- Home
- »
- Medical Devices
- »
-
Neurology Clinical Trials Market Size, Industry Report, 2030GVR Report cover
![Neurology Clinical Trials Market Size, Share & Trends Report]()
Neurology Clinical Trials Market (2025 - 2030) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design, By Indication, By Indication by Study Design, By Indication by Phase, By Value Chain, By Region, And Segment Forecasts
- Report ID: GVR-4-68038-352-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Neurology Clinical Trials Market Summary
The global neurology clinical trials market size was estimated at USD 5.84 billion in 2024 and is projected to reach USD 8.42 billion by 2030, growing at a CAGR of 6.39% from 2025 to 2030. The market growth can be attributed to rising CNS disease burden, emerging trend of outsourcing, growing number of clinical trials & decentralized trials, emerging technological advancements, and demand for disease-modifying therapies.
Key Market Trends & Insights
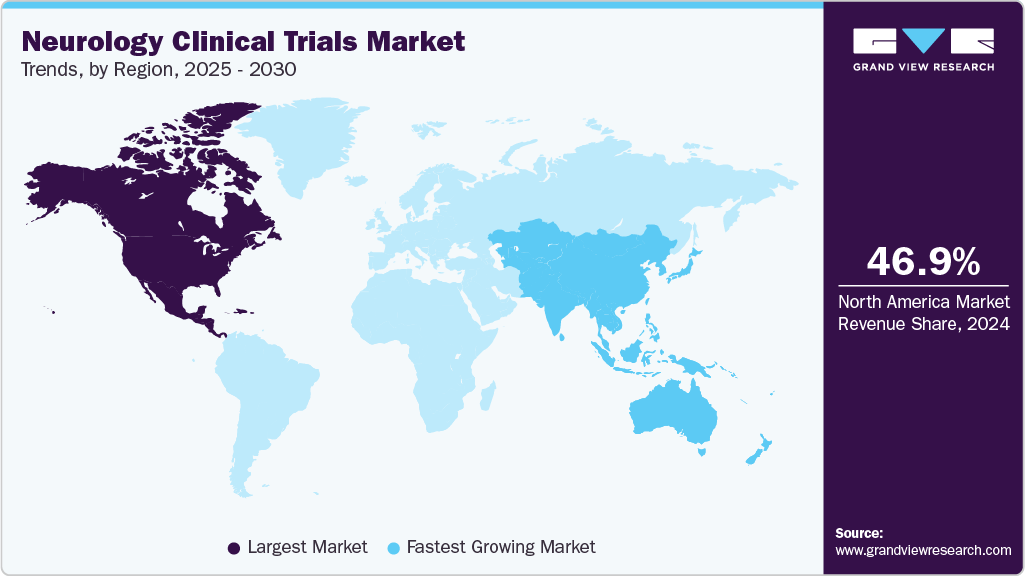
- North America dominated the global neurology clinical trials market with a share of 46.96% in 2024.
- The U.S. neurology clinical trials market accounted for the highest share of the North America region in 2024.
- By phase, the Phase III segment accounting for a revenue share of 42.14% in 2024.
- By study design, the interventional studies segment accounting for the largest revenue share in 2024.
- By indication, the Alzheimer’s disease segment accounted for the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 5.84 Billion
- 2030 Projected Market Size: USD 8.42 Billion
- CAGR (2025-2030): 6.39%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Besides, the rising number of diseases has increased the demand for neurology therapeutics. In addition, research & development (R&D) activities are enhancing core competencies, further fueling the growth. Moreover, the economic benefit of outsourcing compared to conducting an in-house study is also expected to propel market demand over the forecast period. The rising prevalence of Alzheimer’s disease, epilepsy, and stroke is significantly contributing to the market growth. Growing aging populations in developing and developed countries have led to a surge in age-related neurological disorders.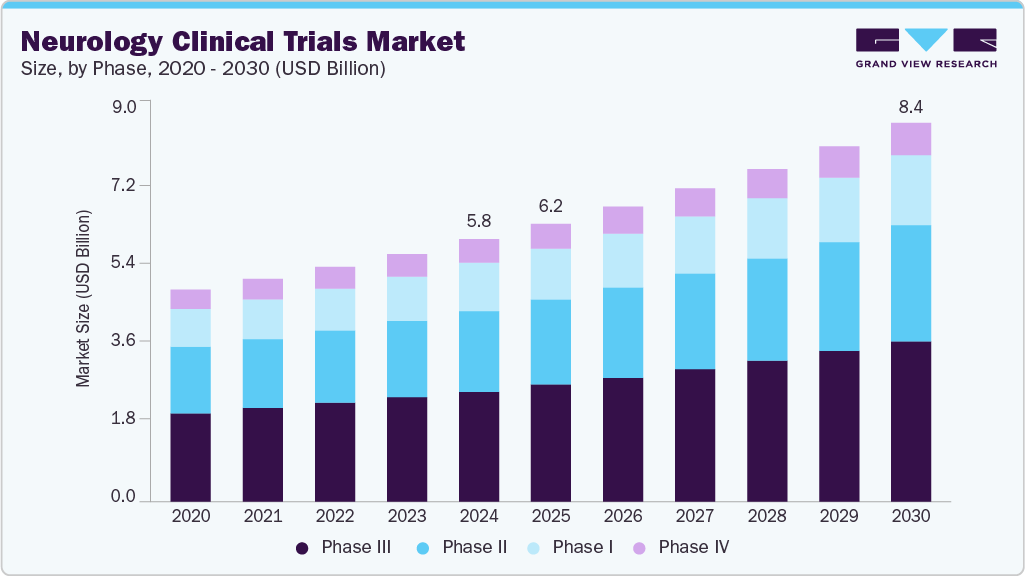
Furthermore, according to the research article published by IQVIA in March 2024, the CNS drugs accounted for 14% of the total industry pipeline drugs and are the second largest therapeutic area in active clinical trials. This strong development activity by market players demonstrates the significant investment and research and development activities in the market. For instance, in March 2025, Novartis announced to present its strength in its neuroscience portfolio at the American Academy of Neurology. This will include seven-year-old disability progression and safety outcomes from the ALITHIOS open-label extension study of Kesimpta (ofatumumab) in patients with relapsing multiple sclerosis (RMS). Moreover, the company aims to highlight its updates on pipeline candidates such as remibrutinib and iptacopan, underscoring its commitment to advancing innovative treatments for neurological disorders. Thus, constant development by market players and their growing product pipelines to meet the growing demand for patients would further contribute to market growth.
