- Home
- »
- Medical Devices
- »
-
Needle-free Injectors Market Size, Share & Trends Report, 2030GVR Report cover
![Needle-free Injectors Market Size, Share & Trends Report]()
Needle-free Injectors Market (2023 - 2030) Size, Share & Trends Analysis Report By Product (Fillable, Prefilled), By Technology, By Type (Liquid-based Needle-free Injectors), By Usability, By Application (Vaccine Delivery), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-994-7
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2018 - 2020
- Forecast Period: 2023 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
The global needle free-injectors market size was estimated at USD 1.3 billion in 2022 and expected to expand at a compound annual growth rate (CAGR) of 7.8% from 2023 to 2030. Increasing number of regulatory approvals, rising preference for self-administration of drugs are expected to foster market growth. Needle-free devices provide pain less delivery and negate the risk of infection. Hence, the demand for these injectors is projected to rise during the forecast period.
The COVID-19 pandemic has positive impact on needle-free injectors market. The requirement and demand for mass vaccination needs and self-injectable devices have increased since the initiation of COVID-19. As COVID-19 cases have increased in all regions, the majority of the needle free-injectors market players have participated in R&D activities and started entering into agreements and partnerships with other industry firms and government organizations to develop and improve needle free-injectors.
Needle-free injectors provide higher safety for patients and healthcare workers by eliminating risks related to conventional infusion and injection technology such as drug preparation, needle insertion, and covering & disposing of needles. In addition, American Nurses Association stated that around 74.0% of accidental needlestick injuries are due to contamination and are likely to lead to infection. Treatments for these injuries is expensive. According to the CDC, treatment cost ranges from USD 500 to USD 3,000 or more per injury. These costs get multiplied during infection. The CDC has also projected that the number of needle stick injuries reported each year in hospitals in the U.S. is 385,000. On the other hand, European Agency of Safety and Health at Work stated that about 1.0 million needlestick injuries occur every year across the world.
Rising healthcare costs is the biggest concern faced by developing and developed economies. These regions are struggling to address cost-effective and quality care. Despite supportive health funding and insurance program, there has been a constant rise in healthcare cost in the U.S., which is influencing healthcare providers. Healthcare expenditure in the U.S. is around 2.4 times higher than the global average and is anticipated to increase by 68.0% in the coming ten years. With the help of needle-free injectors, patients can self-administer the drug subcutaneously at home, this is anticipated to reduce the number of visits to hospitals or other healthcare settings, thereby helping patients to limit their healthcare expenses.
Product Insights
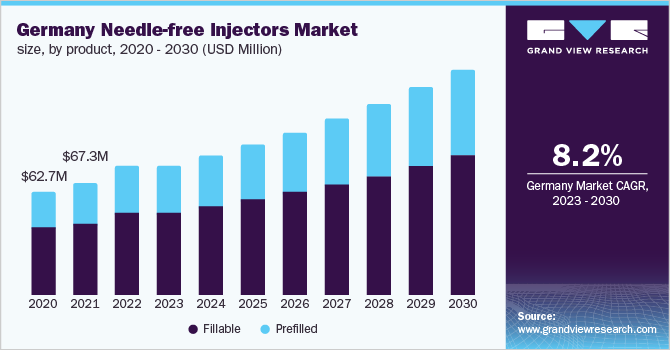
The fillable segment held the largest revenue share in 2022 owing to their high adoption rates and wide availability throughout the globe. The prefilled segment held a considerable market share in 2022. Increasing application of prefilled needle-free injectors in insulin, vaccination, and other drug delivery is projected to augment the market growth. Surging preference for prefilled needle-free injectors by several healthcare professionals may strengthen the market growth.
Usability Insights
The reusable segment dominated the needle-free injectors market and accounted for a significant revenue share in 2021. Reusable needle free-injectors are mostly suggested by healthcare specialists as well as doctors to reduce the risk of infections and other harmful ailments. Reusable injectors provide excessive durability as they are manufactured from superior and precision-engineered materials, which also help doctors to reuse them several times.
