- Home
- »
- Pharmaceuticals
- »
-
Preclinical Bioanalytical Testing Services Market Report, 2033GVR Report cover
![Preclinical Bioanalytical Testing Services Market Size, Share & Trends Report]()
Preclinical Bioanalytical Testing Services Market (2025 - 2033) Size, Share & Trends Analysis Report By Molecule (Small, Large), By Test (ADME, PD, PK), By Workflow, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-789-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Preclinical Bioanalytical Testing Services Market Summary
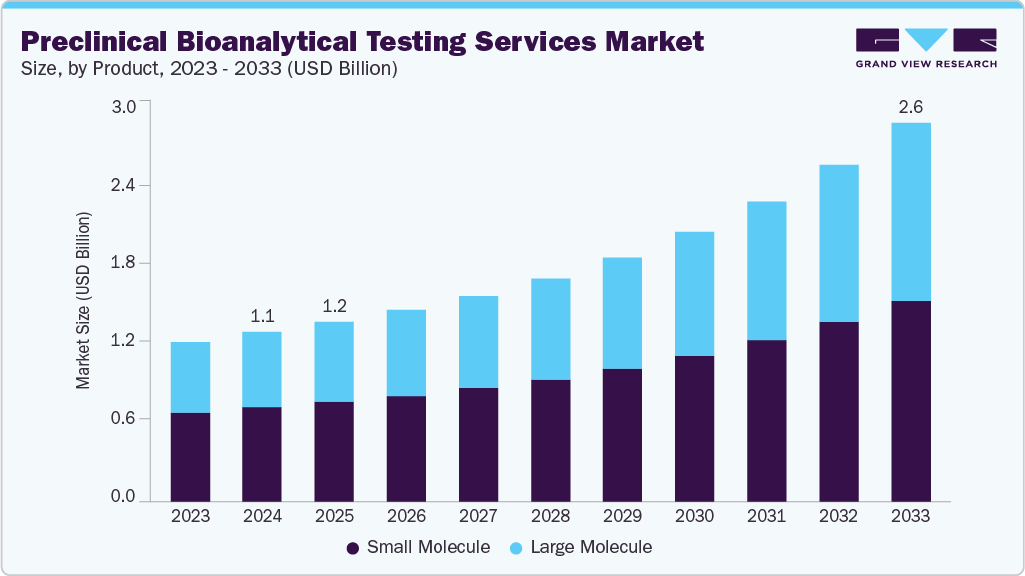
The global preclinical bioanalytical testing services market size was estimated at USD 1.17 billion in 2024 and is projected to reach USD 2.62 Billion by 2033, growing at a CAGR of 9.76% from 2025 to 2033. The market's growth is driven by the rising demand for new drug development, the increasing prevalence of chronic & rare diseases, and the expansion of biologics and cell & gene therapy development.
Key Market Trends & Insights
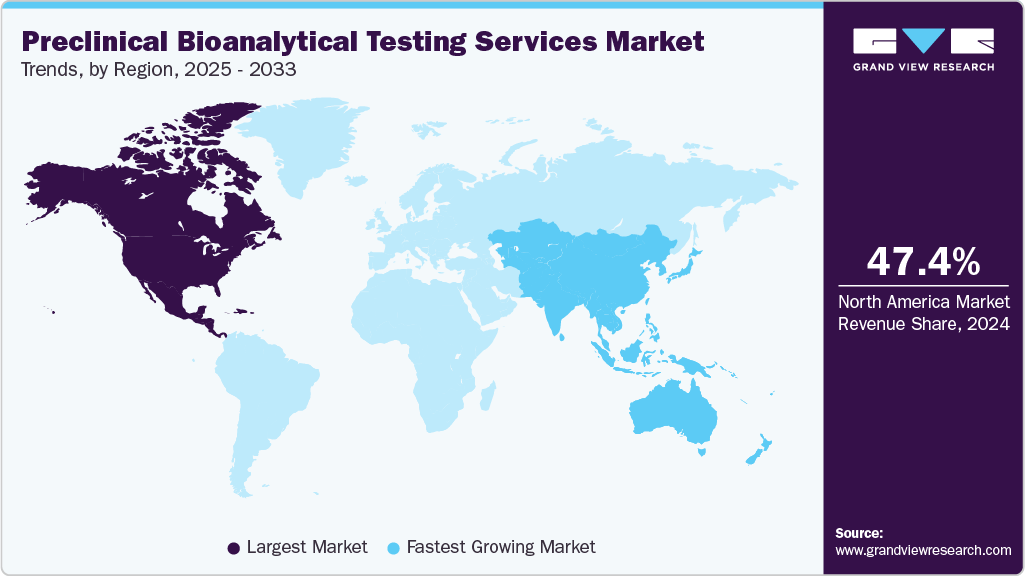
- The North America preclinical bioanalytical testing services market held the largest global revenue share of 47.42% in 2024.
