- Home
- »
- Medical Devices
- »
-
U.S. Cell And Gene Therapy Clinical Trials Market Report, 2033GVR Report cover
![U.S. Cell And Gene Therapy Clinical Trials Market Size, Share & Trends Report]()
U.S. Cell And Gene Therapy Clinical Trials Market (2025 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, II, III, IV), By Indication (Oncology, Cardiology, CNS, Musculoskeletal, Infectious Diseases), And Segment Forecasts
- Report ID: GVR-4-68040-687-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Cell And Gene Therapy Clinical Trials Market Summary
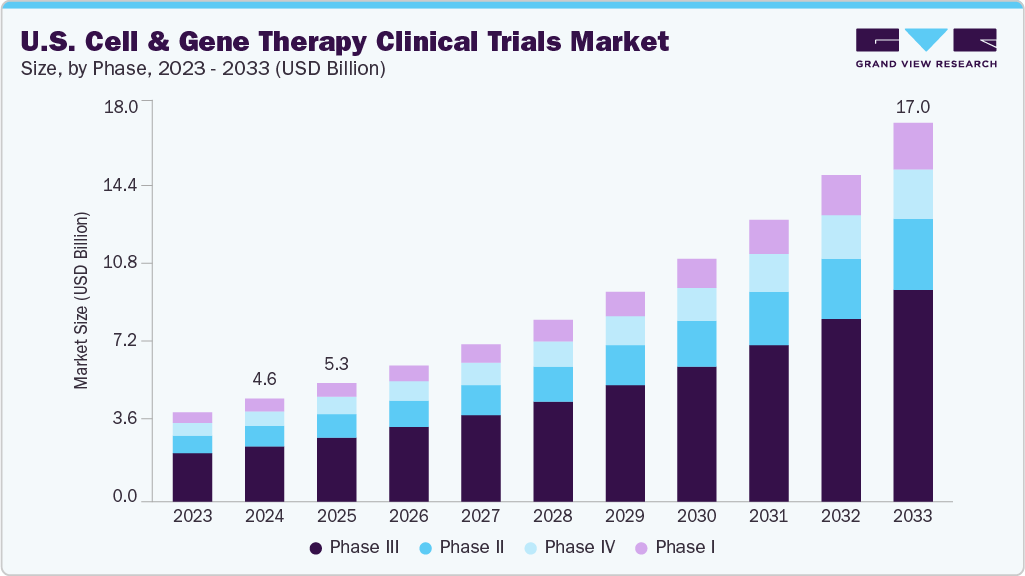
The U.S. cell and gene therapy clinical trials market size was estimated at USD 4.64 billion in 2024 and is projected to reach USD 17.03 billion by 2033, growing at a CAGR of 15.60% from 2025 to 2033. The market is driven by an increase in R&D funding, increasing disease burden in oncology, rare genetic disorders, and neurodegenerative conditions, growing interest in cell and gene therapies for cancer treatment, and a favorable regulatory environment.
Key Market Trends & Insights
- The U.S. cell and gene therapy clinical trials accounted for the largest market revenue share in North America in 2024.
- By phase, the phase III segment led the market with the largest revenue share of 54.35% in 2024.
