- Home
- »
- Healthcare IT
- »
-
Real World Evidence Solutions Market, Industry Report 2033GVR Report cover
![Real World Evidence Solutions Market Size, Share & Trends Report]()
Real World Evidence Solutions Market (2026 - 2033) Size, Share & Trends Analysis Report By Component (Services, Data Sets), By Therapeutic Area (Oncology, Cardiology), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68039-808-0
- Number of Report Pages: 190
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Real World Evidence Solutions Market Summary
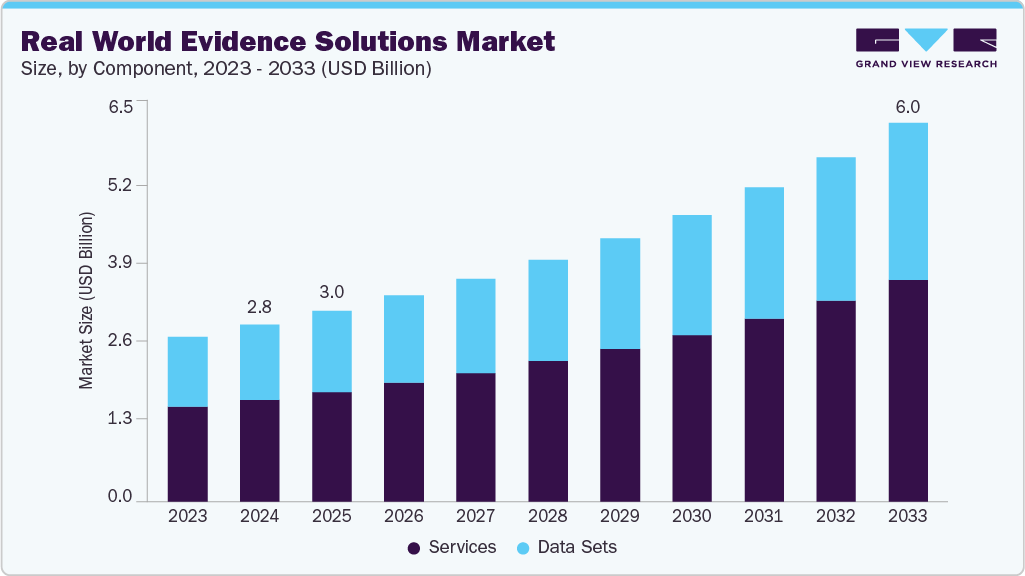
The global real world evidence solutions market size was estimated at USD 3.04 billion in 2025 and is projected to reach USD 6.04 billion by 2033, growing at a CAGR of 9.08% from 2026 to 2033. This growth is attributed to the increase in R&D spending, rising applications of real-world evidence (RWE) in various fields, support from regulatory bodies for using RWE solutions, and the growing volume of real-world data, which are anticipated to boost the market growth.
Key Market Trends & Insights
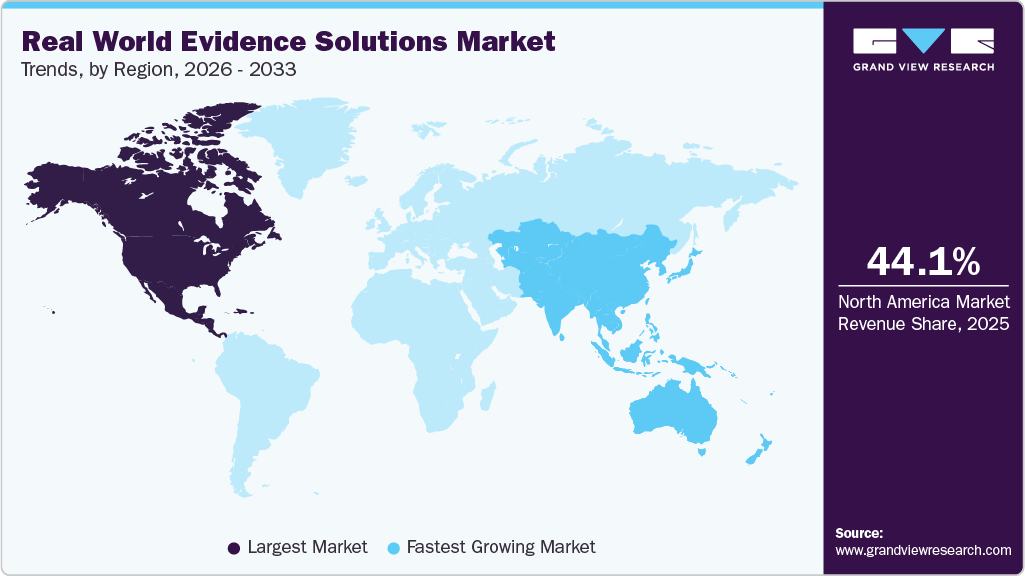
- North America dominated the market for real world evidence solutions with a share of 44.09% in 2025.