Moreover, a rising number of CROs are offering comprehensive services that encompass study design, patient recruitment, data management, and regulatory compliance, thereby streamlining the complex processes integral in neurology trials. Their expertise is particularly valuable in navigating the intricacies of neurological disorders, which often require specialized protocols and patient populations. By leveraging their extensive networks and technological capabilities, CROs facilitate more efficient and cost-effective trials, enabling faster progression from early-phase studies to market approval. This collaborative approach not only accelerates the development of novel therapies for neurological conditions but also ensures adherence to stringent regulatory standards, enhancing the quality and reliability of clinical research in this field. Such factors are anticipated to drive the market.
Opportunity Analysis
The neurology clinical trials market offers significant growth opportunities driven by the growing global prevalence of neurological disorders like Alzheimer’s, Parkinson’s, epilepsy, and multiple sclerosis. Increased investments in precision medicine, gene therapy, and drugs aimed at neuroinflammation are creating new pathways for innovation. The use of artificial intelligence, digital biomarkers, and remote monitoring technologies is facilitating early diagnosis, enhancing patient stratification, and enabling real-time data collection, which collectively improve trial outcomes.
In addition, emerging markets in the Asia-Pacific and Latin America regions offer access to untapped patient populations and reduced trial costs, making them appealing to international sponsors. The adoption of decentralized trial models and wearable cognitive assessment tools further boosts accessibility and patient compliance. Furthermore, regulatory agencies are becoming more adaptable in their approach to neurodegenerative drug development, highlighting the neurology sector as a key strategic focus for biopharmaceutical companies, contract research organizations (CROs), and technology providers.
Impact of U.S. Tariffs on the Global Neurology Clinical Trials Market
U.S. tariffs have had a significant indirect effect on the global neurology clinical trials industry, leading to increased costs and disruptions in supply chains for essential trial materials, such as laboratory equipment, imaging devices, and biologics. The tariffs imposed on goods from China have notably diminished access to affordable medical components and reagents that are vital for trial operations. This situation has resulted in increased operational expenses for U.S.-based sponsors and contract research organizations (CROs), prompting some to relocate trial activities to countries with fewer trade restrictions and lower operational costs.
Furthermore, the uncertainty surrounding tariff policies has complicated long-term planning for multinational neurology trials, particularly those necessitating cross-border regulatory alignment or the transport of biospecimens and diagnostic tools. In addition, these challenges have increased investment in local sourcing, nearshoring, and innovative digital trial technologies to lessen reliance on physical imports. Thus, the introduction of tariffs has created cost pressures and logistical complexities, reshaping trial design, site selection, and global vendor partnerships within the neurology research field.