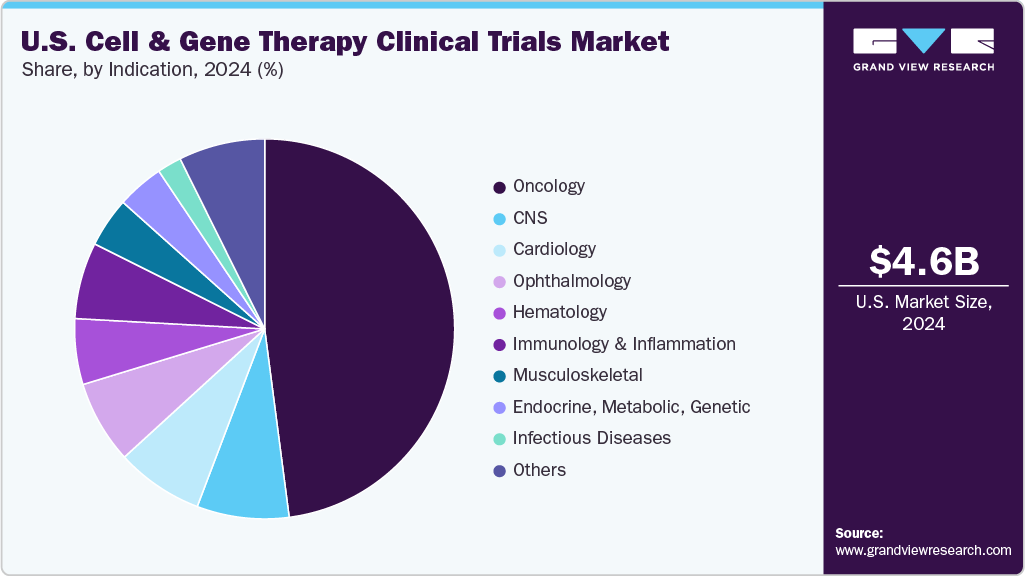
- Based on indication, the oncology segment accounted for the largest market revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 4.64 Billion
- 2033 Projected Market Size: USD 17.03 Billion
- CAGR (2025-2033): 15.60%
- Phase III: Largest market in 2024
- Oncology: Fastest growing market
Besides, the surge in pipeline candidates with over 2,000 CGT assets is currently in development at various preclinical and clinical stages. It has benefited from substantial R&D funding provided by biopharma companies, startups, and government entities. In addition, regulatory advancements, including the FDA’s accelerated approval pathways, RMAT designations, and INTERACT meetings, support the need for cell and gene therapy initiation and development of trials.Moreover, increased investments and M&A activities from large pharmaceutical companies are propelling the expansion of CGT trials, particularly in autologous CAR-T therapies, AAV-based gene therapies, and allogeneic platforms. Service providers and CDMOs are enhancing their CGT-specific capabilities, such as GMP vector production, viral delivery systems, and decentralized trial logistics.
In addition, the market is supported by robust trial infrastructure, which includes leading academic centers, CROs, and advanced CDMOs that facilitate streamlined execution. As the number of investigational CGTs increases, the demand for more precise, scalable, and rapid development solutions is expected to witness new growth opportunities over the estimated time period.
Besides, the rising focus on improving patient access, diversity, and decentralized enrollment is particularly important in rare and pediatric diseases. Recent technological advancements are enhancing trial efficiency and outcomes, with innovations such as next-generation gene editing tools (like CRISPR), allogeneic cell therapy platforms, real-time imaging for treatment monitoring, and automated closed-loop manufacturing systems making significant contributions to the market.
Furthermore, the growing application of artificial intelligence (AI) and machine learning (ML) is optimizing trial design, enhancing patient matching, and facilitating biomarker discovery, thereby driving more efficient and accelerated CGT development in the U.S. Such factors are expected to drive the market over the estimated period.
Opportunity Analysis
The U.S. market offers significant growth opportunities in cell and gene therapy (CGT) clinical trials, driven by a favorable regulatory landscape, strong research and development activities, and a rising demand for advanced therapies. Besides, initiatives from the FDA, including RMAT and Orphan Drug designations, expedite the trial process and foster innovation, especially for rare and complex conditions. The presence of leading academic institutions, experienced contract research organizations (CROs), and specialized CGT contract development and manufacturing organizations (CDMOs) facilitates comprehensive development.
Moreover, robust investment and public-private collaborations are driving early-stage research and clinical expansion. While oncology continues to lead in trial activity, fields such as neurology, ophthalmology, and cardiovascular therapies are gaining momentum in the market. Moreover, innovations like patient registries, digital recruitment tools, and decentralized trial models are enhancing participant access and involvement.
Furthermore, currently there are new emerging opportunities in manufacturing scale-up, allogeneic cell therapy platforms, and combination strategies which further support the market growth. With its established infrastructure and a growing pipeline, the U.S. stands as the most strategic and innovation-oriented market for CGT clinical trials, presenting substantial long-term potential for both sponsors and investors. Such factors are expected to drive the market growth over the estimated time period.