- Asia Pacific is estimated to be the fastest-growing region over the forecast period.
- Based on component, the services segment held the largest market share of 58.14% in 2025.
- By application, the drug development and approvals segment dominated the market with the largest revenue share in 2025.
- Based on end use, healthcare companies held the dominant market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.04 Billion
- 2033 Projected Market Size: USD 6.04 Billion
- CAGR (2026-2033): 9.08%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Regulatory bodies worldwide are increasingly recognizing the value of real-world evidence to supplement clinical trial data, accelerate drug approvals, and enhance patient safety monitoring. This support reduces barriers for pharmaceutical and biotech companies to adopt RWE, thereby driving market growth. Strong regulatory frameworks encourage the collection, analysis, and use of real-world data in decision-making, making RWE a standard part of healthcare research, drug development, and post-market surveillance. To support wider adoption, regulatory bodies are introducing policies and guidance that actively facilitate the practical use of RWE in regulatory submissions and post-market evaluations. For instance,-
In December 2025, the U.S. Food and Drug Administration removed a longstanding requirement that real-world evidence submitted in drug and device applications include identifiable individual patient data, allowing de-identified datasets such as registries, claims, and EHR networks to be considered on a case-by-case basis in regulatory reviews.
-
In January 2025, the UK’s Medicines and Healthcare Products Regulatory Agency launched its pilot Real-World Evidence (RWE) Scientific Dialogue Programme as part of its 2024-27 Data Strategy to help innovators refine evidence-generation strategies and clarify regulatory and health-technology assessment expectations for RWE. The pilot offers confidential discussions and a “safe harbour” workshop, aiming to improve RWE methodologies and support regulatory and HTA decision-making across diverse products and disease areas.
Such regulatory efforts are strengthening the industry’s trust in real-world evidence approaches, thereby driving broader global uptake of RWE solutions.
The growing volume of real-world data is a key market driver, as the volume of health-related data continues to grow at a compound annual rate of 36%. Data from electronic health records, insurance claims, disease registries, wearables, and patient-reported outcomes is increasing in both scale and granularity. Advances in data integration, interoperability, and analytics enable RWE platforms to transform these vast datasets into meaningful, actionable insights. This allows pharmaceutical companies, payers, and healthcare providers to generate more accurate, representative, and timely evidence, supporting drug development, regulatory submissions, and value-based care initiatives.
AI in Real World Evidence Solutions Industry
AI-enabled analytics and multi-source data integration platforms help overcome interoperability challenges by automating data mapping, cleaning, and standardization processes. These advanced solutions enable seamless harmonization of diverse datasets, transforming fragmented data into unified, analysis-ready formats. As a result, organizations can accelerate evidence generation and support informed clinical, regulatory, and commercial decision-making. To address these challenges, several leading players are embedding AI and automation capabilities within their RWE solutions, as illustrated below:
Vendor
Real-World Evidence Capabilities
AI / Automation Capabilities
Saama
RWE analytics, clinical data integration, and insights generation
AI/ML-driven data curation, predictive analytics, automated reporting
SAS Institute Inc.
RWE data analysis and modeling for clinical and commercial insights
Advanced analytics, AI-powered predictive modeling, automated data visualization
Veradigm LLC
Real-world data aggregation and patient insights
AI/ML for patient stratification, trend analysis, and automated outcome reporting
IQVIA
RWE analytics and predictive modeling for clinical and commercial decision-making
AI-driven predictive algorithms, data integration, automated insights generation
These companies demonstrate how AI-powered data integration and automation tools are being leveraged to streamline interoperability, improve analytical efficiency, and enhance the overall quality and usability of real-world evidence across the healthcare value chain.