Technological Advancements
Technological innovations are transforming neurology clinical trials by enhancing diagnosis, patient monitoring, and the accuracy of data collection. The implementation of digital biomarkers, wearable devices, and mobile applications allows for continuous, real-time tracking of neurological function, particularly for disorders such as Parkinson’s and Alzheimer’s. Furthermore, artificial intelligence and machine learning are increasingly utilized to analyze imaging data, forecast disease progression, and refine patient selection processes.

Virtual reality (VR) and cognitive assessment tools contribute to the non-invasive evaluation of brain activity. In addition, electronic data capture (EDC), electronic clinical outcome assessments (eCOA), and telemedicine platforms facilitate decentralized trial models, which improve patient retention and alleviate the burden on trial sites. Collectively, these advancements streamline timelines, reduce costs, enhance clinical outcomes, make trials more adaptive, precise, and focus on patient needs within the neurology research field.
Pricing Model Analysis
Neurology clinical trials utilize various pricing models to manage costs while maintaining operational flexibility effectively. Milestone-based pricing ties payments to critical trial milestones, such as patient enrollment or data lock, aligning incentives and encouraging timely progress while mitigating financial risks. Value-based pricing connects compensation to clinical outcomes or data quality, fostering innovation and accountability by rewarding high-performance results, which is particularly vital in trials with complex endpoints.
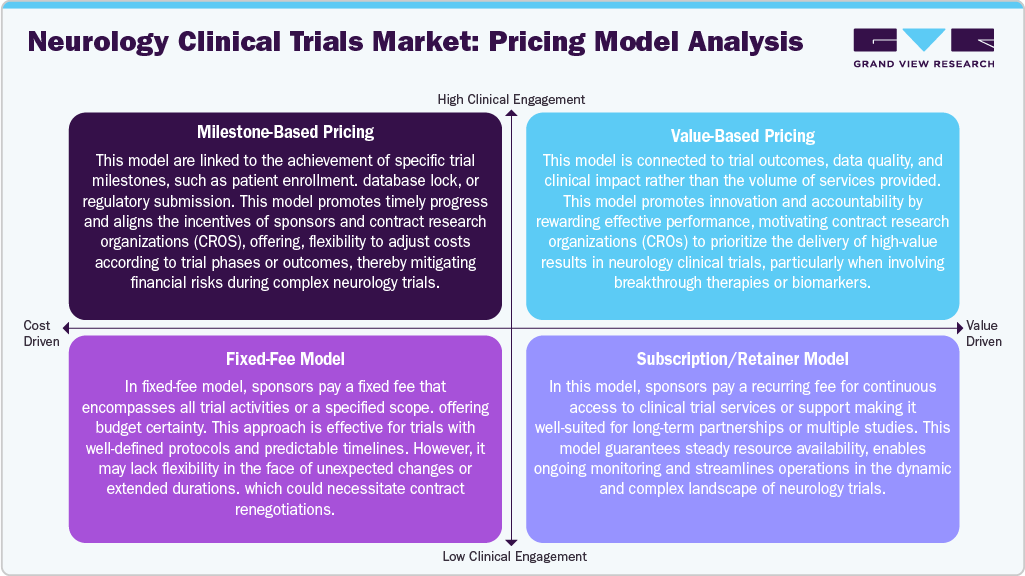
The fixed-fee model offers budget predictability by charging a set price for specific trial activities, making it appropriate for well-organized studies with clear protocols. However, it may lack adaptability for unforeseen changes. Lastly, the subscription or retainer model ensures ongoing access to clinical services through regular payments, making it well-suited for long-term collaborations or multiple concurrent trials. This approach supports steady resource availability and efficient operational management, especially beneficial in intricate neurology trials with changing requirements. Thus, these models represent a shift toward more strategic, transparent, and outcome-oriented pricing in neurology clinical research.
Market Concentration & Characteristics
The neurology clinical trials market growth stage is moderate, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
Neurology clinical trials are evolving with the introduction of innovative biomarkers, digital endpoints, and precision neuromodulation methods. New advancements such as AI-driven diagnostics, brain-computer interfaces, and gene therapies targeting neurodegenerative diseases are transforming trial methodologies. Moreover, adaptive designs and patient-focused technologies are increasing trial efficiency and facilitating recruitment in rare or progressive neurological conditions.

Strict data privacy regulations, changing ethical standards, and heightened regulatory oversight regarding trial endpoints influence neurology studies. The FDA and EMA prioritize patient safety, particularly in trials that involve cognitive function or vulnerable groups. There is a growing clarity in regulations concerning digital therapeutics and real-world evidence, which supports innovative approaches while maintaining compliance and ensuring data integrity.