- The preclinical bioanalytical testing services industry in the U.S. is expected to grow significantly over the forecast period.
- By molecule, the small molecule segment led the market with the largest revenue share of 55.65% in 2024.
- Based on test, the bioequivalence segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.17 Billion
- 2033 Projected Market Size: USD 2.62 Billion
- CAGR (2025-2033): 9.76%
- North America: Largest Market in 2024
- Asia Pacific: Fastest Growing Market
Furthermore, stringent regulatory requirements have heightened the need for precise pharmacokinetic and toxicokinetic analyses during preclinical studies, thereby fueling the demand for specialized bioanalytical testing servicesOther factors contributing to growth include rising advances in bioanalytical technologies, such as LC-MS/MS and immunoassays, increasing cost efficiency, and the growing adoption of advanced bioanalytical technologies.
The rapid expansion of biologics, biosimilars, monoclonal antibodies, and cell and gene therapies is fueling the requirement for advanced preclinical bioanalytical testing services. The most complex molecules require highly sensitive, specific, and validated analytical methods to assess their pharmacokinetics, immunogenicity, and safety. Besides, the drug pipelines are increasingly shifting towards large-molecule therapeutics, and bioanalytical laboratories must develop innovative assays and platforms to meet regulatory expectations. This expansion drives the need for specialized expertise, validation processes, and technologies in preclinical bioanalysis to support the accurate and efficient development of drugs.
Moreover, advancements in high-sensitivity platforms transform preclinical bioanalytical testing services by enabling precise detection and quantification of low-abundance biomarkers, proteins & drug molecules. In addition, LC-MS/MS, ligand-binding assays, and hybrid technology platforms are enhancing the analytical accuracy, reproducibility, and throughput. These innovations support the characterization of complex biologics and cell or gene therapies with improved sensitivity and lower detection limits. In addition, automated sample preparation, microfluidics & multiplexing further streamline workflows, reduce variability, increase efficiency, and ensure reliable data generation, critical for early-stage drug development and regulatory compliance.
Furthermore, regulatory agencies such as the FDA, EMA, and PMDA enforce stringent guidelines for bioanalytical validation, pharmacokinetic analysis, toxicology profiling, and biomarker quantification to ensure the safety and reliability of drugs before human trials. Pharmaceutical companies rely on certified preclinical bioanalytical testing partners with GLP-compliant infrastructure, validated technologies, and robust reporting capabilities to meet regulatory expectations. This increases outsourcing preferences to minimize compliance risks, avoid delays in trial approvals, and enhance submission accuracy. As adherence to strict validation protocols becomes essential, demand for high-quality, audit-ready bioanalytical services surges, accelerating market expansion.
