- Home
- »
- Medical Devices
- »
-
Neurovascular Devices Market Size & Share Report, 2030GVR Report cover
![Neurovascular Devices Market Size, Share & Trends Report]()
Neurovascular Devices Market Size, Share & Trends Analysis Report By Device (Neurothrombectomy Devices, Support Devices), By Therapeutic Applications, By Size (0.027 inch, 0.021 inch), By End-Use, By Region, And Segment Forecast 2023 - 2030
- Report ID: GVR-2-68038-851-0
- Number of Report Pages: 120
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Report Overview
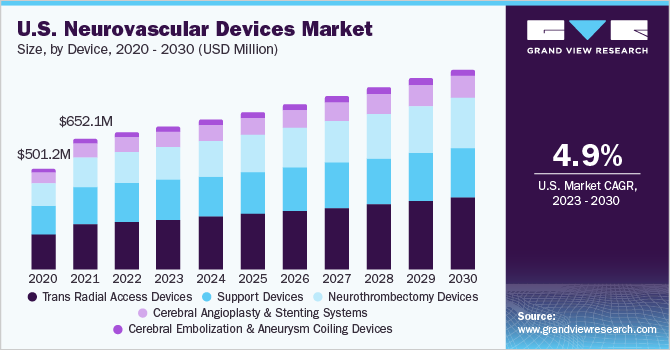
The global neurovascular devices market size was valued at USD 2.84 billion in 2022 and is anticipated to expand at a CAGR of 6.01% during the forecast period. The neurovascular devices market is likely to be driven by the increasing prevalence of neurological illnesses, technological developments, and increased demand for minimally invasive procedures.
Interventional neurology devices are used to diagnose and treat central nervous system and brain vascular disorders. Endovascular, catheter-based procedures, angiography, and fluoroscopy are all part of the interventional neurology. Catheter angiography is one of the oldest in-vivo brain vascular imaging techniques used to diagnose a variety of neurological diseases including cerebral aneurysm, arteriovenous malformations, intracranial stenosis, arteriovenous fistula, and vasculitis.
Increase in incidences of neurological disorders such as brain/ cerebral aneurysm, strokes, epilepsy amongst many others in several countries is expected to drive the neurovascular devices market growth. According to the NHS England, around 1 in 12,500 people in England have ruptured brain aneurysm every year. Furthermore, brain aneurysm affects approximately 3.0% of the persons in the U.K.. Furthermore, the Brain Aneurysm Foundation estimates that 6.0 million Americans are impacted by brain aneurysms each year.
Every year, the disease kills half a million people globally. Approximately 40.0% of ruptures result in death and approximately 66.0% of those who survive has some permanent brain damage. Thus, the rapidly and vast growing patient population base for the target diseases across the key markets is primarily responsible for the significant volume consumption of neurovascular devices, leading to the market growth.
A slew of potential new technologies, merger and acquisitions in the neurovascular space are on the horizon. For instance, in October 2022, the Medtronic Neurovascular Co-Lab Platform was created to help speed much-needed innovation in stroke treatment and care. Their community platform aims to transform ideas and technology into breakthrough global medicines by giving entrepreneurs and physicians with insight and care, and, most importantly, ensuring that innovation reaches as many people as possible around the world. As a result, the availability of new treatment choices and an expanding variety of tactics will drive up demand for the neurovascular devices in the coming years.
Minimally invasive surgeries are gaining popularity owing to the reduced risk and trauma associated with these procedures. Smaller incisions decrease postoperative pain and facilitate speedy recovery leading to high adoption of these procedures and triggering growth of R&D in this field. Several key players are investing in R&D for the launch of innovative minimally invasive surgical instruments. The endovascular coiling used for the treatment of intracranial aneurysm is one of the minimally invasive procedures that are widely recommended by the physicians. In this procedure, a microcatheter is inserted in the groin area through the artery consisting an aneurysm. Platinum coils are then released as the coil promotes clotting of aneurysm and prevents blood from entering other regions of the brain.
Device Insights
Cerebral embolization and aneurysm coiling devices held the largest market share of around 34.79% in 2022. Coil embolization is a minimally invasive procedure for the treatment of aneurysms, wherein, the material closes the sac and reduces the risk of bleeding. A steerable catheter is inserted through the groin and guided to the brain. Rising prevalence of aneurysm is expected to propel the segment during the forecast period. These devices are further classified into embolic coils, flow diversion devices, and liquid embolic agents.
For instance, in January 2023, Luidx Medical Technologies, Inc. announced the success of the Embolic IMPASS Device in an in-vivo research linked to meningeal middle artery (MMA) embolization. This can be used to treat chronic subdural hematomas on the surface of the brain. Instead of surgery, minimally invasive MMA embolization catheter-based may be employed.
