- Home
- »
- Medical Devices
- »
-
Latin America Pharmaceutical Contract Manufacturing & Research Services Report, 2033GVR Report cover
![Latin America Pharmaceutical Contract Manufacturing And Research Services Market Size, Share & Trends Report]()
Latin America Pharmaceutical Contract Manufacturing And Research Services Market (2026 - 2033) Size, Share & Trends Analysis Report by Service (Manufacturing, Research), By Region (Brazil, Colombia, Argentina, Chile, Venezuela, Peru, Uruguay, Ecuador, Bolivia), And Segment Forecasts
- Report ID: GVR-1-68038-745-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Latin America Pharmaceutical Contract Manufacturing & Research Services Market Summary
The Latin America pharmaceutical contract manufacturing and research services market size was estimated at USD 20.2 billion in 2025 and is projected to reach USD 37.2 billion by 2033, growing at a CAGR of 8.1% from 2026 to 2033. The market is driven by cost efficiency, expanding clinical research capabilities, rising demand for outsourced services, and improving regulatory frameworks.
Key Market Trends & Insights
- The Brazil pharmaceutical contract manufacturing & research services market held the largest share in the Latin America market in 2025.
- Based on service, the manufacturing segment held the largest market share of 66.3% in 2025.
- Based on service, the research service segment is expected to grow significantly during the forecast period.
Market Size & Forecast
- 2025 Market Size: USD 20.2 Billion
- 2033 Projected Market Size: USD 37.2 Billion
- CAGR (2026-2033): 8.1%
- Brazil: Largest market in 2025
Some other factors contributing to market growth are the rising demand for generics & biosimilars, coupled with the growing need for local pharmaceutical production, which further drives the need for outsourcing services. In addition, investments from global pharmaceutical companies, improvements in healthcare infrastructure, and government initiatives are promoting domestic manufacturing and innovation. Besides, growing strategic partnerships and technological advancements are boosting regional competitiveness.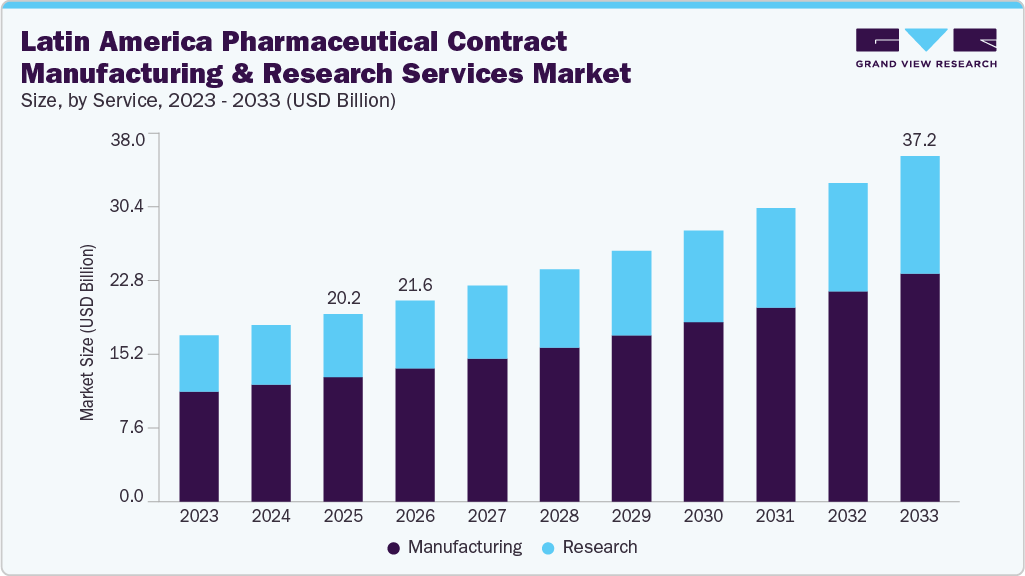
The increasing focus on cost efficiency has encouraged pharmaceutical companies to outsource biopharmaceutical production to specialized contract manufacturers. Besides, the expansion of the pharmaceutical and biologics industry, the rising prevalence of chronic diseases, the increasing healthcare expenditure, and the growing demand for affordable medicines are driving pharmaceutical companies to scale up production. Thus, growing outsourcing to contract manufacturers and research organizations is helping companies meet demand without heavy capital investment.
