- Home
- »
- Clinical Diagnostics
- »
-
Middle East Syphilis Testing Market, Industry Report, 2033GVR Report cover
![Middle East Syphilis Testing Market Size, Share & Trends Report]()
Middle East Syphilis Testing Market (2025 - 2033) Size, Share & Trends Analysis Report By Technology (Molecular Diagnostics, Immunoassay), By Location of Testing (Laboratory Testing, Public Health Labs), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-762-7
- Number of Report Pages: 236
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Syphilis Testing Market Summary
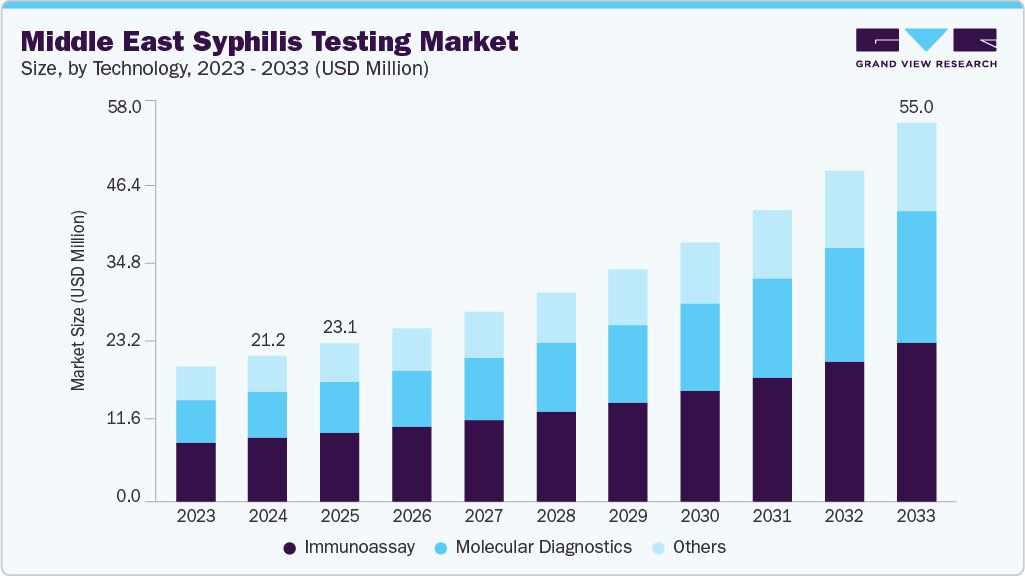
The Middle East syphilis testing market size was estimated at USD 21.25 million in 2024 and is projected to reach USD 55.01 million by 2033, growing at a CAGR of 11.47% from 2025 to 2033. Growing public health awareness and government-led initiatives are driving demand for syphilis diagnostics across the Middle East. The region has placed infectious disease control at the center of its healthcare priorities, with syphilis screening integrated into antenatal care, premarital testing, and expatriate health checks in many Gulf countries.
Rising mobility of populations, large expatriate communities, and cross-border labor flows have made early detection and prevention programs essential. Concurrently, healthcare providers in the region are rapidly adopting advanced diagnostic technologies, including point-of-care rapid tests and laboratory-based immunoassays, to strengthen surveillance and ensure timely treatment.
Saudi Arabia’s health system emphasizes centralized lab capacity and antenatal screening within public hospitals; demand for high-quality immunoassays and molecular confirmatory testing remains strong. Simultaneously, Gulf Cooperation Council (GCC) initiatives and private hospitals are open to rapid POC solutions for screening travelers, migrant worker clinics, and perinatal care. Market entry requires regulatory clearance, adherence to national procurement, and partnerships with major hospital groups. Opportunities exist for accredited dual HIV/syphilis rapid tests that meet Saudi regulatory standards and can be deployed in maternal health and occupational-health screening programs.
The prevalence of syphilis in the Middle East and North Africa (MENA) is higher than anticipated given regional norms, but it is comparable to global levels. According to a 2024 study, the prevalence was 0.48% in the general population overall, but significantly higher rates were found in high-risk groups such as female sex workers (FSW) and men who have sex with men (MSM). Although prevalence is decreasing, it is not doing so quickly enough to reach international goals. Testing includes techniques like rapid plasma reagin (RPR) and Treponema pallidum-specific tests to identify current and previous infections.