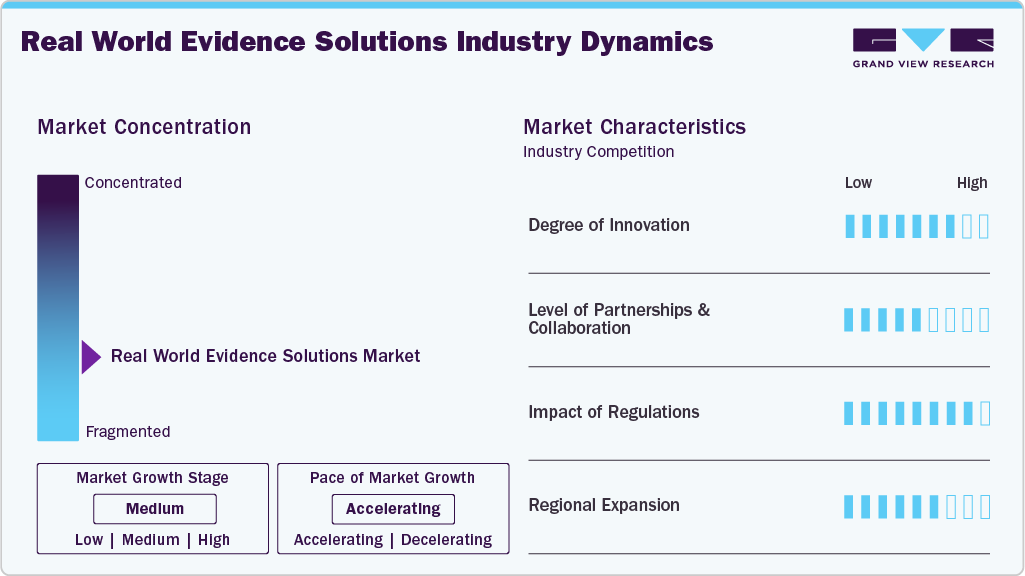
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion. For instance, the real-world evidence market is slightly fragmented, with many product & service providers entering the market. The degree of innovation, the level of partnerships & collaboration activities, and the impact of regulations on the industry are high. However, the regional expansion observes moderate growth.
The degree of innovation in the patient access solutions market is high, driven by the need to reduce claim denials, streamline workflows, and enhance patient engagement. Vendors are rolling out AI-powered platforms that automate eligibility checks, prior authorizations, and digital scheduling, helping providers cut delays and improve accuracy. For instance, in October 2025, Atropos Health launched its new AI-driven platform, the Atropos Evidence Agent, at Stanford Health Care and is collaborating with Microsoft to integrate real-world evidence directly into clinical workflows via ambient AI and EHR systems.
“Utilizing agentic AI to embed real-world evidence (RWE) into workflows with patient-specific insights will enable providers to proactively address patient questions with speed and accuracy. This example shows how ambient AI, ChatEHR, and agentic AI are putting technology to work for the benefit of both providers and patients.”
- Dr. Michael Pfeffer, Chief Information and Digital Officer at Stanford Medicine.
The level of partnerships and collaborations in the Real-World Evidence (RWE) solutions market is moderate, aimed at enhancing data access, integration, and actionable insights. Vendors collaborate with healthcare providers, life-science companies, and technology partners to expand interoperability, incorporate AI-driven analytics, and streamline real-world data generation and utilization across clinical and commercial decision-making. For instance, in October 2025, HealthVerity and Claritas Rx announced a strategic partnership combining HealthVerity’s large-scale, de‑identified real‑world data (claims, EMR, lab) with Claritas Rx’s specialty pharmacy, hub, and co‑pay program insights. The collaboration aims to provide pharma commercial, RWE, and HEOR teams with an integrated dataset offering deeper visibility into treatment access, patient journeys, and outcomes.
The impact of regulations on the real world evidence solutions industry is significant, as clear regulatory guidance from agencies like the FDA, EMA, MHRA, and PMDA establishes standards for data quality, study design, and methodological rigor. This reduces uncertainty for life-science companies, encouraging wider adoption of RWE platforms to support drug development, regulatory submissions, post-market surveillance, and value-based care.
Regional expansion is driving growth in the real world evidence solutions industry as vendors extend their presence across North America, Europe, and Asia-Pacific to tap into rising healthcare digitization, regulatory support, and demand for data-driven insights. Expanding into new geographies allows providers, pharma, and payers to access localized real-world datasets. For instance, in March 2025, OM1 is expanding into Europe to bring its AI-powered real-world evidence (RWE) solutions to life sciences firms, helping them generate high-quality, observational data to support drug development, market access, and regulatory needs.