M&A activity is increasing as major pharmaceutical companies acquire biotech firms that have strong neurology pipelines, particularly in the areas of Alzheimer's, Parkinson's, and rare CNS disorders. These transactions are centered around gene therapy, neuroinflammation, and digital neurotechnology. The goal of these acquisitions is to diversify portfolios, tap into innovative science, and reduce development risks through strategic consolidation.
CROs and clinical trial networks are broadening their offerings to accommodate the complexities of neurology trials by incorporating wearable devices, remote assessments, and neuroimaging technologies. They are also introducing decentralized models, eConsent processes, and virtual cognitive testing platforms to enhance access and compliance. In addition, global site networks are being fortified to integrate specialized neurology centers.
Neurology clinical trials are increasingly expanding into the Asia-Pacific, Eastern Europe, and Latin America regions, driven by rising patient populations, reduced trial costs, and enhanced regulatory environments. Sponsors are increasingly utilizing local CROs, multilingual digital resources, and remote monitoring to navigate logistical challenges and advance recruitment within emerging markets.
Phase Insights
In 2024, the Phase III segment dominated the neurology clinical trials industry, accounting for a revenue share of 42.14%. Phase III clinical trials in neurology are critical, large-scale studies designed to validate effectiveness, ensure long-term safety, and assess the therapeutic value of new medications for neurological disorders. Typically structured as randomized and double-masked studies, these trials may involve placebo or active control groups, and they encompass a wide range of patient populations from various locations. These trials utilize robust study designs and validated assessment tools, increasingly incorporating digital biomarkers and real-world evidence. Innovative methodologies, such as adaptive trial designs, targeted enrollment strategies, and subgroup analyses, enhance statistical significance and help identify patient populations that are most likely to benefit from treatment, which further contributes to market growth.
The Phase I segment is expected to grow significantly during the forecast period. The Phase I studies are designed to evaluate the safety of new devices or drugs, focusing on their tolerability, pharmacokinetics, and pharmacodynamics, particularly for treating neurological conditions. This phase assesses how a drug or device interacts with humans, including its metabolism, absorption, and excretion processes. It also investigates potential side effects that may arise from increased dosage levels and typically involves 20 to 80 healthy volunteers or individuals with the targeted disease.
Besides, given the complex nature of neurological disorders such as Alzheimer’s, Parkinson’s, multiple sclerosis, and rare genetic conditions, Phase I studies often integrate advanced imaging techniques, biomarker analyses, and neurophysiological assessments to better understand drug-target interactions and central nervous system (CNS) penetration. In addition, this shift towards first-in-class neurotherapeutics, including gene therapies, antisense oligonucleotides, and monoclonal antibodies, is expected to drive the innovative trial designs and adaptive methodologies to address variability among patients and the sensitive nature of neurological health.
Study Design Insights
In 2024, the interventional studies segment dominated the neurology clinical trials market, accounting for the largest revenue share. Interventional studies are categorized according to the type of intervention being investigated, which can include drugs or biologics, behavioral interventions, surgical procedures, and devices, all aimed at assessing their impact on neurological conditions. These studies actively manipulate participant conditions in a research setting to evaluate the effects of specific interventions on health outcomes. They play a crucial role in advancing treatment strategies for a wide range of disorders, including Alzheimer's disease, Parkinson's disease, epilepsy, stroke, multiple sclerosis, and various rare neurodegenerative diseases. Besides, over time, there has been a significant demand for phases I–IV interventional studies, with the majority focusing on drugs or biologics, followed by behavioral interventions, surgical procedures, and device-based studies.
The observational segment is expected to grow at a significant CAGR during the forecast period. Observational clinical trials in neurology focus on analyzing health outcomes in groups of patients without changing their treatment they receive. The primary goal of these studies is to gain insights into the natural progression of diseases, identify potential risk factors, and assess the effectiveness of treatments in real-world contexts. Furthermore, as new drugs emerge from the development pipeline, they are expected to drive the growth of observational studies. These trials are essential for gaining a comprehensive understanding of the progression and real-world implications of neurological disorders, all while preserving the integrity of patient care.
