- Home
- »
- Medical Devices
- »
-
U.S. Cath Labs Market Size & Share, Industry Report, 2033GVR Report cover
![U.S. Cath Labs Market Size, Share & Trends Report]()
U.S. Cath Labs Market (2026 - 2033) Size, Share & Trends Analysis Report By Component (Services, Equipment, Medical Devices), By Facility Type (Independent, Hospital-based), Key Companies And Competitive Analysis And Segment Forecasts
- Report ID: GVR-4-68039-160-4
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Cath Labs Market Summary
The U.S. cath labs market size was estimated at USD 16.8 billion in 2025 and is projected to reach USD 17.7 billion in 2033, growingat a CAGR of 5.5% from 2026 to 2033. The market is expected to grow significantly owing to the high incidence of cardiovascular diseases and the technological advancements in diagnostic techniques.
Key Market Trends & Insights
- By component, the services segment accounting for the largest revenue share of 45.85% in 2025.
- By facility type, the hospital-based labs segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 16.8 Billion
- 2033 Projected Market Size: USD 17.7 Billion
- CAGR (2026-2033): 5.5%
In addition, the growing expenditure on healthcare by the citizens and the initiatives by the federal government to increase awareness regarding cardiac health are increasing the growth opportunities for the market. Approximately 20.1 million residents in the U.S. above 20 years of age have coronary artery disease. 805,000 Americans have a heart attack annually. West Virginia has the highest prevalence of heart disorders in the U.S., with an age-weighted prevalence of 6.3% as per the CDC.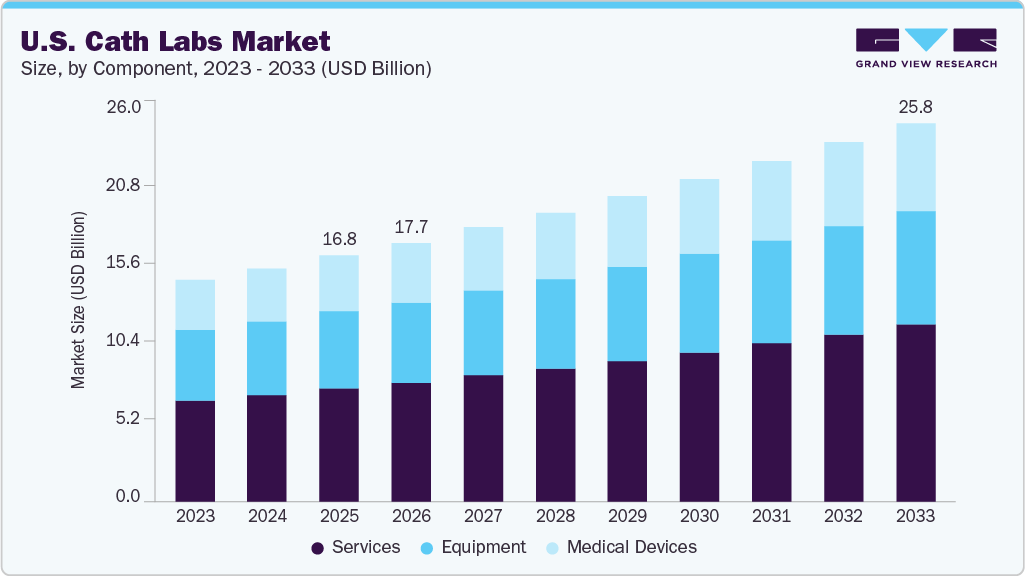
The section highlights the key factors driving the growth of the U.S. cath labs industry, including rising heart diseases, increasing adoption of new technologies, and growing emphasis on clinical excellence, among others.
Market Drivers
The state has nearly 25% of its population above 65 years of age and a 37.7% prevalence of obesity, contributing to the high incidence of heart disease. Overall, the southern states in the U.S. have the worst cardiac health, as per a survey by the University of Alabama and the CDC. The high incidence of cardiovascular disorders is increasing the requirement for early diagnosis, thus increasing the demand for cath labs.
Moreover, technological advancements are significantly impacting market growth. The innovation in diagnostic techniques is enabling accurate, non-invasive, and quick diagnosis. For instance, Beaumont adopted a new cardiac lab technology, control rad, to reduce the patient and staff exposure to hazardous radiation while imaging. The control rad is reported to reduce the exposure to radiation by 50-85%, thus increasing safety during diagnostic procedures. The improvements in soft tissue visualization and the adoption of robots in the cath labs are transforming the level of diagnosis provided. The manufacturers of diagnostic equipment are focused on developing compact, workflow-compatible, and cost-effective robots for effectively managing diagnostic operations while keeping the operating cost low for the labs.
