- Home
- »
- Clinical Diagnostics
- »
-
Multiplex Testing Reagents Market, Industry Report, 2033GVR Report cover
![Multiplex Testing Reagents Market Size, Share & Trend Report]()
Multiplex Testing Reagents Market (2025 - 2033) Size, Share & Trend Analysis By Type (Biotin-based Reagents, Streptavidin-based Reagents), By Application (Autoimmune Disease Diagnostics, Oncology), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-793-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Multiplex Testing Reagents Market Summary
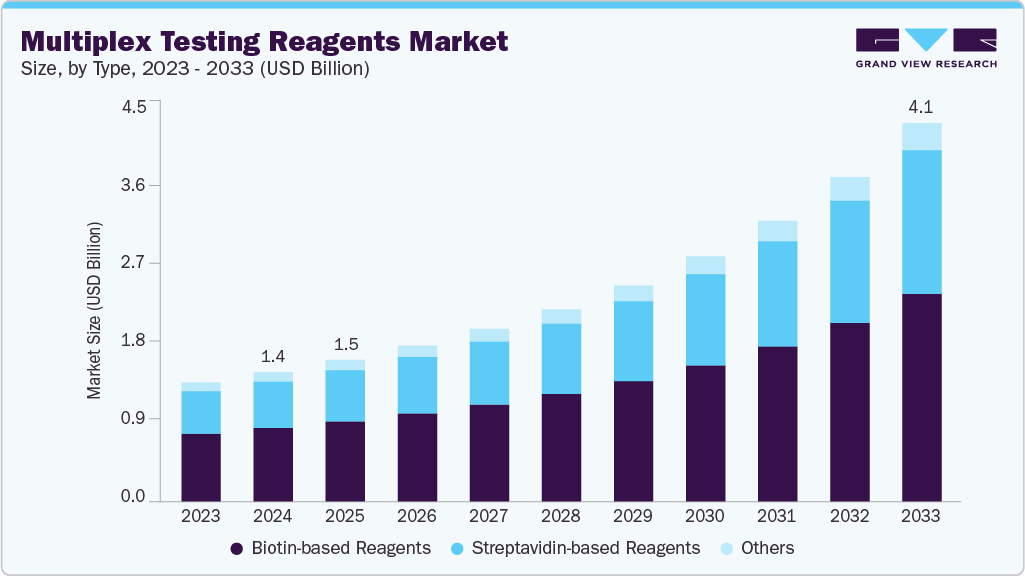
The global multiplex testing reagents market size was estimated at USD 1.42 billion in 2024 and is expected to reach USD 4.15 billion by 2033, growing at a CAGR of 13.08% from 2025 to 2033. Multiplex testing reagents enable the simultaneous detection of multiple analytes in a single test, offering higher efficiency and cost-effectiveness compared to conventional single-target assays.
Key Market Trends & Insights
- The North America multiplex testing reagents market dominated with the largest revenue share of 32.90% in 2024.
- The U.S. led the North American market and held the largest revenue share in 2024.
- Based on type, the biotin-based reagents segment dominated the global market in 2024.
- Based on application, the drug discovery & development segment held the largest revenue share of 26.61% in 2024.
