- Home
- »
- Pharmaceuticals
- »
-
Myasthenia Gravis Treatment Market Size Report, 2030GVR Report cover
![Myasthenia Gravis Treatment Market Size, Share & Trends Report]()
Myasthenia Gravis Treatment Market Size, Share & Trends Analysis Report By Treatment Type (Thymectomy, Cholinesterase Inhibitors, Rapid Immunotherapies), By End-use (Hospitals, Clinics), By Region and Segment Forecasts, 2023 - 2030
- Report ID: GVR-4-68038-002-6
- Number of Report Pages: 120
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Market Size & Trends
The global myasthenia gravis treatment market size was valued at USD 2.01 billion in 2022 and is anticipated to grow at a compound annual growth rate (CAGR) of 9.0% from 2023 to 2030. The key factors driving this market include the rising adoption of immunotherapies, the approval of promising drugs, the emergence of biologics, and increasing awareness of rare diseases. Myasthenia gravis (MG) is a chronic, progressive, and rare neuromuscular disorder that is characterized by weakness of the head, limb, spinal, eye, and respiratory muscles. In this autoimmune disorder, antibodies are produced against the acetylcholine receptor (AchR) that impedes the normal transmission of electrical signals from nerves to muscles.
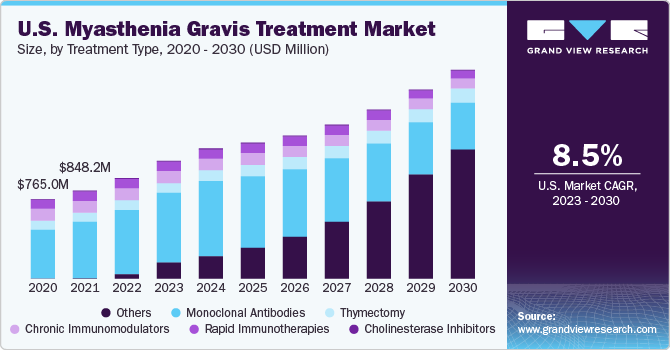
According to the National Organization for Rare Disorders (NORD), in the U.S., around 14-40 per 100,000 individuals suffer from myasthenia gravis. Reports suggest that the frequency of the disorder is on the rise over the last several decades. This can be attributed to the better identification of patients and the increasing prevalence of autoimmune disorders worldwide.
Myasthenia gravis occurs more frequently in females than males, and although symptoms may appear at any age, its onset usually peaks in men in their 50s or 60s and women during their 20s or 30s. There is no proven cure for myasthenia gravis; however, patients can be treated with medications and surgery to manage the symptoms.
Currently, research is focused on better understanding the specifics of the autoimmune problems associated with MG and improving their diagnosis and treatment. Additionally, the market is observing a paradigm shift in research from suppressing the immune system to rebalancing it. Another important area of research is the physiology of the neuromuscular junction (NMJ), aimed at improvement in its function regardless of probable immune system attacks.
Treatment Type Insights
Medications can dramatically inhibit the immune system and largely manage the associated symptoms. Medications include immunotherapies such as Soliris (eculizumab), corticosteroids, and immunosuppressive drugs such as Prograf (tacrolimus) and Neoral/Sandimmune (cyclosporine). The other major treatment alternative, surgery, is the most invasive and usually involves a thymectomy (thymus removal). The thymus deteriorates with age (thymic involution); however, complete loss of the thymus in such a manner will significantly weaken a patient’s immune system.
The monoclonal antibodies segment held the largest market share of over 60% in 2022 and is expected to maintain its leading position through the forecast period, owing to the high precision in treating myasthenia gravis and better efficacy. For instance, Alexion Pharmaceuticals’ eculizumab monoclonal antibodies drug Soliris is used for the treatment of myasthenia gravis. Recently, it received approval from the U.S. Food and Drug Administration (FDA) and the European Commission for treating adult patients with generalized myasthenia gravis who are anti-AchR antibody-positive. Soliris is the first and only complement inhibitor approved for this disorder. The increasing use of monoclonal antibodies and immunosuppressants is aimed at reducing lifelong exposure to corticosteroids and improving long-term results.
The market is witnessing an upsurge in the number of pipeline studies for drug development that is expected to drive the market, resulting in an anticipated fastest growth of 41.8% CAGR in the pipeline studies segment over the forecast period.
End-use Insights
Based on end use, the market has been segmented into hospitals, clinics, and others. The hospital segment held the largest market share of around 55% in 2022 and is expected to grow at a lucrative CAGR of 9.0% over the forecast period. This can be attributed to access to a large patient pool and the high demand for advanced equipment for MG treatment in hospital settings. There has been an increase in patient visits for diagnosis and treatment of MG due to the availability of a broad range of treatment options in such facilities. Furthermore, one of the major factors contributing to its large share is the increased investment in state-of-the-art healthcare infrastructures.
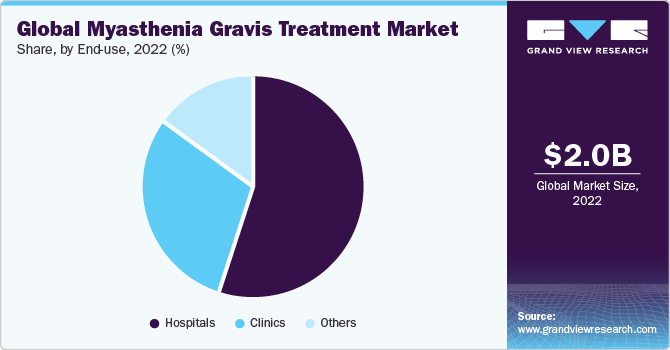
The clinic segment held the second-largest share of around 30% in 2022 and is expected to retain the position through the forecast period. With the growing need for specialized treatments, many patients prefer specialty clinics for rapid diagnosis and effective MG treatment. A large segment of the end users also depend on eCommerce channels for obtaining therapeutics for myasthenia gravis. These segments are significantly contributing to the growth of the myasthenia gravis treatment market.