The disposable segment accounted for the largest revenue share in 2022. Disposable needle free-injectors are easy to use for numerous, low-volume injections or variable dosing. Increasing awareness regarding benefits provided by drug delivery devices and surging concerns over the hazards related to needle stick injuries may enhance the segment growth.
Technology Insights
The gas powered needle-free injectors segment held the largest revenue share in 2022. Gas powered needle-free injectors are suitable for transdermal, subcutaneous, or intramuscular use. Increasing benefits associated with this technology, including improved dosage accuracy, rapid drug administration, faster response rate, and better diffusion into the tissue are predicted to augment the market demand.
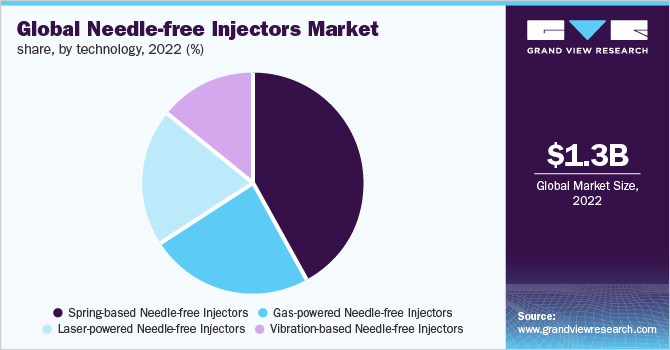
Laser-powered needle-free injectors segment held a considerable market share in 2021. Recent advancements and innovations in laser powered needle-free injectors are anticipated to propel market growth.
Type Insights
The segmental growth is attributed to the increasing ability of a liquid jet to penetrate the skin and underlying fat layer without harming the integrity of the drug molecule or harming the skin. Hence surging demand for liquid-based needle-free injectors is anticipated to foster market growth. The powder-based needle free injectors segment held a significant CAGR during the forecast period. Several market players across the globe are focusing on developing powder-based needle-free injectors to strengthen their product portfolio.
Delivery Site Insights
The subcutaneous segment held the largest market share in 2022. The segmental growth is attributed to the greater effect as the drug moves to the capillaries and is carried to the bloodstream. The intradermal segment accounted for a significant CAGR during the progression period. Increasing preference for intradermal administration by several patients across the globe is anticipated to fuel market growth.
Application Insights
Vaccine delivery is among the leading application segment in needle-free injectors market. Increasing preference for vaccine administration is predicted to surge the demand for needle free-injectors during the forecast period. The pain management segment is expected to witness a significant CAGR during the forecast period. Increasing cases of medical conditions such as pain disorders, arthritis, and migraine is predicted to secure the market position.
End-use Insights
The hospitals segment captured the largest revenue share of in 2022. Hospitals require needle-free injectors in large quantities at nurse’s stations, operation theaters, emergency rooms, outpatient departments, and ambulatory services. Hospitals generally issue an open tender for the procurement of such devices. Growing hospitalization of patients coupled with a surge in the number of hospitals across the world is expected to contribute to market growth during the forecast period.
The homecare settings segment gained momentous growth in 2022. Advanced features of needle-free injectors are improving patient adherence toward drug delivery devices, and this is expected to aid in the growth of the home care segment.
Regional Insights
North America is anticipated to dominate the market at 42.5% in 2022 and is expected to grow over the forecast period owing to the presence of major market players in the U.S. coupled with the increasing prevalence of chronic and lifestyle-associated diseases. In addition, the growing trend was found throughout the U.S. and among Canadian citizens towards self-administration of medications. Hence the above-mentioned factors are anticipated to boost regional growth.
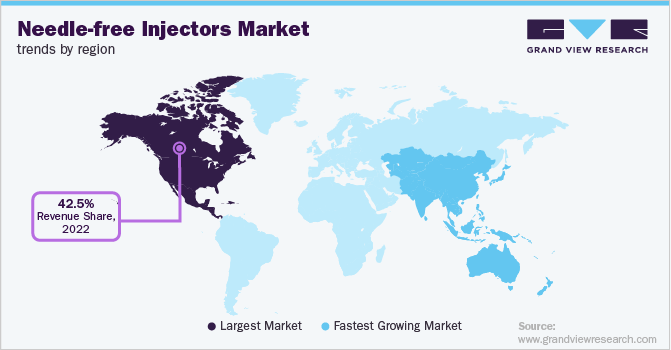
Europe accounted for the second-highest revenue share and is expected to witness a substantial CAGR during the forecast period. Advancements in medical technologies, rising adoption of needle-free injectors, and increasing healthcare spending are expected to contribute to the market growth during the forecast period.