- On the basis of end use, the pharmaceutical & biotechnology companies segment held the largest revenue share of 38.71% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.42 Billion
- 2033 Projected Market Size: USD 4.15 Billion
- CAGR (2025-2033): 13.08%
- North America: Largest Market in 2024
Their ability to provide comprehensive diagnostic data in shorter timeframes has driven adoption across clinical, research, and pharmaceutical applications. The market’s growth is further fueled by the increasing demand for personalized medicine, where rapid and precise detection of multiple biomarkers is critical for tailoring treatment strategies. Technological advancements in assay design, automation, and detection platforms have also enhanced the reliability and throughput of multiplex testing, making it an attractive choice for laboratories aiming to optimize workflow while minimizing sample volume and operational costs.A primary factor driving the growth of multiplex testing reagents is the rising prevalence of chronic diseases and infectious conditions worldwide. Chronic illnesses such as cardiovascular disorders, cancer, autoimmune diseases, and metabolic syndromes often involve multiple biomarkers, making multiplex assays an efficient tool for comprehensive monitoring and diagnosis. In addition, the global burden of infectious diseases, including viral, bacterial, and emerging pathogens, has increased the need for high-throughput diagnostic solutions capable of detecting multiple pathogens simultaneously. Multiplex reagents allow clinicians to screen for co-infections and identify pathogen profiles in a single test, reducing diagnostic delays. This is particularly important in public health scenarios where early detection can prevent outbreaks. Consequently, growing disease prevalence directly correlates with increased adoption of multiplex testing reagents, thereby driving market expansion.
Technological innovation is a key driver of market growth for multiplex testing reagents. Advanced assay platforms, such as bead-based flow cytometry, microarrays, and next-generation sequencing-compatible reagents, have enhanced the sensitivity, specificity, and throughput of multiplex testing. Automation and integration with laboratory information systems have further streamlined workflows, allowing laboratories to process larger sample volumes with reduced human error. Novel reagent chemistries and labeling techniques have improved the detection of low-abundance biomarkers, expanding clinical and research applications. In addition, miniaturization and microfluidic technologies have enabled point-of-care multiplex testing, bringing laboratory-grade analysis closer to patients. These technological improvements not only improve diagnostic accuracy but also reduce turnaround time and costs, making multiplex testing reagents increasingly attractive for hospitals, diagnostic laboratories, and research institutions globally.
The trend toward personalized and precision medicine is fueling demand for multiplex testing reagents. Personalized medicine relies on comprehensive biomarker profiling to tailor therapies to individual patients, optimizing treatment outcomes while minimizing adverse effects. Multiplex testing allows simultaneous analysis of genetic, proteomic, and immunologic markers, providing a holistic view of disease states. This is particularly critical in oncology, where tumor heterogeneity requires multiple biomarker assessments to guide targeted therapies. Similarly, in autoimmune and metabolic disorders, multiplex reagents facilitate accurate disease classification and monitoring of treatment response. As healthcare providers increasingly focus on individualized care, the need for multiplex testing reagents rises. The ability to obtain multiple diagnostic results from a single patient sample aligns with efficiency goals, patient comfort, and cost reduction, further strengthening the market’s growth trajectory.
Expanding research activities and clinical applications represent another growth factor for multiplex testing reagents. Pharmaceutical and biotechnology companies are increasingly relying on multiplex assays for drug discovery, clinical trials, and biomarker validation studies. Academic and government research institutions utilize these reagents to investigate complex disease mechanisms, study immune responses, and perform large-scale epidemiological studies. Multiplex testing’s capability to analyze multiple parameters simultaneously accelerates research timelines and enhances data quality. Moreover, the adoption of multiplex reagents in routine clinical diagnostics, such as infectious disease screening, allergy panels, and autoimmune disorder monitoring, has broadened market opportunities. Government initiatives promoting advanced diagnostics and funding for translational research further support market growth. Consequently, the combination of research and clinical applications continues to drive sustained demand for multiplex testing reagents worldwide.
Market Concentration & Characteristics
The multiplex testing reagents industry is experiencing significant innovation, driven by advancements in molecular diagnostics and personalized medicine. Companies are developing high-throughput platforms that enable the simultaneous detection of multiple biomarkers, enhancing diagnostic efficiency and accuracy. This innovation is crucial for applications in oncology, infectious diseases, and autoimmune disorders.
The market has seen robust mergers and acquisitions, as companies seek to expand their technological capabilities and market reach. Strategic acquisitions of biotech firms and assay technology developers are helping larger diagnostics companies strengthen their assay portfolios and access new markets. These consolidations accelerate innovation cycles and enable faster deployment of advanced testing solutions.

