- Home
- »
- Medical Devices
- »
-
Live Biotherapeutic Products And Microbiome CDMO Market 2030GVR Report cover
![Live Biotherapeutic Products And Microbiome CDMO Market Size, Share & Trends Report]()
Live Biotherapeutic Products And Microbiome CDMO Market (2025 - 2030) Size, Share & Trends Analysis Report By Application (C. Difficile, Crohn’s Disease, IBS, Diabetes), By Region (North America, Europe, APAC, MEA), And Segment Forecasts
- Report ID: GVR-4-68040-061-6
- Number of Report Pages: 137
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Live Biotherapeutic Products And Microbiome CDMO Market Summary
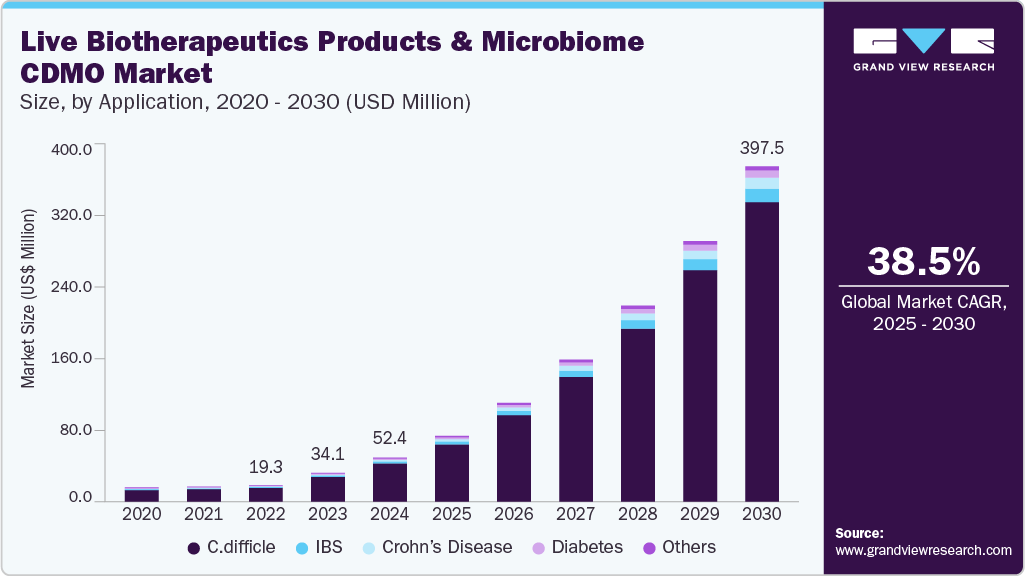
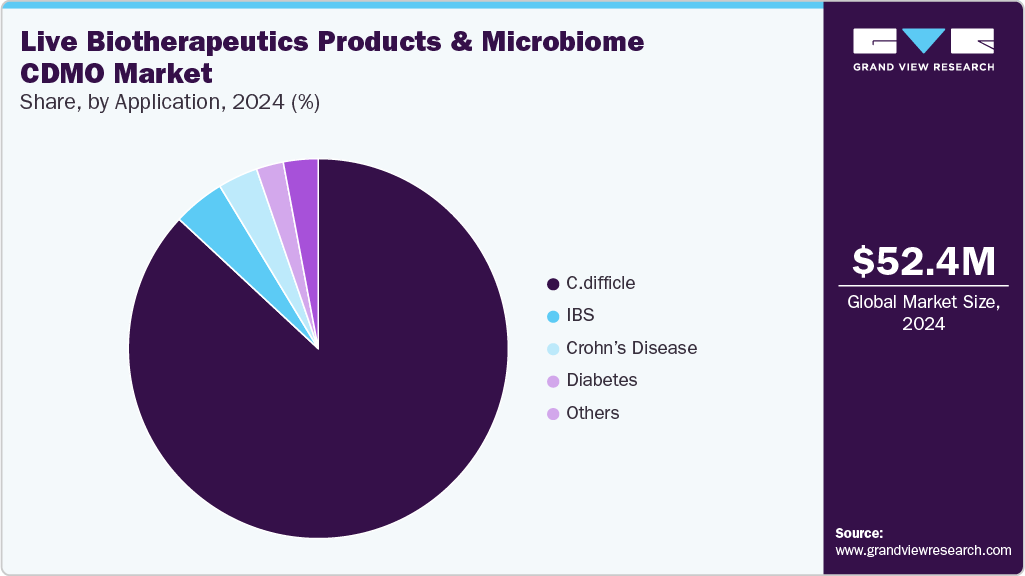
The global live biotherapeutic products and microbe CDMO market size was estimated at USD 52.37 million in 2024 and is projected to reach USD 397.49 million by 2030, growing at a CAGR of 38.5% from 2025 to 2030. The rising importance of the potential advantages of live biotherapeutic products and microbiome-based therapeutics in treating various diseases, including inflammatory bowel disease, cancer, and neurological disorders, among patients and healthcare providers, is the prime factor propelling the market growth.