In addition, product innovation in the market is no longer confined to in-house R&D within large biopharma companies; instead, Latin America’s smaller and mid-sized biotech firms are playing a critical role in discovering novel molecules, which are often later acquired by established players. This shift has led contract research organizations (CROs) to increasingly secure partnerships and contracts with emerging biotech companies, ensuring sustained service demand and volume growth.
Moreover, strong government initiatives in Latin America aim to enhance domestic production and healthcare independence. Countries like Brazil and Mexico are actively supporting local biologics manufacturing through public funding, tax incentives, and technology transfer programs. In Brazil, government-supported partnerships under public healthcare policies have led to a significant increase in local biologics production, thereby reducing reliance on imports. Besides, regulatory bodies such as ANVISA have simplified approval processes, facilitating scaling of manufacturing operations. These policy-driven initiatives are strengthening regional capabilities, attracting investment, and driving growth in the pharmaceutical manufacturing sector across Latin America.
Furthermore, companies in the region are strengthening their global footprint by upgrading and expanding domestic specialty manufacturing facilities while actively pursuing international collaborations. The growing presence of market players such as Thermo Fisher Scientific and IQVIA in key countries, including Brazil, Mexico, Argentina, and Chile, is further accelerating market growth. In addition, supportive regulatory improvements, increasing clinical trial activity, and a growing skilled workforce are enhancing the region’s attractiveness as a hub for pharmaceutical development and outsourcing services.
Opportunity Analysis
The Latin American pharmaceutical contract manufacturing and research services market presents substantial growth opportunities, driven by evolving outsourcing strategies and the expansion of regional markets. The market is expected to witness new growth opportunities driven by the increasing trend of nearshoring by North American pharmaceutical companies to Latin America hubs. These companies are looking to enhance supply chain resilience, reduce transportation costs, and mitigate geopolitical risks, making Latin America a favorable outsourcing destination. In addition, rising demand for biologics & biosimilars and strong potential for clinical research activities are further contributing to the market growth. Most global pharmaceutical companies are increasingly outsourcing complex manufacturing processes, which requires CDMOs in the region to develop advanced capabilities. As the region also holds strong potential for clinical research, driven by a diverse and sizable patient population, this facilitates quicker patient recruitment and shortens clinical trial timelines.
Moreover, the growth of the domestic pharmaceutical industry, alongside the rising demand for generic drugs, further enhances opportunities for contract manufacturers to expand their operations. Government initiatives encouraging local production and attracting foreign investments are also contributing to market growth. Furthermore, integrating digital technologies, such as automation and data analytics, enables service providers to improve efficiency, ensure regulatory compliance, and enhance overall service quality, thereby strengthening their competitive advantage in the market. Such factors are expected to drive the market growth.
Impact of U.S. Tariffs on the Latin America Pharmaceutical Contract Manufacturing & Research Services Market
U.S. tariffs on pharmaceutical imports have significantly affected the Latin America pharmaceutical contract manufacturing and research services market by altering cost structures, supply chains, and outsourcing strategies. These tariffs raise the costs of imported raw materials, intermediates, and finished pharmaceutical products, thereby increasing overall manufacturing and development expenses for pharmaceutical companies. This shift has led to changes in global supply chains, with companies increasingly exploring alternative regions to reduce tariff-related risks. Thus, companies are reconsidering their sourcing and production strategies to retain cost efficiency.
Moreover, the region is emerging as an attractive nearshoring destination due to cost benefits and geographical proximity to the U.S. However, the imposition of tariffs also introduces uncertainty in trade policies, which may delay investment decisions and outsourcing agreements with CROs and CDMOs within the region. Furthermore, broader economic slowdowns stemming from trade tensions can affect pharmaceutical R&D spending and reduce demand for outsourcing. However, despite these factors, the tariffs are driving supply chain diversification, ultimately fueling Latin America's growth as a hub for pharmaceutical outsourcing services.