Moreover, there is a growing emphasis on clinical excellence, innovation, and patient-centric care, as demonstrated by leading cardiovascular centers such as the Mount Sinai Fuster Heart Hospital. Institutions ranked among the nation’s top cardiac care providers continually invest in research, advanced technologies, and evidence-based clinical practices, which accelerate the development and adoption of next-generation cath lab systems, minimally invasive intervention tools, and precision-guided devices. Their culture of discovery, pioneering new treatments, setting procedural benchmarks, and translating breakthroughs into real-world practice drives continuous technological upgrades across hospitals nationwide. Combined with a compassionate, outcomes-focused care model, these high-performing centers stimulate broader healthcare investments, industry partnerships, and infrastructure expansion, thereby significantly fueling growth and advancement in the U.S. cath lab market.
The federal government is taking initiatives to increase awareness regarding cardiac health and reduce the increasing prevalence of heart disease. Million Hearts, an initiative supported by the CDC, works for the prevention of heart attacks throughout the nation. The goal of the initiative is to reduce 1 million cardiovascular disease incidences in the next 5 years. The Heart Truth, a national health education program by the National Heart, Lung, and Blood Institute, is increasing awareness about heart disease in the U.S. and is the first initiative by the federal government in response to the increased prevalence of cardiac disorders. The month of February is designated American Heart Month to raise awareness of cardiovascular health. Such initiatives are expanding residents’ diagnostic options.
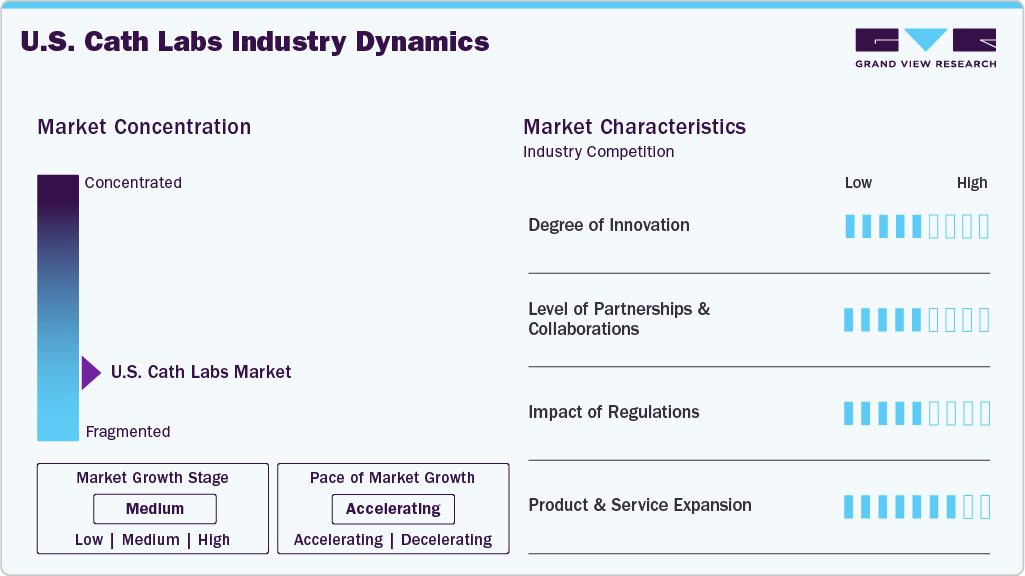
Market Concentration & Characteristics
The chart below illustrates the relationship between the U.S. cath labs market concentration, characteristics, and participants. The degree of innovation is medium, the level of partnerships & collaborations activities is medium, the impact of regulations on the market is high, and the regional expansion of the market is low.
Innovations include the development of robotic-assisted catheterization systems to enhance precision and reduce radiation exposure for both patients and medical staff. For instance, the Corindus Vascular Robotics system allows physicians to perform complex procedures remotely while maintaining control over the catheter’s movements. This technology not only improves procedural outcomes but also expands access to specialized care in remote areas where experienced interventional cardiologists may not be available.
The U.S. cath labs industry saw notable consolidations as larger operators sought to expand their reach and capabilities. For instance, in September 2023, Philips announced a long-term strategic partnership with Leeds Teaching Hospitals NHS Trust. This collaboration aims to enhance cardiac care in the region, particularly through the introduction of advanced medical technologies. On World Heart Day 2023, celebrated in September, Philips highlighted this partnership by unveiling a new fully featured Biplane Catheter Lab at Leeds Teaching Hospitals.
The impact of regulations on the U.S. cath lab market is high, with strict guidelines influencing device development and deployment. Regulatory bodies such as the FDA ensure that all medical devices used in cath labs meet stringent safety and efficacy standards before they can be marketed. For instance, the FDA’s premarket approval process requires extensive clinical trials for new devices, which can delay their entry into the market but ultimately enhance patient safety by ensuring that only effective products are available. Moreover, the Centers for Medicare & Medicaid Services (CMS) plays a crucial role in regulating reimbursement policies for procedures performed in cath labs.