Syphilis testing in the UAE utilizes Rapid Plasma Reagin (RPR) and Treponema pallidum Hemagglutination (TPHA) tests, and a recent study in 2016 showed a 0.51% prevalence among expatriate workers undergoing health checks. Another study in 2021 found a 0.45% syphilis seropositivity rate among individuals screened in a tertiary hospital, with higher rates among non-Saudi expatriates compared to Saudis.
Table 1 Temporal Trends in Syphilis Epidemiology in the UAE 2018-2022
2018
2019
2020
2021
2022
Grand Total
Total Number of Cases
11 (4 %)
79 (27 %)
75 (26 %)
105 (36 %)
20 (7 %)
290 (100 %)
Age in Years (Mean ± SD)
39 ± 8
37 ± 9
37 ± 11
35 ± 9
32 ± 7
36 ± 10
Gender n (%)
Female
4 (36 %)
31 (39 %)
16 (21 %)
37 (35 %)
9 (45 %)
97 (33 %)
Male
7 (64 %)
48 (61 %)
59 (79 %)
68 (65 %)
11 (55 %)
193 (67 %)
Nationality n (%)
Locals
7 (64 %)
45 (57 %)
46 (61 %)
55 (52 %)
10 (50 %)
163 (56 %)
Expats
4 (36 %)
34 (43 %)
29 (39 %)
50 (48 %)
10 (50 %)
127 (44 %)
Number of Symptomatic cases
2 (18 %)
18 (23 %)
54 (72 %)
40 (38 %)
16 (80 %)
130 (45 %)
Source: Research Gate Articles, Grand View Research
The data shows a diverse but overall increasing trend in syphilis cases, with the highest numbers recorded in 2021 (105 cases, 36%). By 2022, the number of reported cases dropped to 20 (7%), suggesting improved control measures or reduced case detection/reporting. The average age of affected individuals declined gradually from 39 years in 2018 to 32 years in 2022, indicating a younger population being affected in recent years.
The distribution of genders shows a persistent male predominance, with men making up roughly two-thirds of all cases over time. In terms of nationality, the distribution was fairly balanced, with locals accounting for 56% of all cases and foreigners for 44%. Symptomatic cases increased significantly, peaking at 72% in 2020 and 80% in 2022, highlighting early or asymptomatic detection challenges. Primary and secondary syphilis remained relatively low at 3% overall, pointing toward a higher proportion of latent or late-stage diagnoses.
Market Concentration & Characteristics
The market is characterized by a moderate to high degree of innovation. Dual rapid testing is changing due to innovation. For instance, PrisesBio announced in January 2025 the launch of a syphilis test service in the UAE through its platform (syphilistestuae). The offering seeks to increase the region's access to syphilis screening, most likely by using telehealth or local distribution channels to make testing easier. This highlights the rising demand in Middle East markets for accessible and point-of-care STD diagnostics.
In the Middle East, collaborations and strategic partnerships are becoming powerful drivers of accessibility and affordability in the syphilis testing industry. While outright acquisitions remain relatively limited, regional collaborations create many of the same benefits as consolidation, from improving distribution reach to lowering costs through shared resources. These alliances are helping scale up testing capacity and make advanced diagnostics more widely available across public and private healthcare systems.

The regulatory environment in the Middle East plays a decisive role in shaping the diagnostics landscape, including the adopting of biomarker tests. Stringent requirements for medical device approvals, mandatory screening protocols for infectious diseases, and health authority oversight ensure that only validated and clinically reliable assays reach patients. For instance, in countries such as the UAE and Qatar, syphilis screening is a required component of medical exams for employment and visas, establishing a systematic path for diagnostic adoption. In addition to protecting the public's health, this emphasis on compliance increases trust in sophisticated assays like renal biomarkers, which are being evaluated increasingly for their capacity to identify early illness.