Component Insights
Based on components, the services segment dominated the market with the largest revenue share of 58.14% in 2025. The segment growth is attributed to the high adoption of real world services by pharmaceutical and biotechnology companies and healthcare providers. Moreover, business initiatives undertaken by market players are among the key factors driving market growth. For instance, in February 2024, Gilead Sciences, Inc. announced its plans to present RWE and new clinical data from its antiviral research and development programs at the 31st Conference on Retroviruses and Opportunistic Infections (CROI 2024)
However, the data sets segment is expected to grow at a significant rate during the forecast period. Real-World Data (RWD) is data collected outside the context of clinical trials from sources such as Electronic Medical Records (EMRs), insurance claims, patient-reported results, and biometric devices. Growth in this segment is primarily due to the increasing need for further understanding of real-world compliance, epidemiology, costs, large amounts of medical data generated in hospitals, and the reliance on results-based research.
Application Insights
On the basis of application, the drug development and approvals segment accounted for the largest revenue share of 28.74% in 2025. Real world evidence solutions allow pharmaceutical companies, healthcare providers, and payers to efficiently manage operations and accelerate the process of drug development and its approval. This fuels market growth. Furthermore, governments are undertaking various initiatives to promote the use of RWE in medical device development and approvals. For instance, in December 2023, the FDA published draft guidance outlining its assessment of RWD in medical device clearance or approval decisions, along with anonymized examples of its RWE utilization.
The reimbursement/coverage and regulatory decision-making segment is expected to grow at the fastest CAGR during the forecast period. Most nations in the recent past have leveraged RWE for conducting pharmacovigilance activities and post-marketing effectiveness assessments; however, RWE frameworks that address the usage of RWE during the product lifecycle, comprising pre-marketing activities & post-marketing labeling changes for effectiveness, have only recently been issued.
Therapeutic Insights
On the basis of therapeutic area, the oncology segment held the largest market share of 23.10% in 2025. This growth is attributed to several applications of RWE in cancer drug development processes, such as decreasing the cost of clinical trials, improving the probability of regulatory success, and increasing product approval rates in a shorter timeline. According to GLOBOCAN, in 2022, approximately 10.0 million new cancer cases were detected, and around 9.7 million cancer-related deaths were recorded globally. According to the Indian Council of Medical Research (ICMR), the incidence of cancer in the country increased from 1.39 million cases in 2020 to 1.46 million cases in 2022. Therefore, the growing demand for effective drug development in the oncology therapeutic space has further increased the deployment rate of RWE solutions to expedite the pace of drug discovery.
The cardiology segment is expected to grow at a significant CAGR during the forecast period. RWE solutions offer a potentially rich source of information from clinical trial experiments conducted at different locations and set-ups. In the therapeutic area of cardiology, RWE solution correlates with the evaluation of the potential risks or benefits of an intended drug, using real-world patient data from several other sources. This information helps researchers in clinical decisions revolving around a diverse patient population encountered in the comparison studies. The benefits of RWE in cardiology have been evaluated by the American College of Cardiology Foundation (ACCF), which indicated that about 64% of U.S. cardiologists were very interested in the educational programs regarding RWE software, considering its varied applications.
End Use Insights
On the basis of end use, the healthcare companies segment accounted for the largest revenue share in 2025. The growth can be attributed to the rising significance of RWE studies in drug approvals, the necessity to avoid expensive drug recalls, and the increasing need to evaluate drug performance in real-world settings. For instance, in February 2024, PINC AI Applied Sciences (PAS), Premier, Inc.'s division, and Datavant expanded their collaboration to enhance healthcare research, improve clinical trial operations, and promote trial diversity & equity.