Indication Insights
The Alzheimer’s disease segment accounted for the largest market share in 2024. Clinical trials for Alzheimer’s disease (AD) aim to slow disease progression, enhance cognitive function, and manage behavioral symptoms in an increasingly aging population. As the focus shifts from symptomatic treatments to disease-modifying therapies, current trials increasingly address key areas such as amyloid-beta accumulation, tau pathology, neuroinflammation, and synaptic dysfunction. Recent advancements in biomarker development, encompassing cerebrospinal fluid (CSF) proteins, PET imaging, and blood-based diagnostics, facilitate earlier diagnosis and more precise patient selection. Clinical endpoints are evolving to incorporate both traditional cognitive assessment scales (e.g., ADAS-Cog, CDR-SB) as well as real-world functional outcomes. Such factors are expected to drive market growth.
The Huntington's disease segment is expected to grow at a significant CAGR during the forecast period. Clinical trials for Huntington’s disease (HD) aim to develop targeted therapies for this rare, inherited neurodegenerative disorder, characterized by progressive motor impairment, cognitive decline, and psychiatric issues. Besides its monogenic origin, resulting from a CAG repeat expansion in the HTT gene, HD offers a unique framework for precision medicine approaches, including gene silencing, antisense oligonucleotides (ASOs), and gene-editing techniques, which further support the market growth.
Value Chain Insights
In 2024, the in-house segment held the largest market share, driven by the increasing demand for neurology clinical trials and a growing number of sponsor organizations, primarily biopharmaceutical companies and academic institutions, opting not to outsource core trial operations. These trials offer essential benefits, allowing for comprehensive oversight of protocol design, site selection, patient engagement, data integrity, and regulatory compliance. Besides, this model is particularly preferred for early-phase trials, especially when proprietary technologies, sensitive intellectual property, or a high degree of scientific precision is required, such as in gene therapies for rare neurological conditions or precision-targeted biologics for Alzheimer’s and Parkinson’s diseases. Moreover, this approach provides strategic advantages, including streamlined communication, quicker decision-making processes, and enhanced alignment between clinical goals and corporate strategy, which further support the market growth.
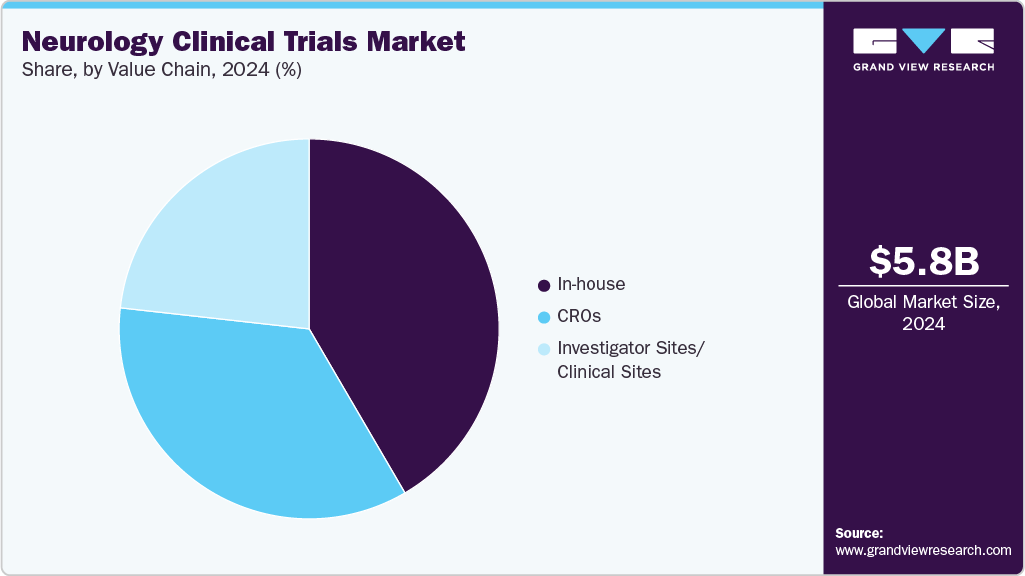
The contract research organizations (CROs) segment is expected to grow at a significant CAGR during the forecast period. CROs play a crucial role in the planning, execution, and management of neurology clinical trials, providing specialized knowledge and operational scalability throughout all phases of drug development due to the complexity and variability of neurological disorders such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, epilepsy, and rare neurogenetic conditions. Besides, biopharmaceutical sponsors are increasingly collaborating with CROs to reduce development risks, timelines, and gain access to therapeutic and regulatory expertise. Thus, neurology trials become increasingly innovative and data-driven, and CROs serve as essential facilitators of efficient, high-quality, and compliant clinical development within the global neurological research landscape.