Key Market Trends & Insights
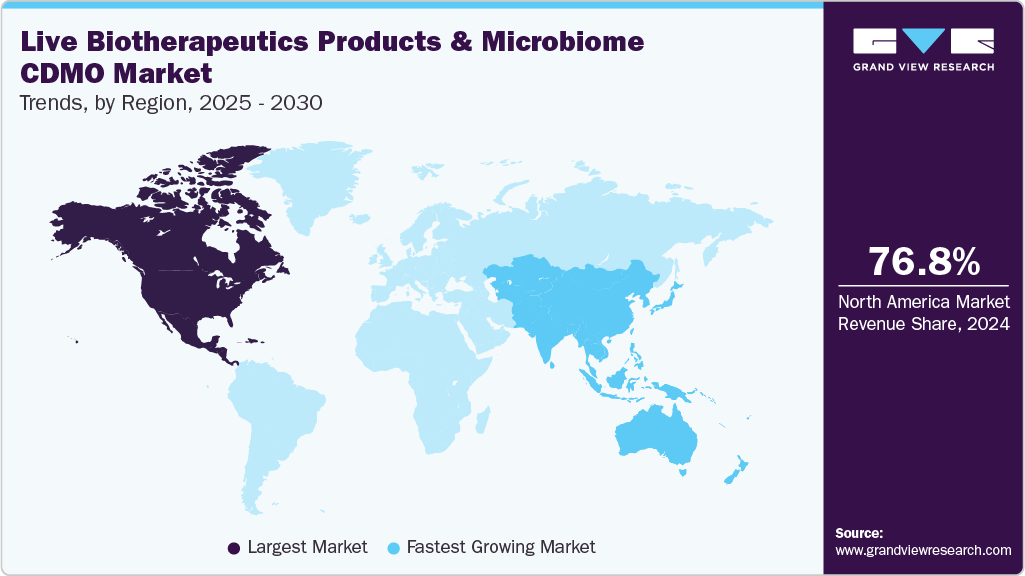
- North America live biotherapeutic products and microbiome CDMO market held the largest revenue share of 76.84% in 2024.
- The U.S. live biotherapeutic products and microbiome CDMO industry led North America with the largest share in 2024.
- By application, the C. Difficile segment held the largest market share of 86.93% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 52.37 Million
- 2030 Projected Market Size: USD 397.49 Million
- CAGR (2025-2030): 38.5%
- North America: Largest market in 2024
Furthermore, the increasing prevalence of chronic diseases and the growing demand for personalized medicine also expand the live biotherapeutic products and microbiome-based therapeutics market. CDMOs specializing in these fields have the expertise and infrastructure to support the development and manufacturing of these complex therapies, which require specialized equipment and facilities. As a result, pharmaceutical and biotech companies can outsource their production to CDMOs, allowing them to focus on research and development. Moreover, a growing interest in LBPs and microbiomes as potential therapeutic agents leads to increased research and development activities in these areas. This has resulted in a greater demand for CDMOs with expertise in these fields. Advances in sequencing technologies and bioinformatics have allowed for a deeper understanding of the microbiome and its role in health and disease. This has enabled the development of more targeted and effective live biotherapeutic products and microbiome-based therapies.
Furthermore, regulatory bodies, such as the FDA, have recognized the potential of live biotherapeutic products and microbiomes and have guided the development and approval of these products. This has increased investor confidence and encouraged companies to invest in developing these products. Overall, the LBPs and microbiome CDMO market is expected to experience significant growth in the coming years due to these drivers and the increasing interest in these innovative therapies.
Opportunity Analysis
The live biotherapeutic products and microbiome CDMO market is projected to witness significant innovation, such as innovative microbiome science and personalized medicine, thereby accelerating market demand. Increasing detection of the microbiome’s impact on human health boosts demand for customized live microbial therapies, requiring specialized and scalable manufacturing solutions. CDMOs with expertise in advanced bioprocessing are well-equipped to support biotech companies in efficiently transforming complex microbial formulations into safe, consistent, and regulatory-compliant products. As regulatory guidelines evolve and clinical validation progresses, CDMOs that offer adaptable manufacturing platforms combined with stringent quality assurance will capitalize on emerging opportunities in the development of novel therapeutics targeting chronic and immune-related conditions, thereby catalyzing growth in the microbiome-based treatment landscape.