Asia Pacific is expected to register the fastest CAGR around 8.16% over the forecast period, which can be attributed to favorable government initiatives for use of medical devices, increasing geriatric population base, and increasing healthcare expenditure in this region. Increase in aging population in several Asian countries, which is highly prone to chronic diseases, is expected to fuel the demand for needle-free injectors.
Key Companies & Market Share Insights
Major companies functioning in needle free-injectors market have well-known vigorous business strategies such as strategic partnerships and collaborations, joint ventures, mergers & acquisitions, new product launches & product innovation, to expand their market reach. Some of the prominent players in the needle free-injectors market include:
-
PharmaJet
-
Crossject
-
Portal Instruments
-
NuGen Medical Devices
-
Inovio Pharmaceuticals
-
Antares Pharma, Inc
-
Aijex Pharma International Inc.
-
Ferring B.V.
-
Penjet Corporation
-
Medical International Technology Inc.
Needle-free Injectors Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 1.4 billion
Revenue forecast in 2030
USD 2.4 billion
Growth rate
CAGR of 7.8% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD Million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company share, competitive landscape, growth factors & trends
Segments covered
Product, technology, usability, type, application, delivery site, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; U.K.; Germany; France; Italy; Spain; Japan; China; India; South Korea; Australia; Brazil; Mexico; Argentina; Columbia; South Africa; Saudi Arabia; UAE
Key companies profiled
PharmaJet; Crossject; Portal Instruments; NuGen Medical Devices; Inovio Pharmaceuticals; Antares Pharma, Inc; Aijex Pharma International Inc.; Ferring B.V.; Penjet Corporation; Medical International Technology Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional, and segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Needle-free Injectors Market Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global needle-free injectors market report based on product, technology, usability, type, application, delivery site, end-use, and region:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Fillable
-
Prefilled
-
-
Technology Outlook (Revenue, USD Million, 2018 - 2030)
-
Spring-based Needle-free Injectors
-
Gas-powered Needle-free Injectors
-
Laser-powered Needle-free Injectors
-
Vibration-based Needle-free Injectors
-
-
Usability Outlook (Revenue, USD Million, 2018 - 2030)
-
Disposable
-
Reusable
-
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Powder-based Needle Free Injectors
-
Liquid-based Needle Free Injectors
-
Projectile/depot-based Needle Free Injectors
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Vaccine Delivery
-
Insulin Delivery
-
Pain Management
-
Others
-
-
Delivery Site Outlook (Revenue, USD Million, 2018 - 2030)
-
Subcutaneous
-
Intramuscular
-
Intradermal
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals & Clinics
-
Home Care Settings
-
Research Laboratories
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
Germany
-
U.K.
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
Colombia
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
-
Frequently Asked Questions About This Report
b. The global needle-free injectors market size was estimated at USD 1.3 billion in 2022 and is expected to reach USD 1.4 billion in 2023.
b. The global needle-free injectors market is expected to grow at a compound annual growth rate of 7.76% from 2023 to 2030 to reach USD 2.4 billion by 2030.
b. North America dominated the needle-free injectors market with a share of 42.47% in 2022. This is attributable to government support for quality healthcare, high purchasing power parity of patients, and increasing cases of needle-stick injuries.
b. Some key players operating in the ECMO machine market include PharmaJet, Crossject, Portal Instruments, NuGen Medical Devices, Inovio Pharmaceuticals, Antares Pharma, Inc, Aijex Pharma International Inc., Ferring B.V., Penjet Corporation, Medical International Technology Inc.
b. Key factors that are driving the needle-free injectors market growth include increasing preference towards self-administration, growing hospitalization of patients coupled with a surge in the number of hospitals across the world.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