Technological Advancements
Technological advancements are significantly reshaping the pharmaceutical contract manufacturing and research services market in Latin America by improving efficiency, quality, and competitiveness on a global scale. The incorporation of advanced bioprocessing technologies, such as single-use systems and enhanced cell culture techniques, enables more flexible, cost-effective production of biologics and biosimilars. Moreover, the use of automation and digital manufacturing solutions, including robotics and manufacturing execution systems, minimizes human error, enhances consistency, and ensures regulatory compliance. The integration of data analytics and artificial intelligence is also revolutionizing drug development and manufacturing by facilitating better decision-making, predictive maintenance, and enhanced clinical trial outcomes. Continuous manufacturing technologies are gaining momentum, enabling uninterrupted production, minimizing waste, maintaining consistent product quality, and speeding up production timelines.

In addition, digital clinical trial solutions, such as decentralized trials and remote patient monitoring, are streamlining patient recruitment, improving data accuracy, and lowering overall trial costs. These technological innovations empower CROs and CDMOs in Latin America to offer high-quality, efficient, and scalable services. This competitive outsourcing service within the pharmaceutical landscape is expected to drive the market growth.
Pricing Model Analysis
In the Latin America pharmaceutical contract manufacturing and research services (CRAMS) market, pricing models are used to balance risk, profitability, and client satisfaction. Cost-plus pricing ensures predictable margins by charging the actual cost of services plus a fixed profit, offering transparency for complex projects.
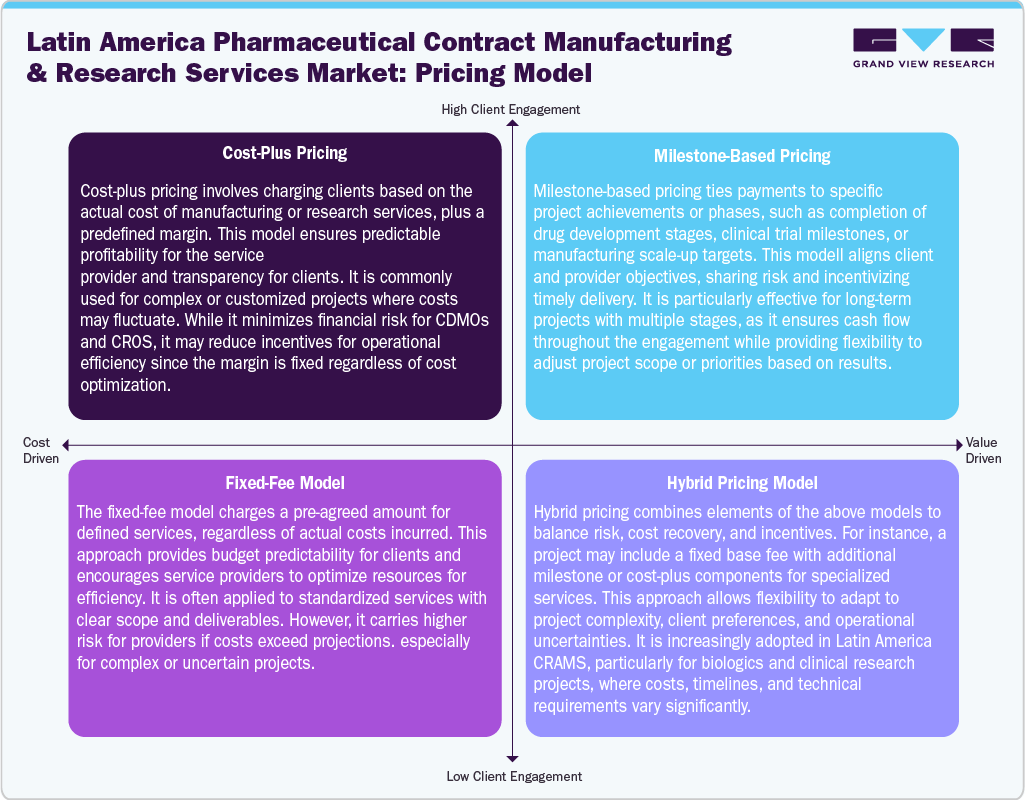
Milestone-based pricing links payments to project achievements, aligning incentives and facilitating cash flow across multi-stage drug development or clinical trials. Fixed-fee models provide budget predictability, encouraging efficiency but requiring accurate cost forecasting to avoid overruns. Increasingly, hybrid models combine these approaches, incorporating fixed fees for standard services and cost-plus or milestone elements for specialized or high-risk activities. This flexibility is particularly valuable for biologics, biosimilars, and advanced clinical research projects in Latin America, where operational complexity and technical requirements vary. Thus, these models enable CDMOs and CROs to manage risk, optimize resources, and maintain competitiveness while meeting diverse client needs.