Companies have introduced rapid tests, ELISA-based platforms, and AI-enabled screening solutions, resulting in a steady expansion of diagnostic portfolios in the Middle East market. The introduction of syphilis rapid antibody testing kits and the growth of home-based STD testing services in Dubai are two recent examples. Convenience, accuracy, and speed are key components of patient-centric care, which is reflected in these advancements. Similar trends are seen in renal diagnostics as providers seek to move laboratory-based biomarker tests closer to the point of care.
Countries such as Saudi Arabia, UAE, and Qatar continue to act as a regional hub for healthcare innovation, attracting multinational companies and startups looking to expand their footprint across the Middle East. The country’s well-developed healthcare infrastructure, coupled with its role as a gateway to Gulf and North African markets, makes it a strategic entry point for new diagnostic technologies. For instance, partnerships between global firms and local providers to introduce AI-driven kidney disease detection tools underscore the UAE’s position as a launchpad for advanced diagnostics.
Technology Insights
The immunoassay segment currently holds the largest share of 43.91% in 2024. This reflects its strength in providing rapid, reliable, cost-effective tests, especially serologic tests for syphilis antibodies widely used in hospital laboratories, screening programs (e.g., antenatal care), and point-of-care settings. The appeal continues to lie in its adaptability for centralized lab workflows and simpler clinic-based or outreach use. Recent studies from 2024 show increased research into automated immunoassays for syphilis detection, with a 2024 Dubai study using chemiluminescence immunoassay for screening and another evaluating syphilis trends between 2018 and 2022. Internationally, there is a focus on integrating syphilis and HIV testing, including new over-the-counter tests, though these are not specific to Dubai.
The molecular diagnostics represents the fastest-growing technology segment, driven by rising demand for early, highly sensitive detection of infections. Their growth is driven by demand for early detection (especially of asymptomatic or early-stage infections), higher specificity, and the capability to detect pathogens directly rather than relying on immunologic response. Countries increasing lab infrastructure, modernizing regulatory frameworks, and investing in multiplex and syndromic testing panels enable this shift.
Location Of Testing Insights
Laboratory-based testing continued to represent the largest share of 59.52% of the syphilis diagnostics market in 2024 due to its well-established status as the gold standard in clinical practice. Automated immunoassays, ELISAs, and confirmatory treponemal/non-treponemal workflows that guarantee high accuracy and consistency are crucial components of centralized labs and hospital-based diagnostic centers. Because these platforms can handle high test volumes, they are especially useful for nationwide screening campaigns and prenatal care programs where dependability is crucial.
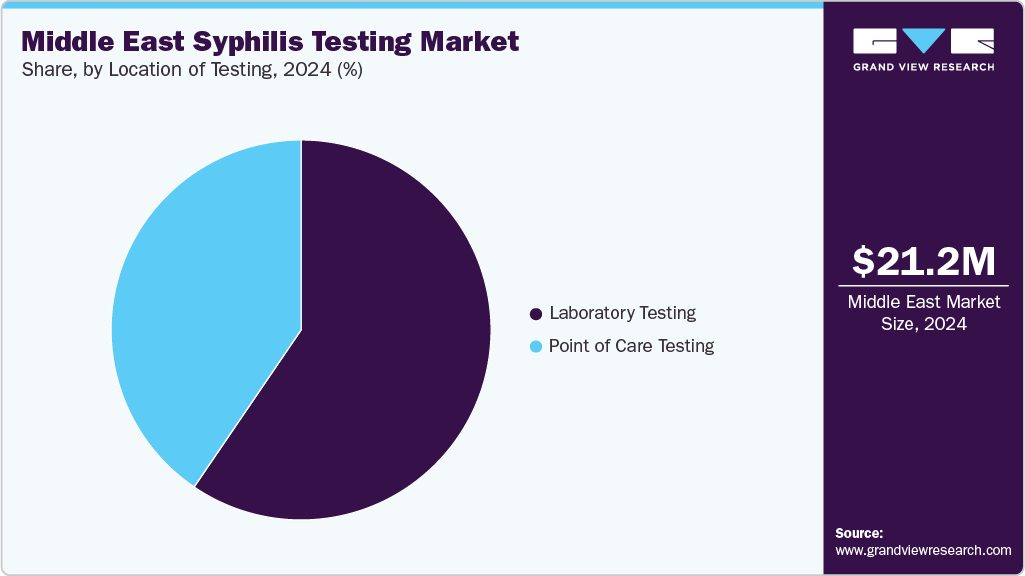
Point-of-care (POC) testing is experiencing the fastest growth, reflecting shifting public health priorities toward accessibility, speed, and patient-centered care. The surge in congenital syphilis, which demands immediate detection and treatment, has highlighted the limitations of lab-centric models that rely on multiple visits and long turnaround times. In response, regulators and companies are accelerating the adoption of rapid and at-home testing solutions. In January 2025, in February 2025, AccuBioTech expanded its product offering via the Medlab Middle East exhibition 2025 in Dubai (February 