Regional expansion efforts are evident as the rise in outpatient procedures has led to the establishment of freestanding cath labs that operate independently from hospitals. These facilities often provide same-day discharge for patients undergoing routine procedures, thereby improving patient satisfaction and reducing healthcare costs. A notable example is the expansion of outpatient cath lab services by large healthcare systems such as HCA Healthcare and Tenet Healthcare, which have opened multiple locations across multiple states to serve the growing patient population.
Component Insights
The services segment led the U.S. cath labs market, accounting for the largest revenue share of 45.85% in 2025. Its dominance is attributed to the rising prevalence of cardiovascular diseases in the U.S. The service segment is sub-segmented into therapeutic and diagnostic services. Therapeutic services accounted for a dominant share in 2022 due to the majority of patients having heart diseases in the U.S. requiring long-term therapy. Based on components, the market is categorized into service and equipment.
Alabama, West Virginia, Louisiana, and Kentucky are the least heart-healthy states in the U.S., thus having a higher need for diagnostic and therapeutic services. Among all cardiovascular disorders, heart attacks are most prevalent, with every 40 seconds one American having a heart attack, wherein 1 in 5 attacks is a silent one. In such cases, direct therapeutic services are required. The southeastern and northeastern states have funding from national and state organizations for the management of heart diseases, thus fueling segment growth in the region.
The equipment segment is expected to grow at the fastest CAGR over the forecast period. Its growth is attributed to the increasing need for diagnostic and therapeutic surgical procedures. The preference for minimally invasive surgeries resulted in the launch of new equipment on the market. For instance, in October 2023, GE HealthCare announced that it had received FDA clearance for its Allia IGS Pulse system. This innovative technology represents a significant advancement in image-guided systems specifically tailored for cardiac imaging. The Allia IGS Pulse is designed to enhance the precision and efficiency of cardiac procedures, which are critical in diagnosing and treating various heart conditions.
Facility Type Insights
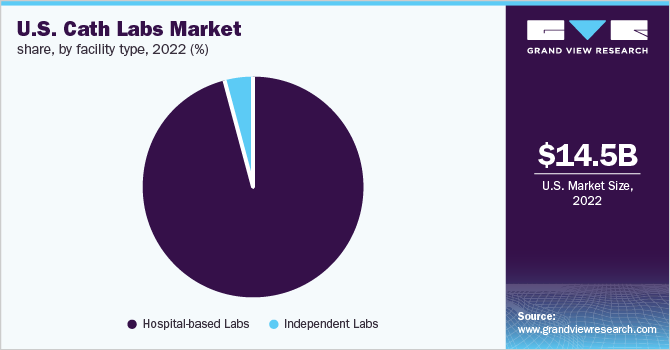
The hospital-based labs segment held the largest revenue share of the U.S. cath labs industry in 2025. Its dominance is attributed to the increasing number of corporate hospital-based cath labs, favorable reimbursement, and the accessibility of top-quality care and equipment in hospital-based settings. Based on facility type, the market is categorized into hospital-based labs and independent labs.
The regulations favored hospitals and provided better reimbursement for hospital inpatient settings. In addition, the hospital-based cath lab unit is among the top cath labs in the nation. In New York, The Mount Sinai Hospital's cath labs have been rated the best in the state. The adoption of the latest technologies for safe and effective diagnosis and treatment is driving a higher preference for such settings. For instance, in May 2024, HCA Florida University Hospital became the first hospital in the U.S. to implement a groundbreaking electrophysiology technology designed for advanced cardiac care. This new technology enhances the precision and effectiveness of arrhythmia correction procedures, thereby improving patient outcomes.
The freestanding cath labs segment is expected to grow at the fastest CAGR over the forecast period, attributed to the lower cost of services and less time-consuming treatment. Moreover, patients prefer independent cath labs because they do not require admission after surgery, resulting in a quicker recovery. The ambulatory catheterization centers facilitate better personalized patient care, which is not often possible in a hospital-based setting.
Key U.S. Cath Labs Company Insights
There are several major manufacturers and service providers in the market engaged in strategic initiatives to strengthen their market presence, including new product & service launches, partnership & collaborations, mergers & acquisitions, and geographical expansion. For instance, in February 2023, Abbott announced a definitive agreement to acquire Cardiovascular Systems, Inc. (CSI), a medical device company known for its innovative atherectomy system used in treating peripheral and coronary artery disease.
Key U.S. Cath Labs Companies:
Key Manufacturers in the U.S. Cath Labs Market
- Shockwave Medical
- Boston Scientific Corporation
- GE Healthcare
- Koninklijke Philips N.V.