Opportunity Analysis
The preclinical bioanalytical testing services market presents strong growth opportunities driven by favorable regulatory environments, the rising development of biologics and advanced therapies, increased outsourcing by small and mid-sized biopharmaceutical companies, technological advancements in analytical platforms, and growing expansion into emerging markets. In the market, rising biologics and advanced therapy development are creating demand for specialized analytical solutions to support the expanding pipeline of therapies, further expanding the need for high-quality, regulatory-compliant preclinical testing services. Moreover, increasing outsourcing by small and mid-sized biopharma companies fuels the market growth, as most companies lack the infrastructure and technical expertise for in-depth bioanalytical work, further creating collaboration with CROs and CDMOs to accelerate drug development. Furthermore, expanding into emerging markets such as Asia Pacific and Latin America is expected to offer new growth opportunities supported by cost advantages, skilled scientific talent, and rising local R&D investments, positioning these regions as key growth frontiers for global preclinical bioanalytical service providers.
Technological Advancements
The preclinical bioanalytical testing services industry presents significant growth opportunities, driven by enhancements in precision, efficiency, and data integrity. High-sensitivity LC-MS/MS systems enable the detection of trace-level compounds, thereby improving quantification accuracy in pharmacokinetic and toxicokinetic studies. In addition, the market has increased requirements for ligand-binding and hybrid assays that combine the strengths of LC-MS and immunoassay techniques, enabling the sensitive analysis of complex biologics and biosimilars. Moreover, the growing integration of automation and robotics streamlines sample handling and data processing, reducing human error and fueling productivity for large-scale studies.

The digital data management and AI-driven tools are expected to enhance data traceability, support predictive modeling, and ensure regulatory compliance through real-time monitoring and analytics. Furthermore, multiplex and high-throughput platforms enable the simultaneous analysis of biomarkers, optimizing resource utilization and accelerating study timelines. Thus, these technological advancements are expected to increase testing speed, accuracy, and reproducibility, further supporting the service providers in meeting the growing demand for high-quality preclinical bioanalysis in biologics, biosimilars, and advanced therapies while maintaining cost efficiency and compliance with stringent global regulatory standards.
Pricing Analysis
The preclinical bioanalytical testing services market utilizes a range of pricing models tailored to project scope and client requirements. Ideally, milestone-based pricing links payments to specific project stages, such as method validation or report submission, promoting accountability and shared risk. In addition, the value-based pricing ties costs to delivered outcomes, such as data quality or regulatory success, rewarding performance and innovation.
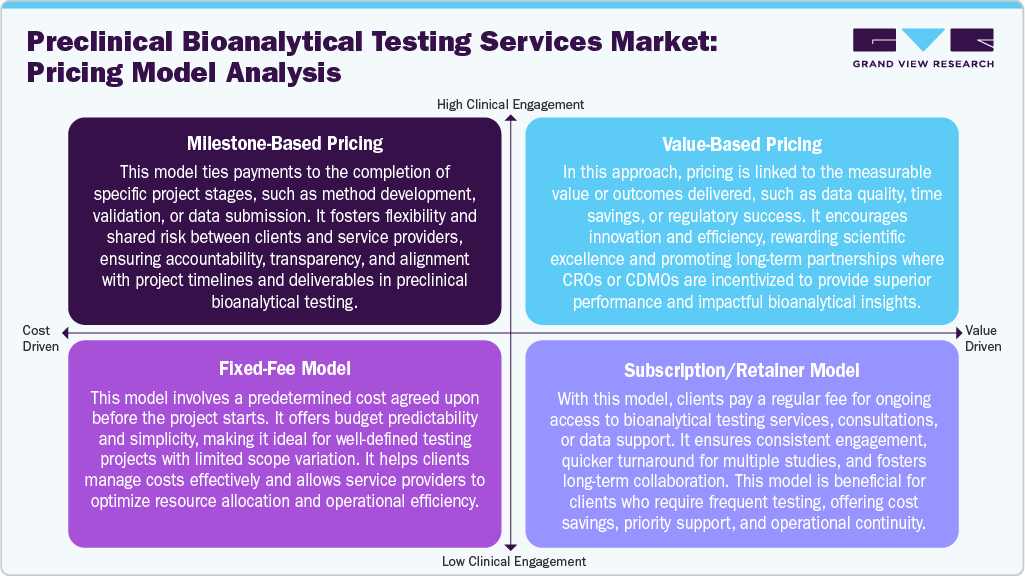
Moreover, the fixed-fee models offer a predetermined price for clearly defined projects, ensuring budget predictability and cost control. Furthermore, the subscription model offers clients ongoing access to testing services and scientific support for a recurring fee, facilitating long-term collaboration and expedited project execution. These pricing structures enable CROs and CDMOs to attract diverse clients while maintaining transparency, efficiency, and profitability. Such factors are expected to drive market growth.