Impact of U.S. Tariffs on the U.S. Cell and Gene Therapy Clinical Trials Market
U.S. tariffs have produced a mixed impact on clinical trials for cell and gene therapy (CGT). Many advanced therapies depend on imported materials such as plasmids, viral vectors, enzymes, and lab equipment. Tariffs on these components, particularly from China and Europe, have raised costs for clinical trial sponsors, especially for small biotech companies with limited budgets. These increased expenses can hinder early-stage research and delay the commencement of trials.
Moreover, contract development and manufacturing organizations (CDMOs) and contract research organizations (CROs) that support CGT trials have similarly felt the effects, as tariffs have raised input costs, impacting service pricing and profit margins. Furthermore, companies are responding by adjusting their sourcing strategies and investing in U.S.-based manufacturing to mitigate tariff exposure. This shift could enhance domestic resilience and fortify the clinical trial ecosystem in the long run. While larger pharmaceutical companies may be able to absorb these extra costs, smaller CGT developers are under greater financial strain. Hence, while U.S. tariffs have created immediate cost challenges, it has also encouraged long-term supply chain localization and infrastructure improvements. The key for the CGT clinical trial sector is to balance short-term financial pressures with strategic adjustments that sustain innovation and ensure patient access to groundbreaking therapies.

Technological advancements are critical in accelerating clinical trials for cell and gene therapy (CGT) in the U.S. Artificial intelligence and machine learning (AI/ML) are optimizing trial design, patient selection, and biomarker discovery, significantly shortening timelines and boosting success rates. Next-generation gene editing tools, such as CRISPR-Cas9 and base editors, provide improved precision in targeting disease-causing genes, enhancing both the safety and efficacy of therapies. Innovations in viral and non-viral delivery methods, including adeno-associated viruses (AAVs), lipid nanoparticles (LNPs), and electroporation techniques, broaden the scope of treatable conditions by improving transgene delivery and minimizing immunogenicity. Allogeneic cell therapy platforms facilitate off-the-shelf treatments, addressing the logistical and cost challenges associated with autologous therapies.