Kaneka Corporation gained U.S-FDA approval in April 2020 for its novel brain aneurysm Embolization Coil (product name: i-ED COIL), which it has been marketing since September 2020 through Kaneka Medical America LLC. Kaneka wants to grow its sales in the U.S beginning in August 2021 by entering a sales partnership arrangement with Asahi Intecc USA, Inc.
Neurothrombectomy devices segment is expected to expand at the highest CAGR during the forecast. Increasing number of growth strategies by the key market players such as product launch, merger & acquisition and others along with the rising prevalence of acute ischemic stroke will drive the segment growth in near future. For instance, in January 2023, Therma Bright Inc. invests in Israeli Ischemic Stroke Blood Clot Retriever Technologies Startup Inretio Inc. The PREVA basket "ensnares" the clot, enclosing it and shielding the brain from any sub-clots that break off during the thrombectomy treatment.
In February 2022, Perfuze, which is developing next-generation catheter-based aspiration technology to treat acute ischemic stroke caused by major artery obstruction, has concluded a USD 23.94 million Series A financing round. Additionally, Rapid Medical, a developer of novel neurovascular devices, announced FDA 510(k) clearance for TIGERTRIEVER 13 for major vessel occlusions during the 19th Annual Conference of the Society of NeuroInterventional Surgery (SNIS) in Toronto in 2022. TIGERTRIEVER 13 is the world's smallest revascularization device, designed to remove thrombus from sensitive brain blood arteries following an ischemic stroke.
Moreover, in April 2021, Perflow Medical reported the first clinical successful application of the Stream17 dynamic neurothrombectomy net in the treatment of hemorrhagic and ischemic stroke patients. Perflow Medical's product portfolio includes the recently commercialized Cascade17 dynamic nonocclusive reconstructive net for coil embolization of cerebral aneurysms, as well as Stream17.
Therapeutic Application Insights
Stroke held the largest market share of around 57.10% in 2022, owing to the factors such as increasing prevalence of hypertension, stroke, and other neurological disorders. According to the CDC, 1 in 6 people worldwide will have a stroke in their lifetime and every year, more than 795,000 people in the U.S have a stroke. Strokes are the second leading cause of death globally, and account for an approximate 140,000 deaths in the U.S annually. There are various initiatives undertaken by the government worldwide to prevention stroke. Some of the examples include, in October 2021, the Udall Center at the University of Minnesota Medical School received a new USD 11.3 million grant from the National Institute of Neurological Stroke.
Advent of technologically advanced products is also powering the segment growth. For instance, in January 2023, Infinity Neuro has announced that its Inspira aspiration catheters have acquired CE Mark approval and are now available for purchase in Europe. This is Infinity Neuro's first offering; the company intends to launch a broad spectrum of products for the treatment of ischemic and hemorrhagic stroke during 2023 and beyond.
Cerebral aneurysm segment is expected to grow at the highest CAGR during the forecast period, primarily as a result of the increasing prevalence of cerebral aneurysm. According to the Brain Aneurysm Foundation, around 6.5 million people in the U.S have an unruptured brain aneurysm, with approximately 30,000 developing a rupture each year. Also, the increasing number of clinical trials and the introduction of technologically improved products will fuel category expansion in the near future.
For instance, in February 2023, VESALIO reported the successful first use of its NeVa VS device for the treatment of post-aneurysmal subarachnoid haemorrhage (aSAH) cerebral vasospasm in the U.S. Vasospasm is the most common aSAH consequence and the leading cause of mortality and morbidity. Furthermore, in February 2023, EndoStream Medical, a business that develops solutions for brain aneurysms, announced the enrollment of the first patient in the TORNADO-US clinical study in the U.S.
The Nautilus intrasaccular system from the company will be evaluated for the treatment of cerebral aneurysms in the trial. Moreover, in April 2021, the FDA has authorized Medtronic's clot-resistant implant for the treatment of brain aneurysms, which is expected to drive segment growth in the near future.
Size Insights
0.021" held the largest market share of around 27.26% in 2022. Segment growth can be attributed to various advantages provided by the 0.021” devices, along with various product launches and product approvals. For instance, in April 2021, Bendit Technologies' 0.021" Bendit21 microcatheter has been cleared by the U.S FDA for use in the brain, peripheral, and coronary vasculature. The approval came several months after the Bendit21 microcatheter was successfully used in two life-saving surgeries in the U.S.
Furthermore, in October 2021, Evasc Neurovascular has introduced the eCLIPs Bifurcation Flow Diverter, the third version of their eCLIPs device. The latest generation of eCLIPs features a shapeable delivery wire, a reduced size suitable with 0.027" and 0.021" ID microcatheters, and electrolytic detachment for more effective treatment of bifurcation cerebral aneurysms.