Market Concentration & Characteristics
The Latin America pharmaceutical contract manufacturing & research services market is in an emerging growth stage, characterized by the degree of innovation, regulatory impact, level of M&A activities, service expansion, and regional expansion.
Innovation is a key growth driver in the Latin America CRAMS sector, as contract development and manufacturing organizations (CDMOs) and contract research organizations (CROs) increasingly adopt advanced technologies. These include single-use systems, continuous manufacturing processes, and digital solutions. Although the region is still developing compared to more mature markets, the growing adoption of biologics, biosimilars, and data-driven processes is driving efficiency, cost reductions, and enhanced service quality.

M&A activities in Latin America are on the rise as both global and regional companies aim to expand their capabilities and geographic reach. Strategic partnerships and acquisitions provide a pathway to access new technologies, strengthen infrastructure, and penetrate high-growth segments such as biologics and clinical research. This consolidation enhances overall competitiveness and enables service providers to offer integrated solutions to pharmaceutical companies.
The regulatory landscape in Latin America is improving, with agencies increasingly aligning with international standards, such as Good Manufacturing Practices (GMP). Countries like Brazil and Mexico are enhancing their compliance systems, which not only boosts credibility but also adds complexity to operations. Companies with strong regulatory knowledge stand to benefit from expedited approvals and greater trust from multinational clients.
Market providers in the region are broadening their offerings from basic manufacturing and research to comprehensive end-to-end solutions encompassing drug development, clinical trials, and commercial-scale production. This integrated approach helps attract global clients, fosters long-term contracts, and amplifies revenue potential across the pharmaceutical value chain.
Regional expansion is expected to drive the establishment of cost-effective manufacturing and research hubs across Latin America. This strategy improves supply chain efficiency, enhances market access, and supports scalability. It also enables service providers to meet both local and international demands, reinforcing Latin America’s position as an emerging hub for global outsourcing.
Service Insights
On the basis of service, in 2025, the manufacturing segment held the largest market share in the market, accounting for a revenue share of 66.3%. The segment growth is attributed to rising demand for generics, biologics, and biosimilars, local regulatory improvements, the presence of multinational pharmaceutical companies, and lower labor, operational, and infrastructure costs compared to other regions. In addition, a growing trend among pharmaceutical companies to outsource the entire therapeutic development process from drug substance (API) production to finished drug products due to limited in-house capacity and specialized expertise is anticipated to drive the market growth. Moreover, growing investment in advanced manufacturing technologies and facility expansion enable CDMOs to handle complex formulations, scale efficiently, and meet growing regional and international market demand. Such factors are expected to drive market growth over the forecast period.
Furthermore, the manufacturing segment includes advanced drug delivery formulations, API/bulk drugs, packaging, and finished-dose formulations. The finished dose formulations segment is expected to grow significantly during the forecast period. Finished Dose Formulations include tablets, capsules, injectables, liquids, and inhalables. This segment is driven by growing outsourcing requirements among pharmaceutical companies to leverage regional cost advantages, scalable manufacturing infrastructure, and regulatory-compliant facilities. Besides, CDMOs offer end-to-end capabilities, from formulation development and stability testing to large-scale production and packaging, which further drives market growth. In addition, growing focus on high-quality, ready-to-market products enables faster commercialization and reduces time-to-market for multinational clients.