Regulatory frameworks significantly influence the global landscape. In the U.S., the FDA oversees in vitro diagnostic (IVD) products, ensuring safety and efficacy. Proposed changes to regulations, such as stricter premarket review requirements, aim to enhance public health protection but may impact the development and availability of new diagnostic tests.
The market for multiplex testing reagents is expanding with the development of new products that offer enhanced sensitivity and specificity. Innovations include multiplex PCR kits and lateral flow assays that can detect multiple pathogens or biomarkers simultaneously. These advancements are particularly beneficial in point-of-care diagnostics and personalized medicine, where rapid and accurate results are essential.
The multiplex testing reagents market is witnessing regional expansion, particularly in the Asia Pacific region. Countries like China are increasingly sourcing laboratory reagents from domestic suppliers to reduce costs and shorten delivery times, responding to rising import tariffs and concerns about supply chain reliability. This shift is supported by robust biotech sector growth and government initiatives to enhance local manufacturing capabilities.
Type Insights
On the basis of type, biotin-based reagents accounted for the largest market share of 56.85% in 2024. The dominance of this segment is primarily attributed to biotin’s high binding affinity with streptavidin, which ensures superior assay sensitivity, specificity, and reproducibility. Biotin-based reagents are widely used in multiplex immunoassays, PCR, and molecular diagnostic platforms due to their ability to enable efficient signal amplification and multi-target detection in a single reaction. Moreover, the increasing demand for accurate and high-throughput diagnostic tools in infectious disease testing, oncology, and genetic screening has accelerated the adoption of these reagents. Major market players such as Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN, and Merck KGaA continue to innovate biotin-based chemistries to improve assay performance and automation compatibility, further reinforcing this segment’s leadership and growth potential in multiplex diagnostic applications.
The streptavidin-based reagents segment is anticipated to experience the fastest growth during the forecast period in the multiplex testing reagents industry. This growth is driven by the increasing adoption of streptavidin-biotin chemistry in multiplex assays due to its exceptional binding strength, low background interference, and compatibility with diverse detection platforms. Streptavidin-based reagents enable high signal amplification, making them ideal for applications in immunoassays, molecular diagnostics, and proteomics where precision and sensitivity are critical. Moreover, the growing use of multiplex assays in infectious disease diagnosis, cancer biomarker profiling, and genetic testing is further fueling demand for these reagents. Key manufacturers such as Thermo Fisher Scientific, Merck KGaA, Bio-Rad Laboratories, and PerkinElmer Inc. are investing in developing next-generation streptavidin conjugates with enhanced stability and reduced non-specific binding, which is expected to accelerate adoption and solidify this segment’s position as a key growth driver in the multiplex testing market.
Application Insights
Based on application, the drug discovery & development segment accounted for the largest market share of 26.61% in 2024. This dominance is attributed to the increasing utilization of multiplex assays in pharmaceutical research to accelerate target identification, biomarker validation, and high-throughput screening. Multiplex testing enables simultaneous analysis of multiple analytes, reducing reagent consumption, cost, and time while improving data reliability-key advantages in modern drug development workflows. In addition, the growing emphasis on personalized medicine and precision therapeutics has boosted demand for multiplex-based platforms that support comprehensive pharmacological profiling. As R&D investments continue to rise globally, the Drug Discovery & Development segment is expected to sustain its strong market position throughout the forecast period.
The others segment is anticipated to experience the fastest growth during the forecast period in the market for multiplex testing reagents. This segment includes emerging applications such as neurological disorder research, cardiovascular disease diagnostics, environmental testing, and veterinary diagnostics. The rapid expansion of these areas is driven by growing awareness of early disease detection, increased funding for translational research, and advancements in high-throughput multiplex platforms. Furthermore, the integration of multiplex assays in epigenetic and metabolic studies is broadening their utility beyond conventional diagnostics. Leading manufacturers, including Thermo Fisher Scientific, Bio-Rad Laboratories, Merck KGaA, and PerkinElmer Inc., are actively developing specialized multiplex reagent kits to address these new applications, positioning the “Others” segment as a key driver of future market expansion.