Others segment is expected to develop at the highest CAGR during the forecast period. The others segment includes devices sized at 0.015”, 0.060”, 0.84”, 0.091”, and 0.074”. The availability and benefits offered by these products are expected to drive segment expansion. For example, the AVIGO 0.014" hydrophilic guidewire seamlessly supports tracking for 021 and 027 systems. Furthermore, this device aids in catheter crossing and stability. Therefore, the abundance of products in this area is likely to promote market growth during the forecast period.
End-use Insights
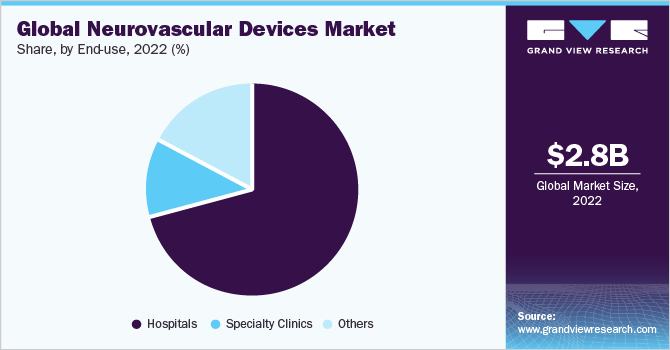
Hospital held the largest market share of around 70.95% in 2022. The growth of this segment can be attributed primarily to the increasing patient pool suffering from neurovascular disorders, such as ischemic & hemorrhagic stroke, brain aneurysm, Traumatic brain injury (TBI), and Arteriovenous Malformation (AVM). For instance, according to World Stroke Organization, throughout their lifetime, 1 in 4 persons over the age of 25 years is expected to experience a stroke. Every year, 13.7 million people are expected to experience their first stroke, and 5.5 million of them may die. Without suitable action, it appears that the annual death toll will increase to 6.7 million.
Moreover, more than 15.0 million people are living with the impact of hemorrhagic stroke globally. Furthermore, availability of technologically advanced neurovascular catheters, coupled with favorable reimbursement policies, can further enhance neurovascular device market growth in hospitals during the forecast period. For instance, Medtronic offers comprehensive services to secure and maintain coverage & payment for various neurovascular devices.
In addition, increasing number of patients being admitted to hospitals due to the surgeries, therapies, and treatments is expected to favor the neurovascular device market. Thus, subsequent increase in number of patients across the globe, launch of technologically advanced products, and favorable reimbursement policies are leading to a growing demand for the hospital treatments.
Other segment is expected to expand at the highest CAGR during the forecast period. The others segment includes Ambulatory Surgical Centers (ASCs), emergency care centers, and long-term care centers. Increase in number of ASCs and other healthcare modern facilities is expected to drive the segment. For instance, according to ASC Becker, the total number of Medicare-certified ASCs in the U.S. is nearly 5,700. Thus, increasing number of ASCs and rising awareness about availability & advantages of other medical facilities are expected to help this segment grow during the forecast period.
Regional Insights
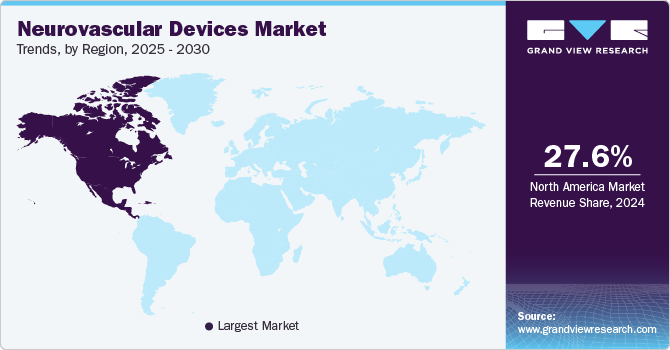
North America dominated the neurovascular devices market with the share of 26.44% in 2022 owing to the presence of key manufacturers such as Penumbra, Inc., Stryker Corporation, Johnson & Johnson, and Merit medical systems, Inc. in the region. In February 2023, Phenox Inc. stated that their pRESET Thrombectomy Device has been cleared by the FDA for use in the treatment of acute ischemic stroke. The product pRESET, which has been marketed in Europe for more than a decade, has now been approved for use in the U.S. Furthermore, rising prevalence of neurological disorders, and increasing demand for the minimally invasive surgical procedures are driving the neurovascular devices market growth in this region.
Furthermore, increased R&D expenditure by the manufacturers and increased government efforts are significantly contributing to regional market growth. Medtronic Neurovascular Clinical Affairs, for example, is financing a clinical trial to evaluate the safety and efficacy of the 'Pipeline Vantage Embolization Device' with Shield Technology in the treatment of intracranial aneurysms within the specified indication for usage.