3-6), showcasing its in vitro diagnostic (IVD) rapid-test portfolio built on immunochromatography technology, including its Syphilis Rapid Test, among other infectious disease assays. The company emphasized that its rapid IVD offerings (for dengue, HIV, HCV, etc.) aim to deliver accurate, fast diagnostics to support infectious-disease screening and control. AccuBioTech's syphilis test strengthens the use of POC diagnostics. Particularly in high-prevalence areas, this trend promotes global healthcare priorities to enhance early detection, prompt treatment initiation, and infection control.
Regional Insights
The syphilis testing market in the Middle East is expanding. Countries with more modern regulatory oversight (e.g., Saudi Arabia, UAE) are better positioned to adopt new molecular technologies. Streamlined approval for IVD devices and frameworks for importing or locally validating molecular kits helps accelerate adoption.
Saudi Arabia Syphilis Testing Market Trends
The syphilis testing market in Saudi Arabia is growing, driven by increasing awareness, diagnostic capacity expansion, and supportive government initiatives. Public hospitals remain central to screening programs, especially in antenatal care, while private labs and pharmacies are stepping in to offer more accessible and rapid testing options.
UAE Syphilis Testing Market Trends
The syphilis testing market in the UAE is a growing market in the MEA. Syphilis testing is frequently included in routine health examinations, such as those needed for employment permits, highlighting the significance of these tests for public health. The tests are conducted in accredited medical facilities and hospitals using tried-and-true techniques such as TPPA (Treponema pallidum particle agglutination) and Rapid Plasma Reagin (RPR) for accuracy and dependability. In January 2025, PrisesBio launched a syphilis antibody rapid test product for the UAE market, giving locals and tourists a quicker and easier way to detect syphilis early. The announcement highlights that diagnostic confidence is ensured by UAE clinicians' extensive training in interpreting these assays. Further, in July 2025, Call My Doctor, a UAE-based healthcare provider, launched a home STD testing service in Dubai. The service enables patients to get tested for sexually transmitted diseases from home or a hotel, with sample collection by certified personnel and lab-backed diagnostic analysis. Such development indicates future trends; the UAE actively integrates advanced technologies such as artificial intelligence into result interpretation to enhance speed and precision.
Kuwait Syphilis Testing Market Trends
The syphilis testing market in Kuwait is growing due to the high burden of diseases in the country, expecting to promote the demand for diagnostic testing. Further, increasing demand for rapid tests, elevated screening in antenatal and migrant health programs, and deployment of molecular and immunoassay technologies. Also, STD awareness campaigns and private health infrastructure growth suggest that diagnostic providers in Kuwait are likely seeing moderate growth in both immunoassay-based syphilis tests and the early uptake of molecular confirmatory testing.
Oman Syphilis Testing Market Trends
The syphilis testing market in Oman is emerging. Oman has achieved a significant public health milestone. On October 19, 2022, the WHO certified Oman as the first country in the Eastern Mediterranean Region to eliminate mother-to-child transmission (MTCT) of HIV and syphilis. As seen in Oman’s EMTCT success, high antenatal screening coverage (often >95%) drives consistent demand for syphilis tests. Similarly, awareness campaigns and mandatory screening (for work visas, travel, etc.) in urban GCC areas push immunoassay and molecular diagnostics usage.