Regional Insights
North America dominated the market with a share of 46.96% in 2024. This growth can be attributed to the increasing prevalence of neurodegenerative disorders such as Alzheimer’s, Parkinson’s, and multiple sclerosis. Besides, the presence of established outsourcing companies and stringent regulatory scenarios supports market growth. Moreover, the rising need for various drugs and medical devices to enhance neuroimaging, biomarkers, and digital health technologies is expected to drive the market growth over the estimated period.
In addition, the region has the presence of countries such as the U.S., Canada, and Mexico contribute to the growing number of clinical trials and increased regional spending. Besides, many companies are shifting their focus toward outsourcing to emerging market players due to the increasing regulatory requirements and long approval timelines. This has led to most outsourcing activities in North America. Furthermore, excessive costs associated with research have encouraged companies to focus on in-house clinical trials or outsource to CROs and investigator sites/clinical sites.
U.S. Neurology Clinical Trials Market Trends
The neurology clinical trials industry in the U.S.accounted for the highest share of the North America market driven by a presence of established CROs, an increasing number of clinical trials and growing trend for outsourcing companies to concentrate on their core competencies, mitigate associated risks, and boost drug and device innovations, thereby gaining a competitive edge. Besides, according to data from Clinicaltrials.gov, as of May 2025, there are anticipated to be 171 global studies focusing on neurology. A significant portion of these clinical trials taking place in the U.S. is creating lucrative opportunities for the clinical trial market. Besides, increased R&D spending on clinical trials coupled with increased prevalence of diseases such as Alzheimer’s disease, epilepsy, stroke, multiple sclerosis, and Parkinson’s disease is expected to drive the need for novel therapeutics and medical devices in the country.
The Canada neurology clinical trials marketis driven by a shifting demographic landscape, robust infrastructure, and supportive government initiatives. Besides, the rising incidence of neurological disorders such as Alzheimer's, Parkinson's, epilepsy, and stroke due to an aging population is anticipated to drive the market expansion. In addition, the presence of a well-established healthcare system, along with favorable government support, is also expected to enhance the development of innovative drugs and medical devices in the country. Moreover, the country's leadership in decentralized and patient-centered trial models supported by digital health technologies is enhancing participant recruitment and data collection processes. These factors are expected to advance neurological research and therapeutic development in Canada.
Europe Neurology Clinical Trials Market Trends
The Europe neurology clinical trials industry is driven by the presence of numerous multinational pharmaceutical companies, increasing investment in research and development, advancements in healthcare research, and the rise of contract research organizations (CROs) specializing in various phases of drug and device development. In addition, the strong network of the National Health Service (NHS), along with leading academic medical centers and government initiatives aimed at streamlining clinical research, provides considerable support to this market.
The neurology clinical trials market in Germany held the highest share in 2024. This growth can be attributed to rapid technological advancements, streamlined approval processes, and enhanced access to research data and quality clinical resources. The country is a key European hub for Contract Research Organization (CRO) services, which is anticipated to further boost market expansion in the coming years. Besides, strong healthcare infrastructure, efficient patient recruitment, a well-trained workforce, and a commitment to regulatory innovation make it an attractive destination for clinical neurology trials. In addition, partnerships among academic institutions, CROs, and biotech companies are accelerating neurological research, providing speed, regulatory clarity, and scientific excellence for global sponsors focused on brain health innovations.
The UK neurology clinical trials marketis anticipated to grow significantly over the forecast period. The country's growth is driven by the presence of numerous multinational pharmaceutical companies, increasing investment in research and development, advancements in healthcare research, and the rise of contract research organizations (CROs) specializing in various phases of drug and device development. In addition, the strong network of the National Health Service (NHS), along with leading academic medical centers and government initiatives aimed at streamlining clinical research, provides considerable support to this market.
Asia Pacific Neurology Clinical Trials Market Trends
The Asia Pacific neurology clinical trials industry is expected to grow at the fastest CAGR over the forecast period. Large and diverse populations of neurological disorders, an aging population, and changing lifestyles drive the region’s growth. Neurological disorders present significant challenges in the region, creating a rising need for effective treatment options and interventions. Besides, the presence of multinational pharmaceutical and medical device companies in countries like China, India, and South Korea is increasingly focusing on both in-house innovations and the use of outsourcing facilities such as Contract Research Organizations (CROs) and investigator sites/clinical sites for clinical trials, which are anticipated to fuel the market over the estimated period.
The neurology clinical trials market in China is driven by rapid technological progress, an increasing demand for new and cost-effective medical devices, and significant healthcare investment. Besides, the country is witnessing growth owing to increased involvement from both domestic sponsors and international companies. In addition, the presence of a vast patient population, a developed regulatory framework, and enhanced infrastructure further creates new opportunities for clinical research.