Advanced manufacturing technologies—such as closed systems, automation, and AI-driven quality control—are enhancing scalability and consistency in the production of clinical-grade materials. In addition, real-time imaging and biomarker tracking technologies improve trial monitoring by allowing for non-invasive and dynamic evaluations of therapeutic efficacy and safety. Thus, these innovations are expected to drive the CGT clinical trials across the U.S.
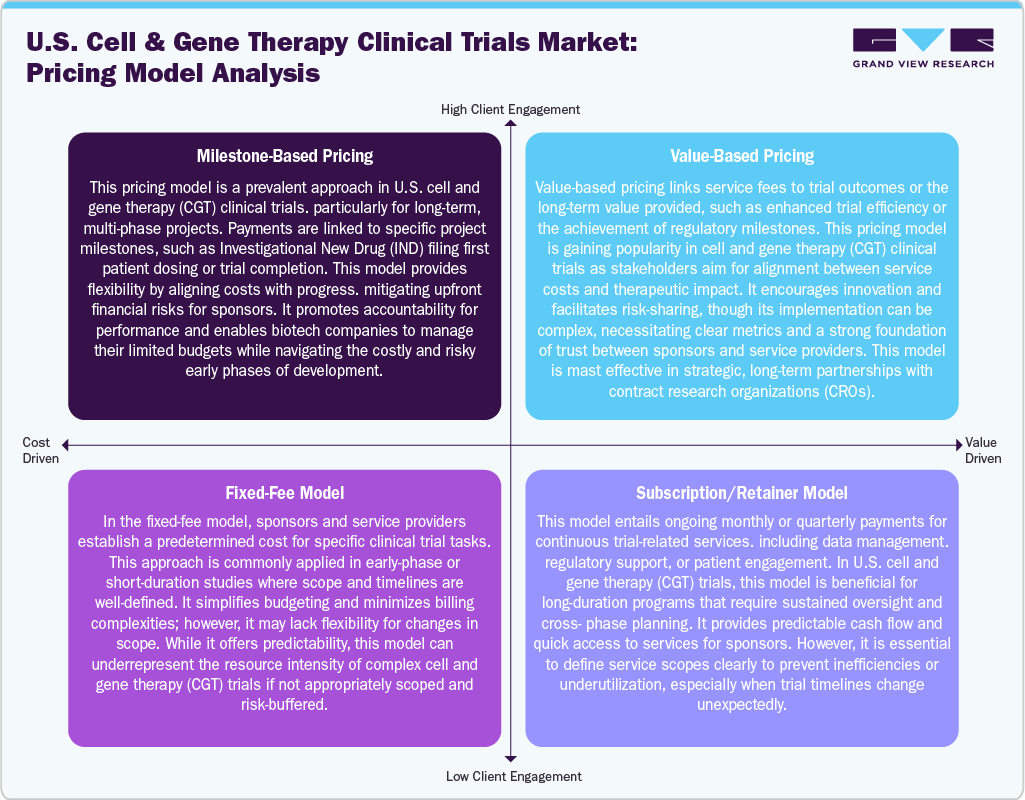
In the U.S. cell and gene therapy clinical trials industry, various pricing models are utilized to align with the complexity and duration of studies. Milestone-based pricing is commonly employed, linking payments to significant project milestones such as IND submissions, patient enrollment, or trial completion. This model fosters accountability for performance and offers budget flexibility. The fixed-fee model provides a predetermined cost for specific trial activities, making it ideal for early-phase trials that have a clear scope and timeline.
Although this approach simplifies budgeting, it may struggle to address unexpected complexities encountered in CGT studies. Besides, the emerging trend in popularity is value-based pricing, where payments are connected to trial outcomes or efficiency improvements. This model encourages shared risks and rewards but necessitates clearly defined success metrics and a high level of trust between partners.
Moreover, the subscription or retainer model facilitates steady access to services via recurring fees, making it well-suited for long-term trials that require ongoing support. While this model offers predictability, it also requires clearly defined service expectations to ensure cost-effectiveness and prevent underutilization.
Market Concentration & Characteristics
The market growth stage of U.S. cell and gene therapy clinical trials is moderate, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
The U.S. cell and gene therapy clinical trials industry is characterized by significant innovation, fueled by advancements in CRISPR-based editing, allogeneic cell platforms, AI-driven trial design, and novel delivery systems. These continuous breakthroughs are transforming therapeutic strategies in areas such as oncology, rare diseases, and regenerative medicine.
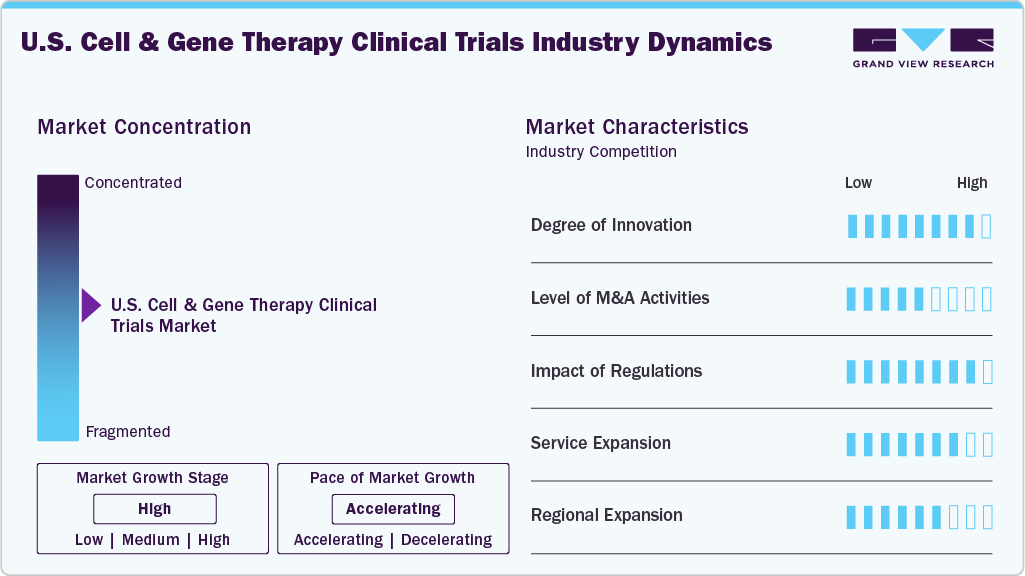
M&A activity continues to drive as CROs, CDMOs, and biotech firms actively pursue acquisitions to enhance their expertise in CGT, acquire proprietary platforms, and broaden their trial capabilities. These strategic activities are driving vertical integration, expanding pipelines, and providing access to specialized manufacturing and early-phase development services.