- Siemens Healthineers
- Abbott Laboratories
- Stryker
- Cordis
- AngioDynamics
- Medtronic
- Shimadzu Corporation
- Johnson & Johnson
- B. Braun Melsungen AG
- Cardinal Health, Inc.
Key Service Providers in the U.S. Cath Labs Market
- Cleveland Clinic
- Cedars-Sinai
- Mayo Clinic
- NewYork-Presbyterian Hospital
- Northwestern Medicine
- Stanford Health Care
- Houston Methodist
Recent Developments
-
In February 2024, Philips introduced a groundbreaking imaging technology known as LumiGuide, which allows physicians to navigate blood vessels using light instead of traditional X-ray methods. This innovation is powered by Fiber Optic RealShape (FORS) technology and represents a significant advancement in the field of interventional radiology and vascular surgery.
-
In November 2023, a surgical team at Mount Sinai Fuster Heart Hospital became the first in the Northeast to perform the newly FDA-approved, one-hour, minimally invasive Symplicity Spyral renal denervation (RDN) procedure.
-
In October 2023, Shockwave Medical launched the Shockwave C2+ Coronary Intravascular Lithotripsy (IVL) catheter in the U.S., following its approval by the U.S. FDA. The C2+ catheter offers a significant improvement over its predecessor, the Shockwave C2 catheter, by delivering 50% more pulses per catheter. This increase in pulses is expected to improve procedural efficiency and patient outcomes.
-
In July 2023, the Cardiac Catheterization Lab at Banner University Medical Center South officially reopened after undergoing significant renovations and expansions. This retooled facility is designed to enhance the delivery of cardiovascular care, providing a larger and more efficient space for medical procedures related to heart health.
U.S. Cath Labs Market Report Scope
Report Attribute
Details
Revenue forecast in 2033
USD 25.8 billion
Growth rate
CAGR of 5.5% from 2026 to 2033
Actual data
2021 - 2025
Forecast data
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Component, facility type, region
Country scope
U.S.
Key manufacturing companies profiled
Shockwave Medical; Boston Scientific Corporation; GE Healthcare; Koninklijke Philips N.V.; Siemens Healthineers; Abbott Laboratories; Stryker; Cordis; AngioDynamics; Medtronic; Shimadzu Corporation; Johnson & Johnson; B. Braun Melsungen AG; Cardinal Health, Inc.
Key service provider companies profiled
Cleveland Clinic; Cedars-Sinai; Mayo Clinic; NewYork-Presbyterian Hospital; Northwestern Medicine; Stanford Health Care; Houston Methodist
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Cath Labs Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. cath labs market report based on component, and facility type.
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Services
-
Therapeutic
-
Angioplasty
-
Stent placement
-
Rotational atherectomy
-
Thrombectomy
-
Cardioversion
-
Transcatheter Aortic Valve Replacement (TAVR)
-
Others
-
-
Diagnostic
-
Angiography
-
Coronary intravascular ultrasound
-
Cardiac catheterization
-
ECG/EKG
-
Fractional Flow Reserve (FFR)
-
Others
-
-
-
Equipment
-
Imaging Equipment
-
Angiography machines
-
X-ray machines
-
IVUS
-
-
Monitors
-
Procedure Room Equipment
-
X-ray table
-
Heart monitoring equipment
-
Display screens
-
-
Control Room Equipment
-
Viewing monitor
-
Hemodynamic monitoring system
-
-
Other Equipment
-
Infusion pumps
-
Anesthetic and respiratory equipment
-
Crash cart
-
-
-
Medical Devices
-
Stents
-
Sheaths
-
Guidewires
-
Pacemakers
-
Defibrillator
-
CPR device
-
Others
-
-
-
Facility Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Independent Labs
-
Hospital-Based Labs
-
Frequently Asked Questions About This Report
b. The U.S. cath labs market size was estimated at USD 14.5 billion in 2022 and is expected to reach USD 15.2 billion in 2023.
b. The U.S. cath labs market is expected to grow at a compound annual growth rate of 5.34% from 2023 to 2030 to reach USD 21.9 billion by 2030.
b. The services segment dominated the U.S. cath labs market in 2022. This is attributable to the rising prevalence of cardiac disorders and increase awareness regarding importance of early diagnostics.
b. Some key players operating in the U.S. cath labs market include Koninklijke Philips N.V.; Shimadzu Corporation; GE Healthcare; Johnson & Johnson; B. Braun Melsungen AG; Siemens Healthineers; Medtronic; Cardinal Health, Inc; Boston Scientific Corporation; Abbott Laboratories
b. Key factors that are driving the U.S. cath labs market growth include high incidence of cardiovascular diseases, technological advancements in diagnostic techniques, increasing number federal initiatives taken by the government to reduce healthcare expenditure
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