The Japan neurology clinical trials market isdriven by rapid technological innovations, an increasing demand for outsourcing services, and rising healthcare expenditures. Besides, the growing incidence of neurological disorders is expected to boost the demand for a variety of pharmaceutical products and medical devices aimed at addressing the challenges faced by patients with these conditions in the country. In addition, a strong commitment to neurological research, along with collaborative initiatives, is further fueling the development of new drugs and devices within the region.
The neurology clinical trials market in Japanheld a significant revenue share in the global market in 2024. The country has become a preferred location for pharmaceutical and medical device companies looking to innovate in the neurology space. In addition, the country’s growth is driven by factors such as a diverse patient pool, fast enrollment for clinical trials, lower labor costs, a growing healthcare infrastructure, a significant talent pool of technical experts, and overall cost efficiency. In addition, increasing government funding, the rise in the prevalence of neurological disorders, and an increased focus on neurology clinical trials, specifically Phase I trials, are expected to further propel market growth during the forecast period.
Latin America Neurology Clinical Trials Market Trends
The neurology clinical trials industry in Latin America is fueled by the developing medical devices industry. Besides, factors such as increasing life expectancy, growing awareness of home healthcare services, and a rising demand for technologically advanced medical devices for long-term care are contributing to this expansion. Furthermore, the expanding demand for medical devices is anticipated to drive the need for cost-effective diagnostic solutions. These factors are expected to drive market growth over the forecast period.
Brazil neurology clinical trials market is driven by advancements in in-vitro diagnostics, an increasing demand for diagnostic devices, and a supportive regulatory environment. In addition, the growing prevalence of chronic diseases, such as cancer, diabetes, and cardiovascular conditions, has increased the need for diagnostic devices for early detection. Besides, the combination of versatility, specialized expertise, focus on emerging regulations, and commitment to quality product innovations makes the Neurology Clinical Trials a unique offering for medical device companies.
Middle East & Africa Neurology Clinical Trials Market Trends
The neurology clinical trials industry in MEA is driven by the rising prevalence of lifestyle-related diseases and neurological diseases, high population growth, and increased demand for medication and medical devices. Besides, the region offers innovative patient diversity, cost advantage, infrastructure, and world-class medical facilities. In addition, the region offers untapped market opportunities for neurology clinical trials with expanding pharmaceutical and medical device companies.
The South Africa neurology clinical trials market is driven by the increasing burden of neurological disorders. Besides, the country is commonly utilized for clinical research due to diverse pools of potential participants across various therapeutic areas, which offer significant advantages for conducting trials. In addition, growing collaboration with local investigators, regulatory bodies, and healthcare establishments is expected to enhance trial management efficiency and ensure compliance, contributing to the market's growth.
Key Neurology Clinical Trials Company Insights
The key players operating across the market are adopting inorganic strategic initiatives such as partnerships, mergers, acquisitions, service launches, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in April 2024, Alzheon, Inc., mentioned the dosing of the first participant in a long-term extension of its pivotal APOLLOE4 Phase 3 trial. This trial evaluates a 265 mg oral tablet of ALZ-801/valiltramiprosate, given twice daily, for individuals in the initial stages of AD who carry two copies of the ε4 allele of the apolipoprotein E gene (APOE4/4 homozygotes). The early stages of AD include patients experiencing mild cognitive impairment attributable to AD, as well as those with mild AD.
Key Neurology Clinical Trials Companies:
The following are the leading companies in the neurology clinical trials market. These companies collectively hold the largest market share and dictate industry trends.
- Syneos Health
- Icon Plc
- Lindus Health
- AbbVie
- GlaxoSmithKline
- Aurora Healthcare
- Labcorp Drug Development
- Pfizer
- AstraZeneca
- Biogen
- Eli Lilly
- Alzheon
- GAP Network
- BDD Pharma
- Re:Cognition Health
Recent Developments
-
In April 2025, GSK is investing £38.5 million (USD 50 million) in collaboration with ABL Bio, a South Korea-based company, to create innovative treatments for neurodegenerative diseases. In addition to the initial payment, GSK will be responsible for up to £2.075 billion (USD 2.66 billion) in costs associated with R&D, regulatory approvals, and commercialization milestones, distributed across multiple potential programs outlined in the agreement.
-
In February 2025, Eli Lilly mentioned investments in obesity treatment, positioning itself as a leader alongside semaglutide manufacturer Novo Nordisk, thanks to its successful drug tirzepatide. In addition, the company plans to leverage its extensive pipeline of incretin therapies by exploring potential applications in neurology and immunology.