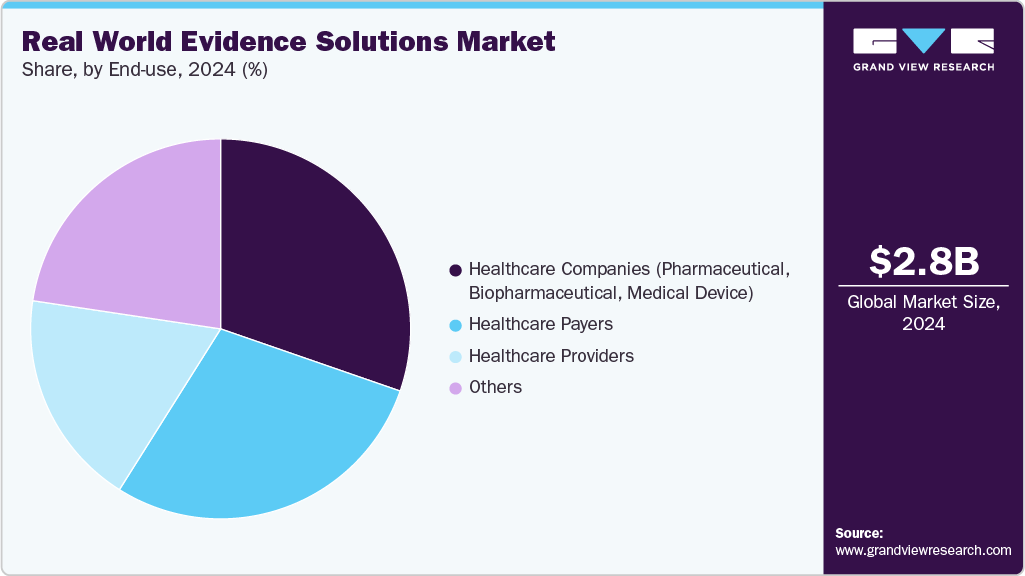
The healthcare payers’ segment is expected to grow at the fastest CAGR during the forecast period due to increasing awareness among payers regarding the importance of medical device/drug safety & their adverse effects and a favorable reimbursement scenario, especially in developed countries. RWE has been used in nearly 16% of all the clinical findings cited by payers in specialty drug decisions, with HER data being the most often cited data set used to derive these insights. RWE solutions help payers in reimbursement assessments. RWE also supports patients in budget management and assessing the risk-benefit of interventions. The incorporation of RWE in value evaluation frameworks, used for determining drug value, can assist payers in making informed, evidence-based reimbursement assessments.
Regional Insights
North America accounted for the largest share of the global real world evidence solutions market in 2025, which can be attributed to significant support from regulatory bodies for using Real-world Evidence (RWE) solutions, and an increase in R&D spending is also anticipated to boost market growth. Furthermore, the shift from volume to value-based care is expected to fuel market growth. In addition, the high share of the region can be attributed to the presence of key players in the U.S. and Canada. Favorable government regulations and an increasing number of RWE service providers in the region are anticipated to drive the market further.
U.S. Real World Evidence Solutions Market Trends
The real world evidence solutions market in the U.S. is growing rapidly as companies leverage advanced digital platforms and AI-driven tools to generate actionable insights from clinical and real-world data. For instance, in April 2025, Scientist.com launched Clinical Labs Navigator, a digital platform that streamlines the sourcing, management, and execution of clinical trial services, improving efficiency, transparency, and compliance across the research lifecycle. Similarly, in May 2024, Atropos Health raised USD 33 million to scale its AI-powered evidence automation platform, enabling faster production of high-quality, personalized clinical insights and supporting broader adoption across healthcare institutions, life sciences, and research organizations. These initiatives highlight the U.S. market’s focus on operational efficiency, advanced analytics, and accelerated evidence generation to drive better clinical and policy decisions.
Europe Real World Evidence Solutions Market Trends
The real world evidence solutions market in Europe is driven by increasing regulatory emphasis on incorporating real-world data into healthcare decision-making, reimbursement assessments, and post-market surveillance. Regional authorities and health technology assessment (HTA) bodies are actively promoting structured frameworks to improve the reliability and transparency of RWE use across member states. For instance, in July 2025, the Innovative Health Initiative launched the GREG project to strengthen the impact of RWE across Europe. The initiative focuses on developing practical guidance and tools to support regulators and HTA bodies in evaluating medicines and medical devices using real-world data.
The UK real world evidence solutions market is expected to grow significantly during the forecast period. The increasing government support in the form of initiatives aimed at promoting the adoption of RWE is driving market growth in the country. For instance, in October 2022, the National Institute for Health and Care Excellence (NICE) conducted a virtual workshop as a part of the IMI-funded project, the European Health Data and Evidence Network (EHDEN), in collaboration with the GetReal Institute. The initiative aims to establish a network of healthcare databases across Europe, each adhering to the Observational Medical Outcomes Partnership Common Data Model (OMOP CDM).