End Use Insights
Based on end use, Pharmaceutical & Biotechnology Companies accounted for the largest market share of 38.71% in 2024. This dominance is primarily attributed to the extensive use of multiplex assays in drug discovery, biomarker validation, and clinical research. These companies leverage multiplex reagent technologies to analyze multiple biomarkers simultaneously, accelerating R&D timelines and improving data accuracy in preclinical and clinical studies. The growing demand for personalized medicine and precision diagnostics has further fueled the adoption of multiplex testing platforms within the biopharmaceutical sector. Additionally, the rising prevalence of infectious and chronic diseases has increased investment in multiplex-based molecular and immunoassay technologies.
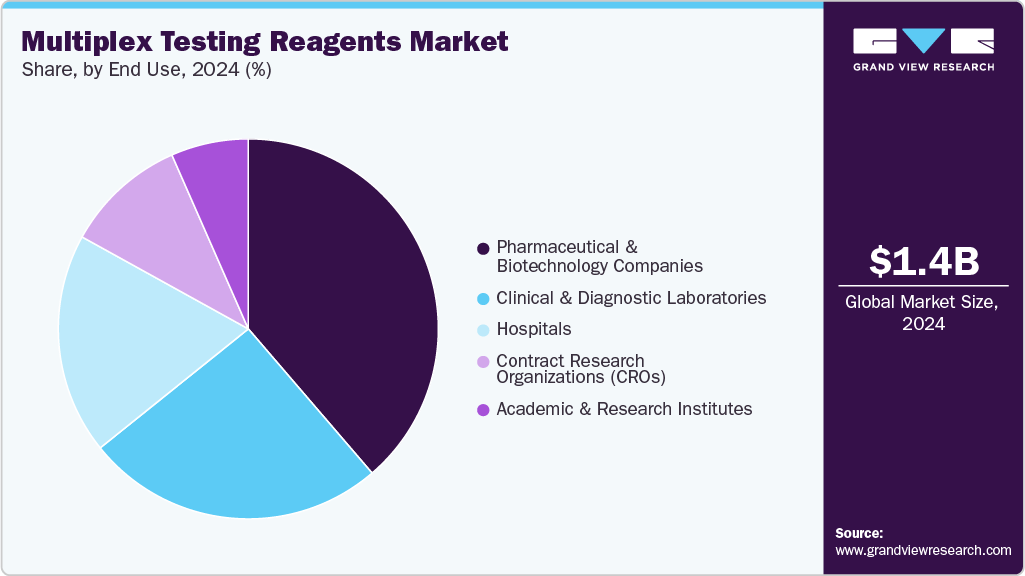
The clinical & diagnostic laboratories segment is anticipated to experience the fastest growth during the forecast period. This growth is primarily driven by the increasing adoption of multiplex assays for infectious disease detection, oncology diagnostics, allergy testing, and the screening of autoimmune disorder. Clinical laboratories are rapidly transitioning from conventional single-analyte testing to multiplex technologies due to their ability to deliver faster, more accurate, and cost-efficient results. Moreover, the rising global burden of chronic and infectious diseases, coupled with the growing demand for personalized and precision diagnostics, is accelerating the use of multiplex reagent platforms. Advancements in automation, digital PCR, and high-throughput immunoassay systems are also enhancing test efficiency and scalability.
Regional Insights
The North America multiplex testing reagents industry accounted for the largest revenue share of 32.90% in 2024, driven by growing R&D activities, including the development of novel drugs, diagnostics, and treatment options. The rising prevalence of chronic conditions such as stroke and cancer, along with increased government funding for new biomarker detection, is expected to fuel segment growth. Leading regional players are also likely to strengthen market expansion.