Market Concentration & Characteristics
The preclinical bioanalytical testing services market growth stage is moderate, and the pace of growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
In preclinical bioanalytical testing services, innovation is driven by the implementation of advanced technologies, including high-sensitivity LC-MS/MS, ligand binding assays, and novel biomarker analytics. Integrating digital platforms, automation, and AI-enabled data analysis significantly enhances accuracy, efficiency, and throughput. Thus, CROs adopting these technologies can provide faster and more reliable bioanalytical results, which are essential for supporting complex biologics, biosimilars, and gene and cell therapy programs.

The preclinical bioanalytical testing services market has seen an increase in M&A activities, as many companies seek to broaden their capabilities, expand their regional footprint, and incorporate complementary technologies. With M&A activities, the CROs and CDMOs can enhance their service offerings with specialized knowledge in bioassay development, mass spectrometry, and biomarker validation, ultimately providing comprehensive bioanalytical solutions.
Stringent regulatory frameworks governing preclinical bioanalytical testing for biologics and small molecules have a significant influence on market operations. Adhering to GLP and evolving guidelines from the FDA, EMA, and OECD are essential for ensuring data integrity, assay validation, and accurate pharmacokinetic/toxicokinetic reporting.
CROs are extending their offerings beyond traditional small-molecule bioanalysis to encompass biologics, biomarkers, and studies related to gene and cell therapies. This approach enables CROs to offer comprehensive continuity from preclinical to clinical bioanalytical services, attracting a broader client base and strengthening their global market positions.
Global CROs are increasingly broadening their preclinical bioanalytical testing operations into emerging markets like India, China, and Singapore, motivated by cost efficiency and scientific expertise. By establishing GLP-compliant laboratories across Asia-Pacific, Europe, and North America, they can improve client access, reduce turnaround times, and ensure localized regulatory compliance, thereby enabling efficient support for multinational preclinical development programs.
Molecule Insights
The small molecule segment captured the largest revenue share of 55.65% in 2024. The growth of the segment can be attributed to rising demand for early drug development support and increasing outsourcing by pharma and biotech companies. Besides, bioanalytical testing of small molecules is crucial for gaining insights into their ADME properties, determining the appropriate dosage, evaluating the therapeutic effectiveness, and safeguarding patient well-being. Other factors contributing to segment growth include advancements in analytical technologies, stringent regulatory requirements for data accuracy, and the growing pipeline of small-molecule therapeutics targeting complex diseases.
The large molecule segment is projected to grow at the fastest CAGR over the estimated period. Rising requirements for monoclonal antibodies, peptides, and gene therapies drive the segment growth. Additionally, the increasing outsourcing by pharmaceutical and biotech companies, rising investment in R&D for complex diseases, and the need for advanced and sensitive analytical techniques all contribute to segment growth. Moreover, stringent regulatory requirements, a focus on immunogenicity assessment, and the expansion of global biologics pipelines are expected to drive the segment's growth.
Test Insights
The bioavailability segment captured the largest revenue share of the preclinical bioanalytical testing services industry in 2024. The segment’s growth is primarily driven by its significant role in enhancing the need to evaluate absorption, distribution, metabolism, and excretion (ADME) profiles of new drug candidates early in development and during regulatory approval activities. The growing focus on optimizing drug delivery systems and enhancing oral bioavailability allows pharmaceutical and biotechnology companies to invest significantly in advanced analytical platforms such as LC-MS/MS and HPLC. Furthermore, rising investment in drug innovations and the increasing number of generic drug launches are expected to drive demand for bioavailability studies to establish therapeutic equivalence. Regulatory agencies, such as the U.S. FDA and EMA, have also mandated comprehensive bioavailability testing for new and reformulated drugs, strengthening the need for specialized analytical services. Such factors are expected to drive the market.