Supportive FDA frameworks like RMAT, Fast Track, and Orphan Drug status are expediting CGT clinical trials. However, evolving guidance on long-term follow-up, safety monitoring, and CMC requirements is increasing regulatory complexity, requiring sponsors to invest more in compliance and documentation.
Service providers are swiftly broadening their offerings to support CGT trials, incorporating services such as cell sourcing, vector production, decentralized trial models, and real-time patient monitoring. These customized solutions are essential for addressing the specific clinical, logistical, and regulatory requirements of advanced therapies.
Companies are enhancing CGT trial infrastructure in major U.S. biotech hubs like Boston, San Diego, and Philadelphia. These areas provide proximity to academic institutions, access to skilled talent, and diverse patient populations, all of which contribute to improved trial efficiency and foster site-level innovation and collaboration.
Phase Insights
The market is classified into phase I, phase II, phase III, and phase IV. The phase III segment led the market with the largest revenue share of 54.35% in 2024. The growth of the segment is due to the increasing number of late-stage trials aiming to validate safety, efficacy, and durability of treatments in larger patient populations, often targeting high-burden conditions such as cancers, rare genetic disorders, and inherited retinal diseases. Besides, the increasing number of Phase III CGT trials support for growing number of clinical trials, solid preclinical data, and robust investor confidence. Moreover, regulatory incentives like Regenerative Medicine Advanced Therapy (RMAT) and Orphan Drug designations are further accelerating the phase III clinical trials.
The phase I segment is anticipated to grow at the fastest CAGR during the forecast period. Phase I cell and gene therapy (CGT) trials serve as the crucial first step in assessing new therapies, focusing on safety, tolerability, and dosage within small patient populations. These early-stage studies are particularly important for high-risk, high-reward treatments aimed at rare diseases, cancers, and genetic disorders. Besides, innovations in vector design, delivery platforms, and gene-editing technologies have contributed to an increasing Phase I CGT activity, especially in the U.S. Moreover, many of these trials take advantage of FDA programs such as Fast Track and RMAT to facilitate accelerated clinical development. Such factors are expected to drive the market over the estimated period.
Indication Insights
The market is segmented into oncology, cardiology, CNS, musculoskeletal, infectious diseases, dermatology, endocrine, metabolic, genetic, immunology & inflammation, ophthalmology, hematology, gastroenterology, and others. The oncology segment accounted for the largest market revenue share in 2024.Cell and gene therapies (CGT) in the oncology therapeutic area have witnessed significant growth in clinical trials in recent years. Innovative CGT strategies like CAR-T cell therapy, TCR-T, and oncolytic viruses in this therapeutic area are being explored for both hematological malignancies and solid tumors. Besides, the substantial unmet need for targeted and durable cancer treatments has resulted in a notable increase in clinical trial initiations for relapsed or refractory cancers.
In addition, supportive programs from the FDA, such as RMAT & Fast Track designations have further accelerated the expansion of oncology CGT trials supporting the market growth. Moreover, numerous leading companies and academic institutions are actively enhancing their pipelines, focusing on next-generation platforms, allogeneic cell therapies, and combination therapies to improve safety and effectiveness. Furthermore, increased investments from both large pharmaceutical companies and emerging biotech companies are driving innovation in oncology CGT. Such factors are expected to drive the market over the estimated period.
The cardiology segment is expected to grow at the fastest CAGR during the forecast period. The growth of the segment is due to the expanding number of innovative CGT therapies that support repair of damaged heart tissue, stimulate angiogenesis, and tackle the genetic underpinnings of cardiovascular diseases such as heart failure, ischemic cardiomyopathy, and inherited cardiomyopathies. Besides, the global prevalence of cardiovascular disease and the scarcity of curative treatment options is expected to drive the market growth.