The real world evidence solutions market in Germanycan be attributed to the increasing partnerships among market players. In November 2023, Cegedim Health Data, a supplier of clinical RWE and RWD, expanded its European database, THIN, to incorporate German RWD. With data from the UK, France, Spain, Italy, Belgium, and Romania, the database now integrates German electronic health records. This expansion makes the database accessible to healthcare stakeholders, such as academic researchers and health authorities.
Asia Pacific Real World Evidence Solutions Market Trends
The real world evidence solutions market in the Asia Pacific is anticipated to register the fastest growth over the forecast period, owing to the rising government initiatives for the adoption of RWE studies and the presence of many contract research and manufacturing organizations in countries such as China and India. The rising demand for better healthcare services is also expected to fuel market growth. Moreover, major players are contributing to the region's market growth by entering into partnerships or expanding their product portfolios. These include Medpace; Cegedim Health Data; Parexel International Corporation; IQVIA, Inc.; and IBM.
Japan real world evidence solutions market has a significant market share in 2025. The market is expected to be driven by the increasing adoption of RWE solutions by local pharmaceutical companies. Key players in the market are developing devices that integrate RWD to deliver patient-centric care. For instance, in February 2024, ZimVie, Inc., a global player in the life sciences sector specializing in dental and spine markets, introduced the TSX Implant in Japan. This implant integrates design elements supported by over 20 years of clinical and RWD, aiming to provide benefits such as peri-implant health, sustained osseointegration, crestal bone preservation, and prosthetic stability.
The real world evidence solutions market in India is expected to grow significantly during the forecast period. Increasing government initiatives to digitize healthcare infrastructure are expected to drive the market. For instance, the National Digital Health Mission seeks to build a unified digital healthcare infrastructure that links healthcare practitioners to patients, allowing them to access real-time health records. This would enable healthcare providers to trace patients' medical histories and provide quality care.
Latin America Real World Evidence Solutions Market Trends
The real world evidence solutions market in Latin America is gradually gaining traction, fueled by growing interest from health technology assessment bodies and life-science firms to use locally generated RWD for decision-making. While public databases from Brazil, Argentina, Colombia, and Chile are expanding into claims and registry data, fragmented data governance and a lack of formal RWE guidelines remain barriers.
Middle East & Africa Real World Evidence Solutions Market Trends
The real world evidence solutions market in the Middle East & Africa region is increasingly exploring RWE solutions as governments and healthcare systems in countries like Saudi Arabia, the UAE, and Israel invest in digital health and data infrastructure to support value-based care and informed decision-making. In August 2025, the Saudi Food and Drug Authority (SFDA) released a draft framework outlining how real-world data (RWD) and RWE can support marketing authorization of medicines, emphasizing “fit-for-purpose” data, methodological quality, and use cases such as external comparators and evidence transportability. While RWE’s role in reimbursement and clinical policy is gaining recognition, challenges remain, including fragmented health systems, limited local data generation, and trust in data.
Key Real World Evidence Solutions Company Insights
Key players in the market are leveraging innovative strategies by offering AI-powered data analytics, predictive modeling, patient stratification tools, and automated outcomes reporting to enhance decision-making and research efficiency. Integration with interoperable healthcare IT systems, electronic health records, claims databases, and partnerships with biopharma companies, payers, and research organizations are further driving adoption and expanding the reach of RWE solutions.
Real-World Evidence Company Market Share Analysis Insights, 2025
The RWE market is led by integrated data and analytics leaders such as IQVIA, with strong competition from technology and health data firms, including Oracle Corporation and Optum. Established players like Merative, Flatiron Health, and SAS Institute, along with CROs such as ICON plc and Parexel, strengthen the ecosystem through analytics platforms and RWE services. The market remains fragmented, with multiple mid-sized firms and niche data providers driving innovation and competition.
No
Company
Estimated
Company Market
Shares 2025
(%)
Logic / Assumption
1
IQVIA
(Note: Detailed analysis around the market share and insights will be provided in the final deliverable.)
