- Home
- »
- Biotechnology
- »
-
Protein Stability Analysis Market Size, Industry Report, 2020-2026GVR Report cover
![Protein Stability Report]()
Protein Stability Analysis Market (2020 - 2026) Size, Share & Trends Analysis Report By Product (Instruments, Software), By Technique (DSC, DSF), By End Use (CROs), By Region, And Segment Forecasts
- Report ID: GVR-4-68038-998-2
- Number of Report Pages: 175
- Format: PDF
- Historical Range: 2016 - 2018
- Forecast Period: 2020 - 2026
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
The global protein stability analysis market size was valued at USD 985.8 million in 2019 and is expected to register a CAGR of 10.8% over the forecast period. Protein stability analysis is a crucial step during drug development, right from active pharmaceutical ingredient (API) development up to product validation. Key players are engaged in implementing these testing techniques for assessment of the quality and effectiveness of drug products under various environmental factors.
The development of new high-throughput techniques, such as Stable-seq by professionals at The American Society for Biochemistry and Molecular Biology, Inc., which involves genetic selection in combination with high-throughput DNA sequencing is anticipated to induce growth. This approach effectively evaluates the in vivo stability of protein variants, which is important for the identification of components in proteolytic pathways that affect the protein turnover and associated activities.
Outsourcing has been widely employed in the drug development industry as a business strategy to enhance productivity in drug development R&D activities and reduce drug development costs. Several contract research organizations, including 2bind GmbH and LakePharma, Inc., are offering stability analysis solutions with quality, accuracy, and robustness within expected timelines. Moreover, compliance with regulatory expectations supplements the adoption of outsourced services by pharmaceutical companies.
Constant efforts laid down by the research groups to design software tools for analysis of protein stability are expected to drive market growth. CUPSAT, designed by the University of Cologne, is an easily accessible web tool for protein stability analysis based on point mutations. This prediction model serves as a valuable tool for the analysis, with an accuracy greater than 80%.
The use of thermal proteome profiling (TPP), provides a novel approach for protein stability analysis. TPP is based on the principle that proteins develop a high resistance for heat-induced unfolding by ligand binding. Hence, the improvements in instrumentation and sample preparation for TPP enables the identification of even small stability shifts in proteins.
Product Insights
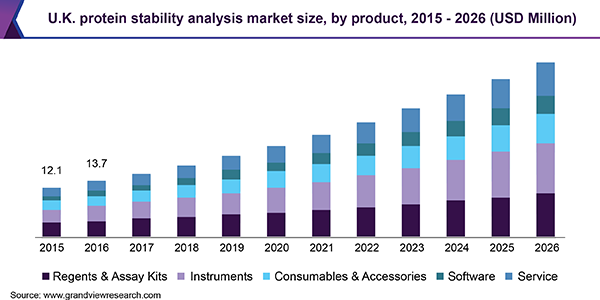
The reagents and assay kits segment dominated the protein stability analysis market in 2019 and is expected to maintain its dominance throughout the forecast period. The implementation of reagents in combination with software enables inexpensive, efficient, and rapid high throughput screening of thermal stability of protein. It aids in the library screening for the identification of ligands that bind to the protein of interest.
Companies such as Thermo Fisher are providing a combination of reagents with software that also allows the identification of mutations and modifications that affects the protein’s stability. Moreover, low pricing, wide applicability, and easy availability of reagents contribute to the largest share of the segment. On the other hand, the instruments segment is expected to witness the fastest growth rate during the study period.
This is because these automated systems deliver highly sensitive protein stability analysis with minimal sample consumption in a lesser time frame. Constant developments in instrument design and incorporation of new features improve productivity during biopharmaceutical discovery and development and generate high quality, reproducible, and reliable protein denaturation data.
Technique Insights
Differential Scanning Calorimetry (DSC) occupied the largest revenue share in the market for protein stability analysis in 2019. The DSC is regarded as a standard stability-indicating technique as it requires minimal assay development. Moreover, due to its high accuracy and reproducibility, DSC is widely utilized in characterizing and selecting suitable proteins for drug development and ligand interaction studies. The other advantages associated include label-free and direct measurement.
Ongoing research studies on DSC usage in protein stability analysis have attributed to the projected revenue share. For instance, a study published in July 2019, evaluated the protein refolding and its complete thermal denaturation by using DSC. Similar kind of study implemented DSC for the measurement of denaturation and solidification of native silk proteins and concluded that silk possessed lower denaturation enthalpy as compared to albumin. Rising application of these techniques in the research arena is anticipated to support the growth in the coming years.
Label-free Differential Scanning Fluorimetry (DSF) is a relatively new technique and exhibits strong growth potential in the near future. This method has gained immense popularity in recent years because it is suitable for analyzing minuscule protein quantities; it is easy to perform, and it can be conducted by using intrinsic tyrosine and tryptophan fluorescence.
End-use Insights
Pharmaceutical and biotechnology companies captured the largest revenue share due to the availability of numerous protein therapeutics products in the market for protein stability analysis and a large number of therapeutics present in preclinical and clinical development stages. Recent approvals of recombinant DNA and monoclonal antibody technologies also provide lucrative opportunities for new therapeutics development.
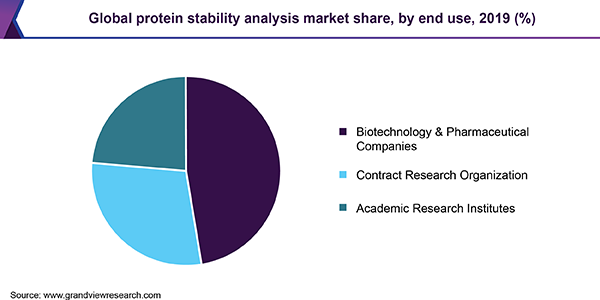
The expansion of the contract research service portfolio has contributed to making protein characterization more efficient and affordable. Contract research organizations (CROs) possess advanced technology and instrumentation capabilities to support the research of pharmaceutical firms through all phases of drug development.