Technology Landscape
The technology landscape in the live biotherapeutic products and microbiome CDMO market is characterized by the integration of next-generation microbial cultivation platforms, precision fermentation systems, and advanced formulation techniques tailored to living organisms. Unlike conventional biologics, LBPs demand closed-loop bioprocessing technologies that maintain strict anaerobic or microaerophilic conditions and real-time monitoring tools to ensure viability and potency throughout production. In addition, lyophilization, encapsulation, and cryopreservation innovations are being adapted to enhance shelf stability and target delivery of live microbes. CDMOs are also adopting modular cleanroom designs, automated sterile fill-finish systems, and digital quality management solutions to meet microbiome-based therapeutics' unique scalability, traceability, and regulatory needs.
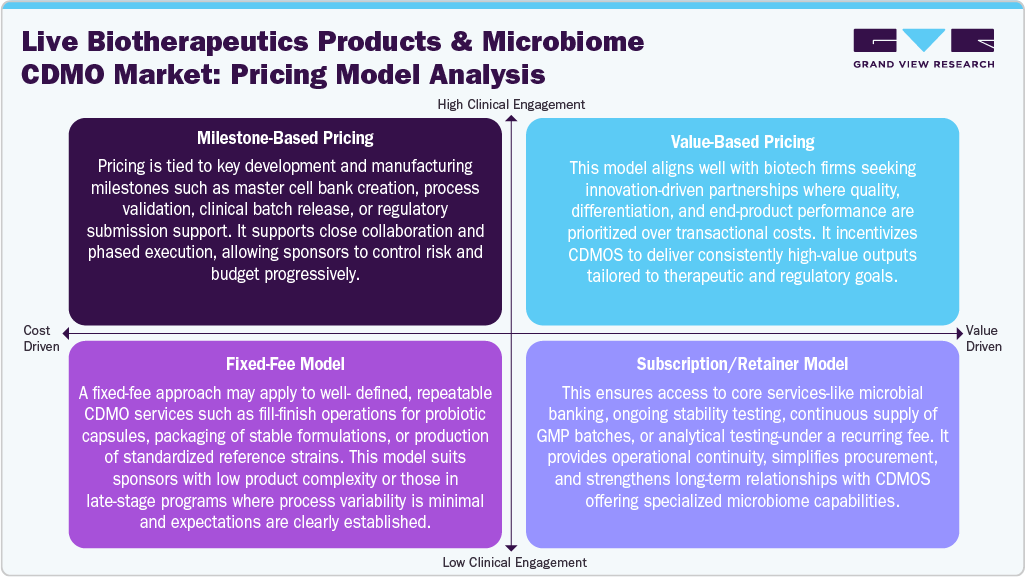
Pricing Analysis
Pricing models in the live biotherapeutic products and microbiome CDMO market are evolving to consider the complexity, customization, and regulatory sensitivity of live microbial manufacturing. Traditional per-batch or volume-based pricing is often inadequate due to the highly specialized nature of LBP development, which requires tailored fermentation, handling of strict anaerobes, and viability-preserving fill-finish processes. As a result, CDMOs are increasingly adopting hybrid models that combine milestone-based pricing for early-stage development with premium service tiers for GMP manufacturing, stability testing, and long-term storage. Pricing is also influenced by the need for rapid scale-up, integration of quality-by-design (QbD) principles, and the incorporation of advanced analytics, making cost structures more value-driven and collaborative in nature.
Market Concentration & Characteristics
The live biotherapeutic products and microbiome CDMO market's growth stage is stable and expected to accelerate over the estimated period. The market is characterized by technologies, regulatory considerations, globalization, and outsourcing of product processes to influence advantages and specialized capabilities.
The live biotherapeutic products and microbiome CDMO innovations are continuously evolving to meet the industry’s demands due to the increased burden of infectious & rare diseases. Several advancements have shaped the live biotherapeutic products and microbiome CDMO market, enhancing drug development, manufacturing processes, and productivity.

Compliance with stringent regulatory requirements, particularly in the pharmaceutical industry, is critical. CDMOs in this market emphasize robust quality assurance practices and adherence to regulatory standards, thereby witnessing lucrative growth opportunities.
Live biotherapeutic products and microbiome CDMO players in the market leverage strategies such as collaborations, partnerships, and acquisitions to promote the reach of their offerings and increase their product capabilities globally.