U.S. Multiplex Testing Reagents Market Trends
The multiplex testing reagents industry in the U.S. is experiencing strong growth due to advanced healthcare infrastructure, significant R&D investments, and early adoption of innovative diagnostics. The increasing burden of chronic and infectious diseases, coupled with an emphasis on precision medicine, drives demand. Supportive FDA regulatory pathways and the presence of key market players further promote rapid innovation and commercialization. Technological advancements in genomics and proteomics, along with the need for efficient diagnostic solutions, position the U.S. as a global leader.
Europe Multiplex Testing Reagents Market Trends
The Europe multiplex testing reagents industry is expanding steadily, supported by strong academic research, favorable healthcare policies, and a rising chronic disease burden. EU initiatives promoting early disease diagnosis and personalized medicine accelerate adoption. Countries such as France, Italy, and the Netherlands are witnessing an increase in investment in diagnostic laboratories. Collaborations between pharmaceutical and biotech firms for drug development further boost clinical trial applications. Regulatory frameworks, while strict, ensure high-quality standards and continued innovation.
The multiplex testing reagents industry in the UK benefits from strong government support for genomics research and NHS initiatives promoting early detection. Projects like Genomics England and public-private partnerships enhance the use of multiplex testing in personalized healthcare. A focus on cancer diagnostics and infectious disease screening is expanding demand, while the UK’s vibrant biotech ecosystem and streamlined regulatory environment foster innovation and commercialization.
The Germany multiplex testing reagents industry is driven by its advanced medical technology sector, extensive healthcare infrastructure, and robust clinical research landscape. Emphasis on precision diagnostics and laboratory automation encourages adoption of multiplex testing. Government funding for cancer and infectious disease research, combined with leading biotech and pharmaceutical companies, supports market growth. Germany’s skilled workforce and efficient regulatory processes further promote the development and distribution of advanced diagnostic reagents.
Asia Pacific Multiplex Testing Reagents Market Trends
The Asia Pacific multiplex testing reagents industry is expected to witness the fastest growth during the forecast period, driven by the increasing number of hospitals, expanding R&D sectors, growing healthcare infrastructure, and regional investments. For example, QuantuMDx received USD 10.9 million from Vita Spring and partnered with Sansure Biotech to develop multiplex capabilities in China. Rising cancer prevalence due to tobacco use also increases the demand for screenings, supporting market growth.
The multiplex testing reagents industry in China is rapidly expanding due to government investment in healthcare modernization and biotechnology. The growing prevalence of infectious and chronic diseases drives demand for rapid, accurate diagnostics. Initiatives like “Made in China 2025” promote biotech innovation, with local companies developing multiplex technologies. Collaborations between Chinese hospitals and global firms accelerate clinical application, while population size and regulatory reforms aid market penetration.
The Japan multiplex testing reagents industry is growing steadily due to an aging population, high healthcare standards, and a focus on early disease detection. Investment in genomic research and personalized medicine makes multiplex reagents vital diagnostic tools. Government initiatives and academic collaborations drive R&D, supported by a robust domestic pharmaceutical industry and a regulatory environment that encourages innovation while ensuring high-quality standards.
Latin America Multiplex Testing Reagents Market Trends
The Latin America multiplex testing reagents industry is experiencing gradual growth, supported by rising healthcare investments and growing awareness of advanced diagnostics. Brazil, Mexico, and Argentina lead the market, driven by improved laboratory infrastructure and demand for infectious disease testing. Regulatory challenges and limited reimbursement structures pose obstacles, but partnerships with global diagnostics companies and public health programs are expanding access. Government emphasis on disease prevention and early diagnosis highlights the market’s potential.
Middle East and Africa Multiplex Testing Reagents Market Trends
The multiplex testing reagents industry in the Middle East and Africa shows emerging growth, fueled by rising healthcare spending and the burden of infectious diseases. Countries like the UAE and Saudi Arabia are investing in diagnostic technologies as part of healthcare modernization. In Africa, international aid and NGO collaborations facilitate multiplex testing adoption, particularly for HIV and tuberculosis. Limited infrastructure and high costs may restrict growth, though ongoing investment is expected to improve market expansion.