Regional Insights
In 2022, North America had the largest market share of over 50% and is projected to lead the market throughout the forecast period. This can be attributed to the high adoption of immunotherapies and monoclonal antibodies, increasing healthcare expenditure, the presence of established research and development facilities, and favorable reimbursement scenarios.
Middle East and Africa is expected to grow at the fastest CAGR of 14.9% over the forecast period. This can be attributed to the rising prevalence of myasthenia gravis in the region, especially among the elderly population, increasing awareness, availability of advanced diagnostics tools and therapeutic options, and the expansion of healthcare infrastructure.
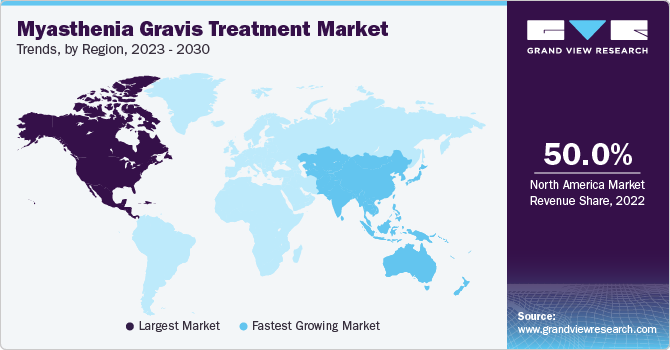
Asia Pacific is anticipated to grow at a lucrative CAGR through the forecast period due to the increase in revenue generation from medications such as immunosuppressants and monoclonal antibodies in this region. Other factors propelling the growth are the initiatives associated with increasing investments in the healthcare sector by the regional governments and manufacturers.
Key Companies & Market Share Insights
Some of the key companies operating in the market such as Alexion Pharmaceuticals and Astellas Pharma hold the largest market shares due to the strong sales of their flagship products, including Soliris and Prograf.
However, generic competition is a major hindrance to market growth due to the patent loss of several marketed products and the availability of low-priced substitutes to branded drugs in the market. To gain an advantage against competitors, companies are adopting strategies such as expansion in emerging countries, new product development, spreading awareness, and getting into collaborations. For instance, in April 2022, AstraZeneca received approval for Ultomiris (ravulizumab-cwvz) from the FDA for generalized myasthenia gravis. In December 2021, the FDA approved efgartigimod drug Vyvgart, a novel treatment for AChR positive tested generalized myasthenia gravis patients.
Key Myasthenia Gravis Treatment Companies:
- Alexion Pharmaceutical Inc.
- Grifols SA
- Avadel Pharmaceuticals, Plc.
- Novartis
- Pfizer, Inc.
- AbbVie Inc.
- F.Hoffmann-La Roche AG
- GlaxoSmithKline Plc.
- Bausch Health Companies Inc.
- Shire plc
Myasthenia Gravis Treatment Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 2.33 billion
Revenue forecast in 2030
USD 4.26 billion
Growth Rate
CAGR of 9.0% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Treatment type, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Alexion Pharmaceutical Inc.; Grifols SA; Avadel Pharmaceuticals plc. Novartis AG; Pfizer; Inc.; AbbVie Inc.; F. Hoffmann-La Roche Ltd.; GlaxoSmithKline plc; Bausch Health Companies Inc.; Shire plc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Myasthenia Gravis Treatment Market Report Segmentation
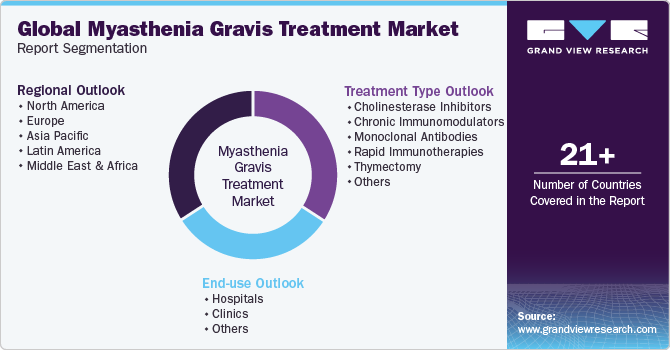
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global myasthenia gravis treatment market based on treatment type, end-use, and region:
-
Treatment Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Cholinesterase Inhibitors
-
Chronic Immunomodulators
-
Monoclonal Antibodies
-
Rapid Immunotherapies
-
Thymectomy
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals
-
Clinics
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global myasthenia gravis treatment market size was estimated at USD 2.01 billion in 2022 and is expected to reach USD 2.33 billion in 2023.
b. The global myasthenia gravis treatment market is expected to grow at a compound annual growth rate of 9.0% from 2023 to 2030 to reach USD 4.26 billion by 2030.
b. North America dominated the myasthenia gravis treatment market with a share of 51.1% in 2022. This is attributable to the high adoption of novel therapies, increasing research and development investments, and favorable reimbursement scenarios.
b. Some key players operating in the myasthenia gravis treatment market include Alexion Pharmaceutical Inc., Bausch Health Companies Inc., Astellas Pharma Inc., Mitsubishi Tanabe Pharma Co., Grifols SA, Catalyst Pharmaceuticals Inc., CSL Behring, Argenx, UCB S.A., Ra Pharmaceuticals, Inc., and Novartis AG
b. Key factors that are driving the market growth include rising incidence of autoimmune disorders, increasing research and development activities, and growing awareness about Myasthenia Gravis
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