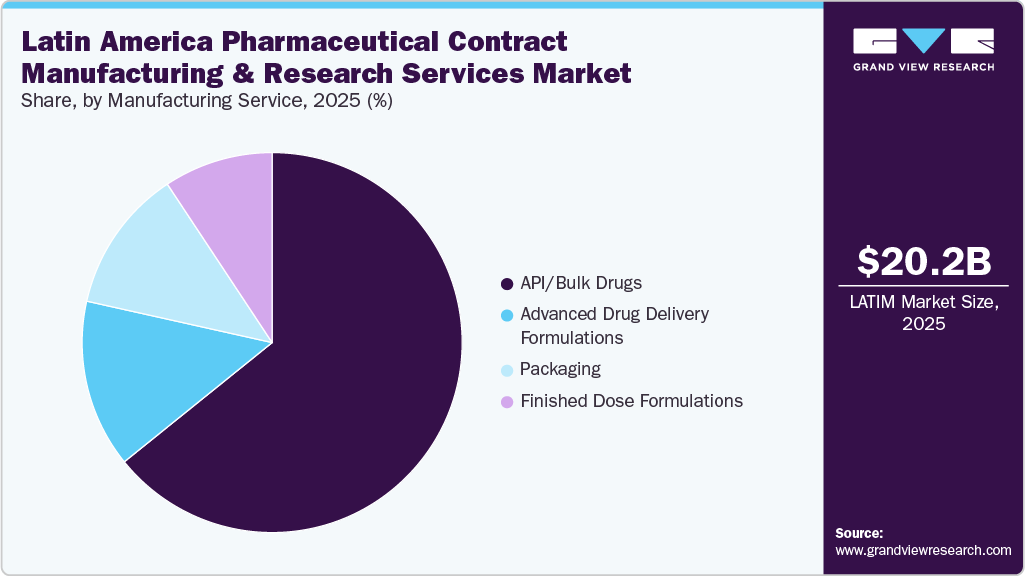
The research service segment is expected to grow significantly during the forecast period. Research services in the Latin American pharmaceutical contract manufacturing and research services market involve a range of offerings from CROs, including preclinical, clinical, and formulation development support. Key services encompass drug discovery, toxicology studies, pharmacokinetics, clinical trial management, and regulatory assistance. The segment growth is driven by growing cost-effective, efficient, and compliant research solutions. The service offers a range of advantages, including diverse patient populations, faster patient recruitment, and a pool of experienced scientific talent. By outsourcing research services to CROs, companies can expedite drug development timelines, reduce operational costs, and tap into specialized expertise, thereby positioning CROs as partners in advancing therapeutics from initial discovery to clinical development.
Regional Insights
The pharmaceutical contract manufacturing & research services industry in Latin America isexpected to significantly grow over the estimated time period. The market is driven by rising demand for local Contract Development and Manufacturing Organizations (CDMOs) and Contract Research Organizations (CROs) amid nearshoring and supply chain diversification. Besides, the growing focus on biologics, biosimilars, and advanced drug delivery systems, along with technological advancements such as single-use systems, continuous manufacturing, and digital solutions, is expected to drive market growth. Moreover, rising strategic partnerships and mergers and acquisitions are broadening capabilities and the industry's geographic reach. Furthermore, regulatory alignment with international standards is enhancing credibility, integrated end-to-end services spanning from research to finished-dose formulations, which are strengthening long-term partnerships with clients and boosting market competitiveness.
Brazil Pharmaceutical Contract Manufacturing & Research Services Market
The Brazil pharmaceutical contract manufacturing & research services industryis driven by a well-established pharmaceutical industry, advanced manufacturing infrastructure, and alignment with international regulatory standards. Besides, the country attracts multinational pharmaceutical companies seeking cost-effective outsourcing solutions for APIs, finished dose formulations, and biologics. In addition, the country benefits from a large, diverse patient population, supporting clinical research and trial activities. Investments in advanced manufacturing technologies, single-use systems, and integrated services strengthen the capabilities of local CDMOs and CROs. Furthermore, strategic partnerships, government support, and expanding service portfolios are fueling the market growth.
Argentina Pharmaceutical Contract Manufacturing & Research Services Market
The pharmaceutical contract manufacturing & research services industry in Argentinais driven by an expanding pharmaceutical industry, a skilled workforce, and cost-effective operations. The country is increasingly attracting multinational pharmaceutical companies looking to outsource services in areas such as active pharmaceutical ingredients (APIs), finished formulations, and clinical research, as they seek to expand in the region. In addition, improving the regulatory framework to align more closely with international standards, and providing further support to ensure quality and compliance for global clients. In addition, ongoing investments in advanced manufacturing technologies, analytical services, and biologics capabilities are further enhancing the offerings of local CDMOs and CROs. Furthermore, strategic partnerships, along with government support for pharmaceutical innovation, drive the country as a competitive outsourcing destination within the Latin American market.