Increasing R&D activities, rising number of mergers & acquisitions, & growing disease burden can influence market dynamics positively.
The local presence of several established pharmaceuticals, the rising burden of infectious and rare diseases, and the growing demand for CDMOs fuel market growth.
Application Insights
C. Difficile segment held a market share of 86.93% in 2024. The live biotherapeutic products and microbiome market is segmented by application into C. Difficile, Crohn's disease, IBS, Diabetes, and Others. C. difficile is a bacterium found in the human gut microbiome and can cause severe diarrhea and colitis, particularly in people who have recently taken antibiotics.
In recent years, there has been growing interest in using live biotherapeutic products to treat various diseases, including those related to the gut microbiome. Live biotherapeutic products are living organisms, such as bacteria or viruses, used to prevent or treat diseases. C. difficile is one of the organisms that can be used as an LBP, and some research has been done into its potential use for treating C. difficile infections. As research into the human microbiome and the potential applications of LBPs continues to grow, the market will likely show more interest in using C. difficile and other organisms for therapeutic purposes.
Regional Insights
North America live biotherapeutic products and microbiome CDMO market accounted for the largest share of 76.84% in 2024 of the global live biotherapeutic products and microbiome market. The market is driven primarily due to the presence of key players and increasing investments in research and development activities in this region. The growing demand for effective treatments for various diseases, such as C. difficile, Crohn's disease, and IBS, has also contributed to market growth in North America.
Moreover, the region has many established pharmaceutical and biotech companies with expertise in developing and commercializing biologic products, including live biotherapeutic products and microbiome-based therapies. Furthermore, the region has a well-established regulatory framework for biopharmaceuticals, which provides a stable environment for companies to develop and manufacture these products.
U.S. Live Biotherapeutic Products And Microbiome CDMO Market Trends
The live biotherapeutic products and microbiome CDMO market in the U.S. held the largest share in 2024, owing to the presence of several pharmaceutical and life sciences companies. Growing interest in clinical trials and the benefits of live biotherapeutics for rare diseases drives the development of new and novel drugs. Moreover, a well-established regulatory framework provides new opportunities for companies to develop and manufacture products with a wide range of applications and target populations with improved knowledge of microbiota's importance in health and disease.
Europe Live Biotherapeutic Products And Microbiome CDMO Market Trends
The live biotherapeutic products and microbiome CDMO market in Europe is expected to grow significantly due to the high prevalence of CDI and the local presence of well-established market players, such as 4D Pharma, Microbiotica, EnteroBiotix, Prokarium, and Eagle Genomics. Moreover, increasing funding, acquisitions, and high R&D spending in European countries contribute to market growth. For instance, in April 2023, Seres Therapeutics, Inc. & Nestle Health Science announced the U.S. FDA approval for VOWST (fecal microbiota spores, live-brpk) known as SER-109.
The live biotherapeutic products and microbiome CDMO market in Germany held the largest share in 2024. The rapidly aging population, growing demand for biotherapeutics, and emerging chronic diseases have increased the pressure on pharmaceutical companies to innovate new drugs for patients. These factors have led pharmaceutical and biotechnology companies to shift their focus on R&D to produce niche live biotherapeutic products and microbiomes.
UK live biotherapeutic products and microbiome CDMO market is anticipated to grow over the forecast period. The UK is one of the key countries in microbiome science and innovation in both the public and private sectors. The country has growing IMT and IMMP opportunities with unique clinical research and genomic & digital capabilities. Moreover, it is the key destination for microbiome investment and innovation.
Asia Pacific Live Biotherapeutic Products And Microbiome CDMO Market Trends
Asia Pacific is expected to grow at a CAGR of 44.82% during the forecast period. The market in Asia Pacific has witnessed significant growth and promising developments in recent years. Some key factors contributing to this growth are the prevalence of IBD and Crohn's disease as well as rising R&D investment and funding. Moreover, growing outsourcing and the increasing number of clinical trials are among the major factors responsible for the fastest market growth in Asia Pacific.
The live biotherapeutic products and microbiome CDMO market in China is expected to grow over the forecast period due to the rising potential for growth. Global leadership in the industry has led companies to innovate niche products such as live biotherapeutic products and microbiomes, with increasing collaboration, acquisition, funding, and FDA approvals.
Japan live biotherapeutic products and microbiome CDMO market held the largest share in 2024 due to a significant increase in the overall number of clinical trials on emerging live biotherapeutics and microbiomes. The government is undertaking steps to offer regulatory support and promote clinical trials in the country.