Qatar Syphilis Testing Market Trends
The syphilis testing market in Qatar is growing. In Qatar, syphilis testing trends indicate modest but steady demand, particularly among migrant populations and through workplace or entry-screening requirements. A survey among Male Craft and Manual Workers (MCMWs) found a lifetime syphilis infection prevalence of ~1.3% and recent infection in a much smaller fraction—suggesting asymptomatic carriage or latent cases are important. Diagnostic service providers in Qatar are expanding “discreet” and molecular panel testing options (e.g., tests that include Treponema pallidum alongside multiple other pathogens) for privacy, convenience, and early detection.
Key Middle East Syphilis Testing Company Insights
Market leaders are involved in extensive R&D to manufacture technologically advanced, cost-efficient testing products. Various organizations are incorporating several strategies, such as mergers and acquisitions, to expand their market presence, which is anticipated to create significant growth opportunities over the forecast period.
Key Middle East Syphilis Testing Companies:
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Danaher Corporation (Beckman Coulter)
- Thermo Fisher Scientific Inc.
- bioMérieux SA
- OraSure Technologies, Inc.
- Chembio Diagnostics, Inc.
- SD Biosensor, Inc.
Recent Developments
-
In March 2025, Genalive, a joint venture between BGI Genomics and Saudi partner Tibbiyah Holding, secured a three-year, ¥950 million RMB contract with the Saudi National Unified Procurement Company (NUPCO). This contract, the largest in NUPCO's history, provides outsourced testing services to 83 public hospitals across Saudi Arabia. The services include whole genome sequencing, exome sequencing, non-invasive prenatal testing, and hereditary cancer screening.
-
In January 2025, PrisesBio launched a syphilis antibody rapid test product for the UAE market, giving locals and tourists a quicker and easier way to detect syphilis early. The announcement highlights that diagnostic confidence is ensured by UAE clinicians' extensive training in interpreting these assays.
-
In October 2024, Kuwait’s Ministry of Health inaugurated the Tatman Clinic at the Fahaheel Health Center, marking the country's third voluntary testing and counseling facility. This clinic is part of a broader national effort to expand public health services focused on HIV and sexually transmitted diseases (STDs). The clinic offers pre-test counseling, education on HIV and STDs, and identification of risk factors. Patients can receive rapid HIV testing, and the clinic also supports testing for syphilis, hepatitis, chlamydia, and gonorrhoea.
Middle East Syphilis Testing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 23.08 million
Revenue forecast in 2033
USD 55.01 million
Growth rate
CAGR of 11.47% from 2025 to 2033
Actual data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, location of testing, region
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Abbott Laboratories; F. Hoffmann-La Roche Ltd.; Bio-Rad Laboratories, Inc.; Siemens Healthineers AG; Danaher Corporation (Beckman Coulter); Thermo Fisher Scientific Inc.; bioMérieux SA; OraSure Technologies, Inc.; Chembio Diagnostics, Inc.; SD Biosensor, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Syphilis Testing Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East syphilis testing market based on technology, location of testing, and region:
-
Technology Outlook (USD Million, 2021 - 2033)
-
Molecular Diagnostics
-
Immunoassay
-
Others
-
-
Location of Testing Outlook (USD Million, 2021 - 2033)
-
Laboratory Testing
-
Commercial/Private labs
-
Public Health Labs
-
-
Point of Care Testing
-
-
Regional Outlook (USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The Middle East syphilis testing market size was estimated at USD 21.25 million in 2024 and is expected to reach USD 23.08 million in 2025.
b. The Middle East syphilis testing market is expected to grow at a compound annual growth rate of 11.47% from 2025 to 2033 to reach USD 55.01 billion by 2033.
b. The immunoassay segment dominated the Middle East syphilis testing market and accounted for the largest revenue share of around 43.95% in 2024.
b. Abbott Laboratories, F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Siemens Healthineers AG, Danaher Corporation (Beckman Coulter), Thermo Fisher Scientific Inc., bioMérieux SA, OraSure Technologies, Inc., Chembio Diagnostics, Inc., SD Biosensor, Inc.
b. The market growth is driven by the growing public health awareness and government-led initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