Key Multiplex Testing Reagents Company Insights
The multiplex testing reagents industry is driven by several leading companies at the forefront of innovation and adoption. Prominent players include Thermo Fisher Scientific Inc., Bio-Rad Laboratories, Inc., Luminex Corporation, QIAGEN N.V., Seegene Inc., Abcam plc., PerkinElmer Inc., Merck KGaA, Illumina, Inc., and Bio-Techne Corporation. These companies are investing heavily in research and development, expanding geographically, introducing continuous product innovations, and forming strategic collaborations, which strengthen their market positions and drive growth in diagnostics and personalized medicine.
Key Multiplex Testing Reagents Companies:
The following are the leading companies in the multiplex testing reagents market. These companies collectively hold the largest market share and dictate industry trends.
- Luminex Corporation
- Bio-Rad Laboratories, Inc.
- Abcam plc.
- Seegene Inc.
- Merck KGaA
- Illumina, Inc.
- Promega Connections
- QIAGEN N.V.
- Thermo Fisher Scientific
- PerkinElmer Inc.
- Bio-Techne Corporation
- BD
Recent Developments
-
In October 2025, Seegene unveiled two multiplex diagnostic platforms, CURECA and STAgora, at the ADLM 2025 conference. These platforms are designed to enhance the detection of complex diseases through advanced multiplex assays.
-
In October 2025, Thermo Fisher Scientific launched a high-throughput multiplex platform capable of analyzing over 100 biomarkers simultaneously. This advancement aims to accelerate diagnostics in oncology, autoimmune disorders, and infectious diseases.
-
In June 2024, Columbia Biosciences announced a partnership with AlphaThera to develop a range of site-specific fluorescent protein-labeling reagents (workflows that commonly use biotin/streptavidin chemistry for capture or detection). This expands available biotinylation and labeling reagents for multiplex workflows.
-
In May 2025, ZEISS announced a collaboration leveraging Kromnigon’s StreptaClick® HRP technology for tissue multiplexing (8-channel multiplex immunofluorescence), demonstrating a streptavidin-based HRP approach optimized for spatial biology workflows
-
In July 2025, Bio-Rad Laboratories introduced four new Droplet Digital PCR (ddPCR) platforms, including the QX Continuum ddPCR system and the QX700 series, acquired through their purchase of Stilla Technologies.
Multiplex Testing Reagents Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.55 Billion
Revenue forecast in 2033
USD 4.15 Billion
Growth Rate
CAGR of 13.08% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Luminex Corporation; Bio-Rad Laboratories, Inc.; Abcam plc.; Seegene Inc.; Merck KGaA; Illumina, Inc.; Promega Connections; QIAGEN N.V.; Thermo Fisher Scientific; PerkinElmer Inc.; Bio-Techne Corporation; BD
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Multiplex Testing Reagents Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global multiplex testing reagents market report based on type, application, end use, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Biotin-based Reagents
-
Biotinylated Antibodies
-
Biotinylated Probes/primers
-
Biotin-tagged Proteins/enzymes
-
-
Streptavidin-based Reagents
-
Streptavidin-conjugated Beads/microspheres
-
Streptavidin-conjugated Enzymes & Fluorophores
-
Recombinant Streptavidin Variants (engineered for higher stability)
-
Others
-
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune Disease Diagnostics
-
Infectious Disease Diagnostics
-
Oncology
-
Allergy Testing
-
Research & Translational Medicine
-
Drug Discovery & Development
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical & Diagnostic Laboratories
-
Hospitals
-
Pharmaceutical & Biotechnology Companies
-
Contract Research Organizations (CROs)
-
Academic & Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