Also, recently in April 2021, the Society of Neurointerventional Surgery (SNIS) and NeuroPoint Alliance (NPA) are collaborated with the Society of Vascular and Interventional Neurology (SVIN) to the NeuroVascular Quality Initiative-Quality Outcomes Database (NVQI-QOD). The strategic alliance marks a major step forward for the Neuro Vasc Technologies in bringing a full neurovascular platform to market.
Asia Pacific is expected to expand at the highest CAGR during the forecast period due to the factors such as rising target disease burden. It is estimated that about 30 million people in India suffer from various forms of neurological diseases and the average prevalence rate is as high as 2,394 patients per 1,00,000 of the population. Furthermore, factors such as rising healthcare expenditure, entry of key players in the region are also contributing towards the market growth. For instance, in March 2022, India Medtronic Private Limited announced the creation of India's first registry dedicated for the collecting of real-world data related to the use of revascularization devices in patients with acute ischemic stroke (AIS).
Key Companies & Market Share Insights
Key market players are focusing on the launch of innovative types of medical devices, growth strategies, and technological advancements. For instance, in April 2022, MicroVention, Inc., a Terumo Corporation wholly owned subsidiary, announced the completion of its first enrollment in STRAIT, a multi-center, prospective observational EU study to evaluate the performance and safety of the BOBBY balloon guide catheter for endovascular treatment of acute ischemic stroke.
In North America and Europe, the BOBBY Balloon Guide Catheter is currently approved. It was designed to improve compatibility and navigability with the SOFIA Plus 6Fr Aspiration Catheter while also simplifying balloon preparation. These advancements in neurovascular devices market are anticipated to boost the market growth during the forecast period. Some of the key players in global neurovascular devices market include:
-
Medtronic
-
Johnson and Johnson Services Inc.
-
Penumbra, Inc.
-
Microport Scientific Corporation
-
Stryker
-
Microvention Inc (Terumo Corporation)
-
Codman Neuro (Integra Lifesciences)
Neurovascular Devices Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 3014.04 million
Revenue forecast in 2030
USD 4.53 billion
Growth Rate
CAGR of 6.01% from 2023 to 2030
Volume in 2022
2,015,661 Unit
Volume forecast in 2030
3,221,883 Unit
Growth Rate
CAGR of 6.03% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million, volume unit, CAGR from 2023 to 2030
Segments covered
Device, therapeutic application, size, end-use, regional
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; U.K.; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Report coverage
Revenue, competitive landscape, growth factors, trends
Key companies profiled
Medtronic; Johnson and Johnson Services Inc.; Penumbra, Inc.; Micro port Scientific Corporation; Stryker; Microvention Inc. (Terumo Corporation); Codman Neuro (Integra Lifesciences)
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Neurovascular Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels in addition to provides an analysis on the latest trends and opportunities in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global neurovascular devices market report based on the device, therapeutic application, size, end use, and region:
-
Device Outlook (Revenue USD Million; Volume Unit; 2018 - 2030)
-
Cerebral Embolization and Aneurysm Coiling Devices
-
Embolic coils
-
Flow diversion devices
-
Liquid embolic agents
-
-
Cerebral Angioplasty and Stenting Systems
-
Carotid artery stents
-
Embolic protection
-
-
Neurothrombectomy Devices
-
Clot retrieval devices
-
Suction devices/aspiration catheters
-
Vascular snares
-
-
Support Devices
-
Micro catheters
-
Micro guidewires
-
-
Trans Radial Access Devices
-
-
Therapeutic Application Outlook (Revenue USD Million; 2018 - 2030)
-
Stroke
-
Cerebral Artery
-
Cerebral Aneurysm
-
Aneurysmal Subarachnoid Hemorrhage
-
Others
-
-
Others
-
-
Size (in Inches) Outlook (Revenue USD Million; 2018 - 2030)
-
0.027"
-
0.021"
-
0.071"
-
0.017"
-
0.019"
-
0.013"
-
0.058"
-
0.068"
-
Others
-
-
End-use Outlook (Revenue USD Million; 2018 - 2030)
-
Hospitals
-
Specialty Clinics
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
Germany
-
U.K.
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global neurovascular devices market size was estimated at USD 2.84 billion in 2022 and is expected to reach USD 3014.04 million in 2023.
b. The global neurovascular devices market is expected to grow at a compound annual growth rate of 6.01% from 2023 to 2030 to reach USD 4.53 billion by 2030.
b. The cerebral embolization and aneurysm coiling devices dominated the global neurovascular devices market and accounted for the largest revenue share of 34.79%.
b. The stroke segment dominated the global neurovascular devices market and accounted for the largest revenue share of 57.10%.
b. North America held the largest share of 26.44% in 2022 due to the presence of key manufacturers in the region, an increase in R&D investments, and a rise in the number of government initiatives.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