2
Oracle
3
Optum
4
Merative (ex-IBM Watson Health)
5
Flatiron Health
6
SAS Institute
7
ICON plc
8
Syneos Health
9
Parexel
10
Thermo Fisher Scientific
11s
Cognizant
12
Other Players
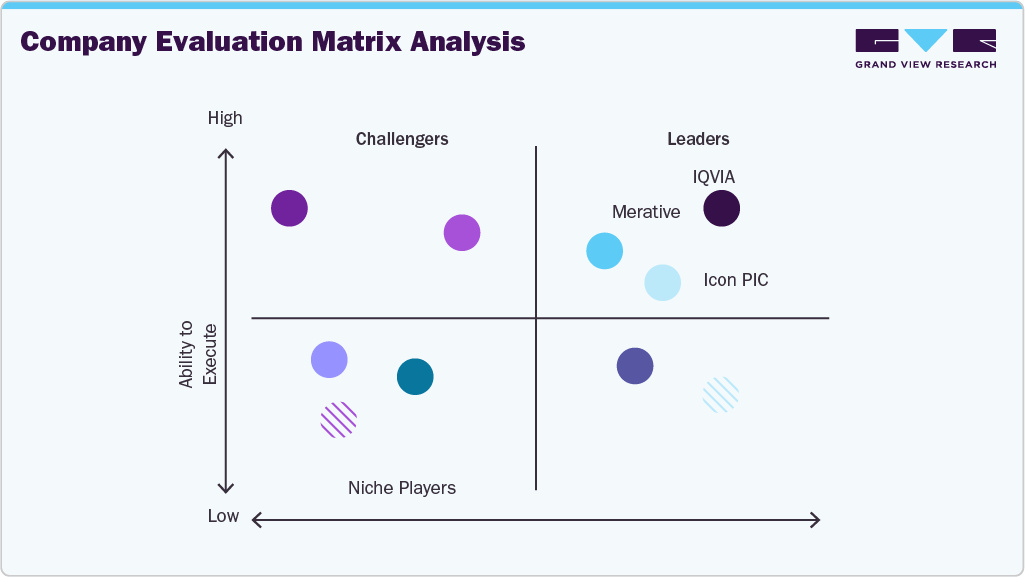
Key Real World Evidence Solutions Companies:
The following key companies have been profiled for this study on the real world evidence solutions market.
- IQVIA
- Merative
- PPD Inc. (now part of Thermo Fisher)
- Parexel International Corporation
- NTT DATA, Inc.
- Icon Plc
- Oracle
- Syneos Health
- Cegedim Health Data
- Medpace
- Optum Inc. (UnitedHealth Group)
- SAS Institute Inc.
- Cognizant
- Aetion, Inc. (acquired by Datavant in May 2025)
- Flatiron Health
- Cytel Inc.
- Trinity
Recent Developments
-
In January 2026, Thermo Fisher Scientific announced the launch of the CoreVitas Obesity Registry, a real‑world evidence initiative designed to collect and analyze comprehensive longitudinal data on patients living with obesity to better understand treatment patterns and outcomes. The registry aims to support healthcare decision‑making, clinical research, and improved patient care through high‑quality real‑world insights.
-
In November 2025, Datavant and AWS launched Datavant Connect powered by AWS Clean Rooms, a cloud-first solution that enables life-science companies to securely discover and analyze fit-for-purpose real-world data across multiple sources without moving raw data. This joint platform, validated by four top-20 pharma firms and 15 major RWD sources, streamlines data discovery and accelerates evidence generation, while preserving privacy.
“Life sciences leaders are under pressure to deliver insights faster, but the tools to do so have historically been slow, fragmented, and hard to scale. Cloud-first insights generation is now transforming how pharmaceutical organizations source fit-for-purpose data outputs more efficiently, accelerating how our industry approaches end-to-end evidence generation.”
-Arnaub Chatterjee, GM and President of Life Sciences at Datavant.
- In October 2025, LyfeSci Research & Innovation, a new physician-led CRO, launched with a strong emphasis on outcomes-focused research, offering integrated services from pre-clinical through to real-world data (RWD) and real-world evidence (RWE) studies. Their patient-and-site-centric model aims to streamline operations, enhance study quality, and accelerate insights that matter to both sponsors and healthcare providers.
“Our goal is to revolutionize the clinical research experience by placing patients at the heart of everything we do. By delivering value that reflects site needs-which in turn address patient priorities and clinical realities-we aim to improve study timelines and elevate the quality of research outcomes by leveraging our scientific expertise and technology capabilities.”
- Tapan Parikh, MD, CEO and Founder.