The bioequivalence segment is expected to grow at the fastest CAGR during the forecast period. This growth is attributed to the increasing demand for generic and follow-on drug products, which necessitate early-stage pharmacokinetic comparisons with reference drugs. The increasing outsourcing by pharmaceutical companies, stringent regulatory requirements, and the need for accurate and reproducible data in preclinical studies are anticipated to support the market's growth.
Workflow Insights
The sample analysis segment captured the largest revenue share of the preclinical bioanalytical testing services market in 2024. The segment growth is driven by increasing demand for accurate and rapid drug development, especially for biologics, biosimilars, and gene therapies. As drug pipelines grow more complex, the demand for precise and high-throughput sample analysis is expected to boost the segment growth. In addition, stringent regulatory requirements, the need for specialized CROs, reducing costs, accelerating timelines, and ensuring high-quality preclinical study outcomes contribute to segment growth.
The sample collection and preparation segment is expected to grow at the fastest CAGR during the forecast period. This growth is due to the increasing complexity of drug development processes and the rising need for efficient sample collection and preparation in preclinical bioanalytical testing to ensure accurate and reproducible results. Advancements in extraction and purification techniques are enhancing the efficiency and reliability of sample preparation, making it a critical component in preclinical bioanalytical workflows.
End Use Insights
The pharma & biopharma companies segment captured the largest revenue share of the preclinical bioanalytical testing services industry in 2024. The segment's growth is driven by the increasing demand for robust, high-quality analytical solutions that support drug development across the discovery, preclinical, and clinical stages. Some of the major service providers are expanding their capabilities and geographical presence to meet the client requirements. For instance, in August 2024, SGS introduced its new specialized preclinical bioanalytical testing services in Hudson, New Hampshire, North America. The company provides advanced bioanalytical services to biopharmaceutical and pharmaceutical companies. The new services, including discovery, preclinical research, and Phase 1-3 clinical trials, support the drug development spectrum. The capabilities will include method transfer, development, and validation, as well as pharmacodynamic (PD) and pharmacokinetic (PK) bioanalysis, immunogenicity testing, ELISA and multiplex assays, bioassays, and additional related analyses.

The CRO segment is expected to grow at the fastest CAGR in the coming years due to the increasing demand for preclinical bioanalytical testing, driven by the rising complexity of drug candidates, including biologics, biosimilars, and gene therapies. In addition, most CROs offer specialized expertise, regulatory compliance, and integrated services across preclinical and clinical phases, enabling companies to accelerate drug development while maintaining quality and precision. Moreover, stringent regulatory requirements from the FDA and EMA necessitate high-quality, reliable testing, contributing to segment growth.
Regional Insights
The North America preclinical bioanalytical testing services market accounted for the largest revenue share of 47.42% in 2024. A robust pharmaceutical and biopharmaceutical industry, strong R&D investment, and the presence of leading CROs drive the growth in the region. Additionally, the expansion of biologics, gene therapies, and novel drugs requires precise analytical testing, further supporting market growth. Moreover, growing innovations, rising demand for highly sensitive advanced analytical technologies, and increasing outsourcing trends enable companies to reduce costs, accelerate timelines, and leverage specialized expertise for preclinical studies. Such factors are expected to drive market growth.
U.S. Preclinical Bioanalytical Testing Services Market Trends
The preclinical bioanalytical testing services industry in the U.S. is driven by the expansion of contract research organizations, increased investment in biopharmaceutical research and development, a rising number of clinical trials, growing demand for personalized medicine, and stringent regulatory oversight by the FDA. The presence of top-tier CROs in the country further promotes the high-quality analytical standards. In addition, the growing demand for biopharmaceutical products and rising drug safety requirements have increased reliance on preclinical bioanalytical testing services, allowing companies to access cost-effective expertise. Such factors are expected to drive the market growth.