CROs are considered as a viable solution to address the capital and resource challenges faced by emerging and small-sized pharmaceutical firms that face budget constraints. They provide a range of analytical testing techniques to support the characterization and development of recombinant peptide and protein therapeutics developed by these firms.
Regional Insights
North America accounted for the largest share in the global market for protein stability analysis in 2019. Several initiatives undertaken by the public and private entities and funding programs initiated by them in the proteomics industry have contributed to the largest share. Grants provided by the National Institute of Health (NIH), National Center for Research Resources, US HUPO, and other government entities accelerate the proteomic research activities in this region.
For instance, since 2012 the NIH’s Disruptive Proteomics Technologies (DPT) Working Group has focused on the identification of gaps and opportunities in technologies and methodologies pertaining to protein analysis. This approach of NIH enables substantial advancement in protein stability analysis across a wide range of biomedical research areas.
Asia Pacific is projected to witness the fastest growth rate in the near future. This is attributive to the efforts undertaken by the CROs to expand their customer base by establishing relationships with the pharmaceutical and biotechnology firms in Asia. Moreover, the increasing number of clinical trials in Asia has also enhanced the adoption of proteomics services in Asia Pacific.
Key Companies & Market Share Insights
Key participants operating in the market for protein stability analysis include Thermo Fisher Scientific, Inc; Enzo Biochem, Inc.; PerkinElmer Inc.; NanoTemper; GE Healthcare; HORIBA, Ltd.; Malvern Panalytical Ltd.; Agilent Technologies, Inc.; SETARAM Instrumentation; Unchained Labs; Waters Corporation; and Applied Photophysics Ltd.
Key companies are adopting significant initiatives to reinforce their market presence and maintain a competitive edge in the space. In addition, market participants are involved in product development, collaborative and partnership models, geographic expansion, and agreement strategies in untapped regions.
For instance, in February 2019, NanoTemper Technologies introduced NT. Robotic autosampler, which is designed to automate the researcher’s workflow for screening a wide variety of candidates for protein stability. This new system benefited the company’s protein stability analysis portfolio due to high flexibility and small size, thereby accelerating the company’s sales.
Protein Stability Analysis Market Report Scope
Report Attribute
Details
Market size value in 2020
USD 1.10 billion
Revenue forecast in 2026
USD 2.0 billion
Growth Rate
CAGR of 10.8% from 2020 to 2026
Base year for estimation
2019
Historical data
2015 - 2018
Forecast period
2020 - 2026
Quantitative units
Revenue in USD Million and CAGR from 2020 to 2026
Report coverage
Revenue forecast; company share; competitive landscape; growth factors and trends
Segments covered
Product, technique, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; U.K.; China; Japan; Brazil; South Africa
Key companies profiled
Thermo Fisher Scientific, Inc; Enzo Biochem, Inc.; PerkinElmer Inc.; NanoTemper; GE Healthcare; HORIBA, Ltd.; Malvern Panalytical Ltd.; Agilent Technologies, Inc.; SETARAM Instrumentation; Unchained Labs; Waters Corporation; Applied Photophysics Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country; regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Segments Covered in the ReportThis report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2015 to 2026. For the purpose of this study, Grand View Research has segmented the global protein stability analysis market report on the basis of product, technique, end use, and region:
-
Product Outlook (Revenue, USD Million, 2015 - 2026)
-
Reagents and Assay Kits
-
Instruments
-
Consumables and Accessories
-
Software
-
Service
-
-
Technique Outlook (Revenue, USD Million, 2015 - 2026)
-
Chromatography
-
Spectroscopy
-
Differential Scanning Calorimetry (DSC)
-
Differential Scanning Fluorimetry (DSF)
-
Dynamic Light Scattering (DLS)
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2015 - 2026)
-
Pharmaceutical & Biotechnology Companies
-
Contract Research Organizations
-
Academic Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2015 - 2026)
-
North America
-
The U.S.
-
Canada
-
-
Europe
-
Germany
-
The U.K.
-
-
Asia Pacific
-
China
-
Japan
-
-
Latin America
-
Brazil
-
-
Middle East and Africa (MEA)
-
South Africa
-
-
Frequently Asked Questions About This Report
b. The global protein stability analysis market size was estimated at USD 0.99 billion in 2019 and is expected to reach USD 1.10 billion in 2020.
b. The global protein stability analysis market is expected to grow at a compound annual growth rate of 10.8% from 2020 to 2026 to reach USD 2.0 billion by 2026.
b. Differential Scanning Calorimetry (DSC) dominated the protein stability analysis with a share of 20.4% in 2019. The DSC is regarded as a standard stability-indicating technique as it requires minimal assay development.
b. Some key players operating in the protein stability analysis market Thermo Fisher Scientific, Inc; Enzo Biochem, Inc.; PerkinElmer Inc.; NanoTemper; GE Healthcare; HORIBA, Ltd.; Malvern Panalytical Ltd.; Agilent Technologies, Inc.; SETARAM Instrumentation; Unchained Labs; Waters Corporation; and Applied Photophysics Ltd.
b. Key factors that are driving the market growth include the rising number of recombinant protein therapeutics, the presence of supportive government initiatives to encourage drug safety programs, expansion of outsourcing activities for protein analysis, and an increase in R&D expenditure for drug development.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