-
In March 2025, Aetion made its Aetion Evidence Platform available on AWS Marketplace, enabling life‑science, payer, and regulatory organisations to more easily access its cloud‑based, analytics‑driven real‑world‑evidence solution. The integration simplifies procurement, enhances scalability and compliance (with HIPAA, GDPR, and 21 CFR Part 11), and accelerates insights derived from real‑world‑data (RWD).
"By integrating with AWS, we offer our new and existing customers a seamless path to deploy and scale real-world evidence studies with enhanced efficiency and security. This is the first step in our vision to expand our collaboration with AWS to bring more value to our customers. Ultimately, our goal is to deliver an end-to-end real-world evidence platform that fully integrates with the customer's AWS-owned instances."
-Jay Podence, senior vice president of Commercial Partnerships at Aetion
-
In April 2024, IQVIA announced an expansion of its strategic partnership with Salesforce, aiming to expedite the advancement of the Life Sciences Cloud of Salesforce. This initiative represents a next-gen customer interaction platform for the life sciences sector. Through these strategic partnerships, IQVIA aims to advance the development of technological solutions designed to accelerate decision-making processes across various domains, including RWE, discovery, clinical development, medical affairs, and patient safety.
-
In April 2024, Parexel partnered with Palantir Technologies Inc. to leverage AI for accelerating & enhancing the delivery of effective and safe clinical trials for global biopharmaceutical clients. The partnership facilitates enhanced clinical trial processes and expands the capabilities of Paraxel in advanced analytics, RWE, and Health Outcomes.
Real World Evidence Solutions Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.28 billion
Revenue forecast in 2033
USD 6.04 billion
Growth rate
CAGR of 9.08% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market value in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Component, application, end use, therapeutic area, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Israel; Kuwait
Key companies profiled
IQVIA; Merative; PPD Inc. (now part of Thermo Fisher); Parexel International Corporation; NTT DATA, Inc.; Icon Plc; Oracle; Syneos Health; Cegedim Health Data; Medpace; Optum Inc. (UnitedHealth Group), SAS Institute Inc.; Cognizant; Aetion, Inc. (acquired by Datavant in May 2025); Flatiron Health; Cytel Inc.; Trinity
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Real World Evidence Solutions Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global real world evidence solutions market report based on component, application, end use, therapeutic area, and region:
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Services
-
Data Sets
-
Clinical Settings Data
-
Claims Data
-
Pharmacy Data
-
Patient-Powered Data
-
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Development & Approvals
-
Medical Device Development & Approvals
-
Reimbursement/Coverage and Regulatory Decision Making
-
Post Market Safety & Adverse Events Monitoring
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Healthcare Companies (Pharmaceutical, Biopharmaceutical, Medical Device)
-
Clinical research
-
Commercial (inclusive of marketing, etc.)
-
HEOR
-
Others (market access, etc.)
-
-
Healthcare Payers
-
Healthcare Providers
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Cardiology
-
Neurology
-
Diabetes
-
Psychiatry
-
Respiratory
-
Other Therapeutic Areas (Immunology, Gastroenterology, etc.)
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
Israel
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Key factors driving RWE solutions market include rising applications of real-world evidence (RWE) in various fields, support from regulatory bodies for using real world evidence (RWE) solutions and growing volume of real-world data are anticipated to boost the market growth.
b. The global real world evidence solutions market size was estimated at USD 3.04 billion in 2025 and is expected to reach USD 3.29 billion in 2026.
b. Some key players operating in the real world evidence solutions market include IQVIA; Merative; PPD Inc. (now part of Thermo Fisher); Parexel International Corporation; NTT DATA, Inc.; Icon Plc; Oracle; Syneos Health; Cegedim Health Data; Medpace; Optum Inc. (UnitedHealth Group); SAS Institute Inc.; Cognizant; Aetion, Inc. (acquired by Datavant in May 2025); Flatiron Health; Cytel Inc.; Trinity
b. The global real world evidence solutions market is expected to grow at a compound annual growth rate of 9.08% from 2026to 2033 to reach USD 6.04 billion by 2033.
b. North America dominated the RWE solutions market with a share of over 44.09% in 2025. This can be attributed to significant support from regulatory bodies for using Real-world Evidence (RWE) solutions, and an increase in R&D spending is also anticipated to boost market growth
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