Europe Preclinical Bioanalytical Testing Services Market Trends
The preclinical bioanalytical testing services industry in Europe is experiencing growth owing to technological advancements, strong pharmaceutical and biotech R&D, and increasing development of biologics, gene therapies, and novel drugs. In addition, the country has stringent regulatory standards from the EMA for clinical research, further supporting the market. Besides, the region has become a hub for clinical research due to its diverse patient populations and well-established regulatory frameworks.
The preclinical bioanalytical testing services market in the UK held a significant share in 2024. The country's market growth is driven by a strong pharmaceutical and biotech sector, growing investment in R&D, and the development of biologics. Besides, increasing advancements in bioanalytical techniques further drive collaboration among CROs and academic institutions, along with government support for life sciences innovation. This accelerates the need for secure, compliant, and reliable bioanalytical testing services to meet quality standards and compliance requirements.
The Germany preclinical bioanalytical testing services market is anticipated to grow due to strong pharmaceutical and biotechnology sectors, advanced R&D infrastructure, highly skilled workforce, and stringent regulatory standards. In addition, the increasing prevalence of chronic diseases has led to a rising demand for biologics, biosimilars, and specialty drugs, further creating a growing need for reliable and compliant preclinical bioanalytical testing services.
The preclinical bioanalytical testing services market in France is driven by the expansion of biologics in the pharmaceutical industry, such as cell and gene therapies and biosimilars, which has intensified the need for outsourced services such as bioanalytical testing. According to an article published by NCBI in February 2024, 44 new cell and gene therapies are anticipated to enter the French market by 2030, and an estimated annual number of treated patients is expected to rise from 1,653 in 2023 to over 69,400 by 2030. These complex products require rigorous testing for safety, efficacy, and quality, further driving the demand for specialized preclinical bioanalytical services.
Asia Pacific Preclinical Bioanalytical Testing Services Market Trends
The Asia Pacific preclinical bioanalytical testing services industry is projected to grow at the highest CAGR over the forecast period. The market's growth is driven by increasing pharmaceutical and biotechnology activities, rising healthcare expenditures, lower service costs, adherence to evolving GDP standards, improved access to medicines, and a growing focus on regulatory compliance and technological advancements.
China's preclinical bioanalytical testing services market is expected to grow over the forecast period. China has become an increasingly attractive destination for outsourcing clinical trials, driven by the growing expansion of domestic and international pharmaceutical and biopharmaceutical manufacturing capabilities. Strong government initiatives aimed at improving healthcare accessibility and efficiency have led to a significant increase in the outsourcing of preclinical bioanalytical testing services operations.
The preclinical bioanalytical testing services market in Japan is expected to witness significant growth over the forecast period. The country's growth is driven by the rising prevalence of chronic diseases and lifestyle-related conditions, which has increased the demand for high-quality analytical services that comply with established regulatory frameworks aligned with PMDA and ICH guidelines. In addition, government policies aimed at improving healthcare access and the country's emphasis on quality and precision operations have fueled the market growth.
India preclinical bioanalytical testing services market is witnessing considerable growth, and the growth is attributed to the expansion of the biopharmaceutical and biotechnology sectors, growing clinical trial activities, and increasing demand for high-quality bioequivalence and bio-similarity studies. Such factors are expected to drive the market.
Latin America Preclinical Bioanalytical Testing Services Market Trends
The Latin America preclinical bioanalytical testing services industry is projected to grow over the forecast period. The growth in the region is driven by the increasing expansion of regional drug manufacturing and growing government initiatives aimed at enhancing access to essential medicines. Besides, the presence of major countries such as Brazil and Argentina is expected to drive high demand for drug discovery across the globe, leading to an increasing need for advanced analytical techniques to improve the accuracy and efficacy of testing.