-
In December 2024, Novartis announced encouraging topline results from the Phase III STEER study. This pivotal trial evaluated the safety and effectiveness of the investigational intrathecal treatment onasemnogene abeparvovec (OAV101 IT) in treatment-naive patients with spinal muscular atrophy (SMA) Type 2, aged two to less than 18 years, who can sit but have never walked independently. Moreover, the efficacy and safety of OAV101 IT were compared to a sham control, which simulates the procedure of administering an investigational drug without providing any therapeutic effect.
Neurology Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 6.18 billion
Revenue forecast in 2030
USD 8.42 billion
Growth rate
CAGR of 6.39% from 2025 to 2030
Historical Year
2018 - 2024
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication, indication by study design, indication by phase, value chain, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Syneos Health; Icon Plc; Lindus Health; AbbVie; GlaxoSmithKline; Aurora healthcare; Labcorp Drug Development; Pfizer; AstraZeneca; Biogen; Eli Lilly; Alzheon; GAP Network; BDD Pharma; Re:Cognition Health
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Neurology Clinical Trials Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global neurology clinical trials market report based on phase, study design, indication, indication by study design, indication by phase, value chain, and region:
-
Phase Outlook (Revenue, USD Million, 2018 - 2030)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2018 - 2030)
-
Interventional
-
Observational
-
Expanded Access
-
-
Indication Outlook (Revenue, USD Million, 2018 - 2030)
-
Alzheimer’s Disease
-
Depression (MDD)
-
Parkinson's Disease (PD)
-
Epilepsy
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Huntington's Disease
-
Muscle Regeneration
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2018 - 2030)
-
Alzheimer’s Disease
-
Interventional
-
Observational
-
Expanded Access
-
-
Depression (MDD)
-
Interventional
-
Observational
-
Expanded Access
-
-
Parkinson's Disease (PD)
-
Interventional
-
Observational
-
Expanded Access
-
-
Epilepsy
-
Interventional
-
Observational
-
Expanded Access
-
-
Stroke
-
Interventional
-
Observational
-
Expanded Access
-
-
Traumatic Brain Injury (TBI)
-
Interventional
-
Observational
-
Expanded Access
-
-
Amyotrophic Lateral Sclerosis (ALS)
-
Interventional
-
Observational
-
Expanded Access
-
-
Huntington's Disease
-
Interventional
-
Observational
-
Expanded Access
-
-
Muscle Regeneration
-
Interventional
-
Observational
-
Expanded Access
-
-
Others
-
Interventional
-
Observational
-
Expanded Access
-
-
-
Indication by Phase Outlook (Revenue, USD Million, 2018 - 2030)
-
Alzheimer’s Disease
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Depression (MDD)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Parkinson's Disease (PD)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Epilepsy
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Stroke
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Traumatic Brain Injury (TBI)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Amyotrophic Lateral Sclerosis (ALS)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Huntington's Disease
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Muscle Regeneration
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Others
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
-
Value Chain Outlook (Revenue, USD Million, 2018 - 2030)
-
In-house
-
CROs
-
Investigator Sites/Clinical Sites
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
Argentina
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global neurology clinical trials market size was estimated at USD 5.84 billion in 2024 and is expected to reach USD 6.18 billion in 2025.
b. The global neurology clinical trials market is expected to grow at a compound annual growth rate of 6.39% from 2025 to 2030 to reach USD 8.42 billion by 2030.
b. North America dominated the neurology clinical trials market, with a share of 46.96% in 2024. This is attributable to the increasing prevalence of neurodegenerative disorders such as Alzheimer’s, Parkinson’s, and multiple sclerosis. In addition, the presence of established outsourcing companies and stringent regulatory scenarios supports market growth. Moreover, the rising need for various drugs and medical devices to enhance neuroimaging, biomarkers, and digital health technologies is expected to drive the market growth over the estimated time period.
b. Some of the players operating in the neurology clinical trials market are Syneos Health, Icon Plc, Lindus Health, AbbVie, GlaxoSmithKline, Aurora healthcare, Labcorp Drug Development, Pfizer, AstraZeneca, Biogen, Eli Lilly, Alzheon, GAP Network, BDD Pharma, and Re:Cognition Health
b. Key factors driving market growth include rising CNS disease burden, emerging outsourcing trend, growing number of clinical trials & decentralized trials, emerging technological advancements, and demand for disease-modifying therapies. Besides, the rising number of diseases has increased research and development (R&D) activities, which further enhance core competencies supporting market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










