapidly growing biotech initiatives are further accelerating the growth of IND CDMO services in the country.
Key Investigational New Drug CDMO Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in June 2025, Samsung Biologics mentioned the launch of “Samsung Organoids,” an advanced drug screening platform aimed at strengthening drug discovery and development. The solution enables precision screening to forecast patient-specific responses, optimize preclinical workflows, and shorten the path to investigational new drug (IND) submissions by data-driven evaluation of candidate molecules.
Key Investigational New Drug CDMO Companies:
The following are the leading companies in the investigational new drug CDMO market. These companies collectively hold the largest market share and dictate industry trends.
- Catalent, Inc.
- Lonza
- Recipharm AB
- Siegfried Holding AG
- Patheon Inc.
- Covance
- IQVIA Holdings Inc.
- Cambrex Corporation
- Charles River Laboratories International, Inc.
- Syneos Health
- Syngene International
- Jubilant Pharmova
- Piramal Pharma Solutions
- Dr. Reddy’s CPS
- Dishman Carbogen Amcis Ltd.
- Biocon Biologics
- Laurus Labs
Recent Developments
-
In April 2025, Porton Advanced announced its CDMO collaboration with Tasly Pharmaceutical Co., Ltd. on the development of the innovative dual-targeting CAR-T therapy, “P134 Cell Injection,” which recently received IND clearance from China’s NMPA. The therapy is being developed for the treatment of recurrent glioblastoma.
-
In March 2024, Lonza mentioned an agreement for the acquisition of the manufacturing facility in the U.S. from Roche for USD 1.2 billion. This is expected to significantly increase Lonza's capacity to produce biologic drugs & expand its presence in the U.S. Lonza plans to invest an additional USD 500 million to upgrade the facility and meet the demand for next-generation biologic therapies.
Investigational New Drug CDMO Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 5.35 billion
Revenue forecast in 2033
USD 10.40 billion
Growth rate
CAGR of 8.66% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Catalent, Inc.; Lonza; Recipharm AB; Siegfried Holding AG; Patheon Inc.; Covance; IQVIA Holdings Inc.; Cambrex Corporation; Charles River Laboratories International, Inc.; Syneos Health; Syngene International; Jubilant Pharmova; Piramal Pharma Solutions; Dr. Reddy’s CPS; Dishman Carbogen Amcis; Biocon Biologics; Laurus Labs
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Investigational New Drug CDMO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global investigational new drug CDMO market report based on service, end use, and region.
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Small Molecule
-
Bioanalysis and DMPK Studies
-
Toxicology Testing
-
Pathology and Safety Pharmacology Studies
-
Drug Substance Development
-
Synthetic Route Development
-
Process Development
-
Form Selection Crystallization Process Development
-
Scale-up of Drug Substance
-
-
Formulation Development
-
Pre Formulation
-
Preclinical Formulation Selection
-
First In Man Formulation/ Process Development
-
-
Analytical & Quality Services
-
Analytical Method Development / Validation
-
Release Testing of Drug Substance and Drug Product
-
Formal Stability of Drug Substance and Drug Product
-
-
Process Optimization
-
Work Up Purification Steps
-
Telescoping & Process Refining
-
Initial Optimization
-
-
-
Large Molecule
-
Cell Line Development
-
Process Development
-
Upstream
-
Microbial
-
Mammalian
-
Others
-
-
Downstream
-
MABs
-
Recombinant Proteins
-
Others
-
-
-
-
-
Contract Manufacturing
-
Small Molecule
-
Oral Solids
-
Semi-Solids
-
Liquids & Injectables
-
Others
-
-
Large Molecule
-
MABs
-
Recombinant Proteins
-
Others
-
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotech Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global investigational new drug CDMO market size was estimated at USD 5.21 billion in 2024 and is expected to reach USD 5.35 billion in 2025.
b. The global investigational new drug CDMO market is expected to grow at a compound annual growth rate (CAGR) of 8.66% from 2025 to 2033 to reach USD 10.40 billion by 2033.
b. The contract development dominated the investigational new drug CDMO market in 2024 with a market share of 85.60%. The segment is gaining momentum due to increasing pharmaceutical & biotech companies outsourcing and rising early-stage R&D to accelerate drug pipelines. Besides, contract development provides comprehensive IND-enabling support, including preclinical studies, bioanalysis, toxicology, formulation, and GMP manufacturing, which further contributes to segment growth.
b. Some of the key market players include Catalent, Inc., Lonza, Recipharm AB, Siegfried Holding AG, Patheon Inc., Covance, IQVIA Holdings Inc., Cambrex Corporation, Charles River Laboratories International, Inc., Syneos Health, Syngene International, Jubilant Pharmova, Piramal Pharma Solutions, Dr. Reddy’s CPS, Dishman Carbogen Amcis Ltd., Biocon Biologics, and Laurus Labs among others.
b. Key factors contributing to market growth are rising outsourcing demand from pharmaceutical & biotechnology companies, increasing pipeline complexity, and the growing need for cost efficiency among companies. Besides, increased emphasis on accelerating time-to-market, while early-phase development risks have been managed through partnerships with specialized contributes to market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