Key Latin America Pharmaceutical Contract Manufacturing & Research Services Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in April 2025, Novo Nordisk announced a plan to invest roughly 6.4 billion reais (≈USD 1.09 billion) to expand its manufacturing facility in Montes Claros to increase the production capacity for injectable therapies, including GLP‑1 drugs used in diabetes and obesity treatment, a signal of Brazil’s importance as both a production hub and export base. These company‑level commitments reflect confidence in Brazil’s contract manufacturing environment and contribute to the region’s broader pharmaceutical ecosystem development.
Key Latin America Pharmaceutical Contract Manufacturing & Research Services Companies:
- Boehringer Ingelheim International GmbH
- Fresenius Kabi AG
- Unither Pharmaceuticals
- Lonza
- Pfizer Inc.
- Charles River Laboratories
- Laboratory Corporation of America Holdings
- IQVIA, Inc.
- Recipharm AB.
- AbbVie, Inc.
- Catalent, Inc.
- West Pharmaceutical Services, Inc.
Recent Developments:
- In March 2025, Benuvia Operations, LLC, a CDMO, announced that it had obtained Good Manufacturing Practices certification (Certificado de Boas Práticas de Fabricação, CBPF) from Brazil’s National Health Surveillance Agency.
Latin America Pharmaceutical Contract Manufacturing & Research Services Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 21.6 billion
Revenue forecast in 2033
USD 37.2 billion
Growth rate
CAGR of 8.1% from 2026 to 2033
Actual Data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, region
Regional scope
Latin America
Country scope
Brazil; Argentina; Colombia; Chile; Venezuela; Peru; Uruguay; Ecuador; Bolivia
Key companies profiled
Boehringer Ingelheim International GmbH; Fresenius Kabi AG; Unither Pharmaceuticals; Lonza; Pfizer Inc.; Charles River Laboratories; Laboratory Corporation of America Holdings; IQVIA, Inc.; Recipharm AB.; AbbVie, Inc.; Catalent, Inc.; West Pharmaceutical Services, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Latin America Pharmaceutical Contract Manufacturing & Research Services Market Report Segmentation
This report forecasts revenue growth at regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Latin America pharmaceutical contract manufacturing & research services market report based on service and region.
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Manufacturing
-
API/Bulk Drugs
-
Advanced Drug Delivery Formulations
-
Packaging
-
Finished Dose Formulations
-
Solid
-
Liquid
-
Semi-solid Formulations
-
-
-
Research
-
Oncology
-
Vaccines
-
Inflammation & Immunology
-
Cardiology
-
Neuroscience
-
Others
-
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Latin America
-
Brazil
-
Colombia
-
Argentina
-
Chile
-
Venezuela
-
Peru
-
Uruguay
-
Ecuador
-
Bolivia
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the Latin America pharmaceutical contract manufacturing and research services market include Boehringer Ingelheim International GmbH, Fresenius Kabi AG, Unither Pharmaceuticals, Lonza, Pfizer Inc., Charles River Laboratories, Laboratory Corporation of America Holdings, IQVIA, Inc., Recipharm AB., AbbVie, Inc., Catalent, Inc., and West Pharmaceutical Services, Inc., among others.
b. Key factors that are driving the Latin America pharmaceutical contract manufacturing and research services market growth include cost efficiency, expanding clinical research capabilities, rising demand for outsourced services, and improving regulatory frameworks. Some other factors contributing to market growth are the rising demand for generics and biosimilars, coupled with the growing need for local pharmaceutical production, which further drives the need for outsourcing services.
b. The Latin America pharmaceutical contract manufacturing and research services market size was estimated at USD 20.2 billion in 2025 and is expected to reach USD 21.6 billion in 2026.
What is the Latin America pharmaceutical contract manufacturing and research services market growth?b. The Latin America pharmaceutical contract manufacturing and research services market is expected to grow at a compound annual growth rate of 8.1% from 2026 to 2033 to reach USD 37.2 billion in 2033.
b. The manufacturing segment dominated the Latin America pharmaceutical contract manufacturing and research services market with a share of 66.33% in 2025. The market growth is attributed to the rising demand for generics, biologics, and biosimilars, local regulatory improvements, the presence of multinational pharmaceutical companies, and lower labor, operational, and infrastructure costs. In addition, a growing trend among pharmaceutical companies to outsource the entire therapeutic development process due to limited in-house capacity and specialized expertise is anticipated to drive the market growth
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