Brazil's preclinical bioanalytical testing services market is expected to grow over the forecast period. The country's growth is driven by the developing pharmaceutical/biopharmaceutical & biotechnology industries, increasing investments in clinical research and drug development, and growing favorable regulatory requirements that attract international CROs, further strengthening the market capabilities.
Key Preclinical Bioanalytical Testing Services Company Insights
Key players in the preclinical bioanalytical testing services market are actively expanding their service portfolios, investing in advanced analytical technologies, and pursuing strategic partnerships to strengthen their global presence. Leading contract research organizations (CROs) and specialized laboratories are enhancing their capabilities in complex assay development, large molecule analysis, and cell and gene therapy testing to meet evolving client needs. For instance, in October 2024, SGS introduced its new specialized preclinical bioanalytical testing services in Hudson, New Hampshire, North America. Through these services, the company provides advanced bioanalytical services to both biopharmaceutical and pharmaceutical companies.
Key Preclinical Bioanalytical Testing Services Companies:
The following are the leading companies in the preclinical bioanalytical testing services market. These companies collectively hold the largest market share and dictate industry trends.
- Labcorp
- Eurofins Scientific SE
- SGS SA
- Charles River Laboratories International
- Syneos Health
- KCAS Bio
- ICON plc
- WuXi AppTec
- Thermo Fisher Scientific Inc (PPD, Inc.)
- Parexel International
- Pharmaron
- BioAgilytix Labs
- QPS Holdings
- Altasciences
Recent Developments
-
In November 2024, Bioneeds India joined the Contract Laboratory Network, offering pharmacology, toxicology, analytical, and regulatory services, supporting pharmaceutical, biotech, and medical device clients with GLP- and ISO-certified preclinical research capabilities.
-
In November 2024, Altasciences mentioned that it has enhanced its bioanalytical service portfolio, offering flexible, high-quality analytical solutions supporting preclinical and clinical studies, reinforcing its 30-year commitment to accelerating global drug development and client success.
-
In October 2023, CEBIS mentioned the offering of end-to-end solutions for clinical research from study start-up to clinical study reports across phases I to IV. Their services include preclinical testing, drug safety, biostatistics, clinical trial supply management, and software solutions such as IRT and EDC, all delivered with competitive efficiency and a strong focus on patient-centric outcomes.
Preclinical Bioanalytical Testing Services Market Report Scope
Report Attribute
Details
Market size in 2025
USD 1.25 billion
Revenue forecast in 2033
USD 2.62 billion
Growth rate
CAGR of 9.76% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Molecule, test, workflow, end use, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Labcorp; Eurofins Scientific SE; SGS SA; Charles River Laboratories International; Syneos Health; KCAS Bio; ICON plc; WuXi AppTec; Thermo Fisher Scientific Inc (PPD, Inc.); Parexel International; Pharmaron; BioAgilytix Labs; QPS Holdings; Altasciences
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Preclinical Bioanalytical Testing Services Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the preclinical bioanalytical testing services market report based on molecule, test, workflow, end use, and region:
-
Molecule Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecule
-
Large Molecule
-
-
Test Outlook (Revenue, USD Million, 2021 - 2033)
-
ADME
-
In-vivo
-
In-vitro
-
-
Pharmacokinetics (PK)
-
Pharmacodynamics (PD)
-
Bioavailability
-
Bioequivalence
-
Biomarker Testing
-
Cell-based Assay
-
Virology Testing
-
Other Tests
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Sample Collection and Preparation
-
Sample Collection, Handling and Storage
-
Protein Precipitation
-
Liquid-Liquid Extraction
-
Solid Phase Extraction
-
Others
-
-
Method Development and Validation
-
Sample Analysis
-
Hyphenated technique
-
Chromatographic technique
-
Electrophoresis
-
Ligand Binding Assay
-
Mass Spectrometry
-
Spectroscopic Techniques
-
Genomic and Molecular Techniques
-
-
Other processes
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharma & BioPharma Companies
-
CDMO
-
CRO
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