The live biotherapeutic products and microbiome CDMO market in India is anticipated to grow over the forecast period, owing to rapidly increasing private healthcare services and academic research, which have led to growing interest and rising awareness of live biotherapeutics. Moreover, rising infectious diseases, hospital-acquired infections, and emerging IBD burden nationwide are driving the market demand.
Latin America Live Biotherapeutic Products And Microbiome CDMO Market Trends
The Latin America Live Biotherapeutic Products and microbiome CDMO market is gaining momentum due to increasing research interest in microbiome-based health solutions and growing demand for localized, GMP-compliant manufacturing infrastructure. As regional biotech firms and academic institutions intensify efforts to explore the role of the gut and skin microbiota in addressing chronic conditions, metabolic disorders, and antibiotic resistance, the need for specialized CDMO partners has grown.
Brazil live biotherapeutic products and microbiome CDMO market is witnessing emerging trends driven by a maturing biotech ecosystem, increased government focus on innovation, and a rising demand for alternative therapies targeting chronic and lifestyle-related diseases. Local startups and research institutes are increasingly exploring native microbial strains for therapeutic development, creating a niche for CDMOs capable of handling region-specific strains under strict anaerobic and GMP-compliant conditions.
Key Live Biotherapeutic Products And Microbiome CDMO Company Insights
The major players operating across the market focus on adopting inorganic growth strategic initiatives such as mergers, partnerships, acquisitions, etc. The prominent strategies companies adopt are service launches, mergers and acquisitions/joint ventures, mergers, partnerships and agreements, expansions, and others to increase market presence and revenue and gain a competitive edge, driving market growth.
Key Live Biotherapeutic Products And Microbiome CDMO Companies:
The following are the leading companies in the live biotherapeutic products and microbiome CDMO market. These companies collectively hold the largest market share and dictate industry trends.
- Arrant Bio
- 4D Pharma
- Cerbios
- Biose Industrie
- Assembly Biosciences, Inc.
- Wacker Chemie AG
- Quay Pharmaceuticals
- NIZO
- Lonza
- Inpac Probiotics
Recent Developments
-
In February 2025, Assembly Biosciences, Inc. announced the positive Phase 1a results for ABI-1179. These results highlight Assembly Biosciences’ capabilities in advancing novel microbiome-derived therapeutics and reinforce the company’s strategic positioning within the live biotherapeutics market.
-
In February 2025 Cerbios-Pharma SA announced to expand its Lugano site for clinical and commercial manufacturing of Highly Potent Active Pharmaceutical Ingredients (HPAPIs), including cytotoxic linker payloads for antibody-drug conjugates (ADCs).
-
In March 2023, Cedars-Sinai established a new Human Microbiome Research Institute that supports investigators studying microbiomes, the diverse collection of fungi, bacteria, and viruses.
Live Biotherapeutic Products And Microbiome CDMO Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 78.05 million
Revenue forecast in 2030
USD 397.49 million
Growth rate
CAGR of 38.5% from 2025 to 2030
Actual data
2018 - 2024
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Arrant Bio; 4D Pharma; Cerbios; Biose Industrie; Assembly Bioscience Inc; Wacker Chemie AG; Quay Pharmaceuticals; NIZO; Lonza; Inpac Probiotics..
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Live Biotherapeutic Products And Microbiome CDMO Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global live biotherapeutic products and microbiome CDMO market report based on application, and region:
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
C. Difficle
-
Crohn’s Disease
-
IBS
-
Diabetes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global live biotherapeutics products and microbiome CDMO market size was estimated at USD 52.37 million in 2024 and is expected to reach USD 78.05 million in 2025.
b. The global live biotherapeutics products and microbiome CDMO market is expected to grow at a compound annual growth rate of 38.48% from 2025 to 2030 to reach USD 397.49 million by 2030.
b. North America dominated the live biotherapeutics products and microbiome CDMO market with a share of 76.8% in 2024. This is attributable to the large presence of key players and increasing investments in research and development activities in this region.
b. Some key players operating in the live biotherapeutics products and microbiome CDMO market include Arrant Bio; 4D Pharma; Cerbios; Biose Industrie; Assembly Bioscience Inc.; Wacker Chemie AG; Quay Pharmaceuticals; NIZO; Lonza; Inpac Probiotics
b. The rising importance of the potential advantages of live biotherapeutic products and microbiome-based therapeutics in treating a variety of diseases, including inflammatory bowel disease, cancer, and neurological disorders, among patients and healthcare providers is the prime factor that propels market growth
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