Moreover, in cardiology the early-phase trials are experimenting with gene transfer vectors and stem cell-based therapies fueled by advancements in delivery technologies and imaging methods is anticipated to support the market over the estimated period.
Country Insights
U.S. Cell And Gene Therapy Clinical Trials Market
The cell and gene therapy clinical trials market in the U.S. accounted for the largest market revenue share in North America in 2024. The country’s growth is due to the increasing number of clinical trials for cell and gene therapy (CGT), growing focus on scientific breakthroughs, and robust support from regulators, investors, and research institutions. Besides, in the country, the FDA accelerated pathways such as Regenerative Medicine Advanced Therapy (RMAT) and Orphan Drug designations are enabling companies to initiate trials more swiftly and deliver treatments to patients more efficiently. Moreover, the country has rising investment from biotechnology and pharmaceutical companies, which is driving the expansion of trials focused on cancer, rare diseases, and neurological disorders.
In addition, Academic institutions, specialized CDMOs, and experienced CROs support efficient trial execution, further supporting the market growth. Furthermore, high funding levels and strategic partnerships, particularly in oncology, rare diseases, and neurology is anticipated to contribute to market growth. Such factors are expected to drive the market growth over the estimated period.
Key U.S. Cell And Gene Therapy Clinical Trials Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in May 2025, Medable Inc. unveiled its innovative digital-first Long-Term Follow-Up model designed for cell and gene therapy trials. This new platform alleviates the challenges faced by patients and sites involved in complex CGT studies, while ensuring scientific integrity through sustainable data collection over the FDA-mandated 15-year follow-up period for certain CGTs, including those utilizing genome-editing technologies like CRISPR-Cas9. In addition, the company mentioned that extended follow-up supports for monitoring any delayed adverse effects, assessing the effectiveness of the therapy, and evaluating its long-term impact on patients, though it can impose considerable challenges.
Key U.S. Cell And Gene Therapy Clinical Trials Companies:
- IQVIA
- ICON Plc
- LabCorp
- Charles River Laboratories
- PAREXEL International Corp.
- Syneos Health
- Medpace
- Thermo Fisher Scientific, Inc.
- Novotech
- Veristat, LLC
Recent Developments
-
In May 2025, Ensoma mentioned U.S. FDA clearance of the IND application for lead program EN-374 in X-linked chronic granulomatous disease, a rare & severe genetic disorder.The FDA’s clearance will support the company to establish a unique in vivo HSC engineering platform, bringing us closer to improving outcomes for patients with X-CGD & other chronic diseases.
-
In February 2024, IQVIA mentioned the partnership agreement with California Institute for Regenerative Medicine (CIRM), WuXi AppTec, City of Hope, and Charles River Laboratories, to enhance cell and gene therapy clinical trial capabilities, accelerating patient recruitment and data analytics. This collaboration aims to streamline trial execution and improve therapy development timelines.
U.S. Cell And Gene Therapy Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 5.34 billion
Revenue forecast in 2033
USD 17.03 billion
Growth rate
CAGR of 15.60% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, indication
Country scope
U.S.
Key companies profiled
IQVIA; ICON Plc; LabCorp; Charles River Laboratories; PAREXEL International Corp.; Syneos Health; Medpace; Thermo Fisher Scientific, Inc.; Novotech; Veristat, LLC
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Cell And Gene Therapy Clinical Trials Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. cell and gene therapy clinical trials market report based on phase and indication.
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Cardiology
-
CNS
-
Musculoskeletal
-
Infectious diseases
-
Dermatology
-
Endocrine, metabolic, genetic
-
Immunology & inflammation
-
Ophthalmology
-
Hematology
-
Gastroenterology
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. cell and gene therapy clinical trials market size was estimated at USD 4.64 billion in 2024 and is expected to reach USD 5.34 billion in 2025.
b. The U.S. cell and gene therapy clinical trials market is expected to grow at a compound annual growth rate (CAGR) of 15.60% from 2025 to 2033 to reach USD 17.03 billion by 2033.
b. The phase III segment dominated the U.S. cell and gene therapy clinical trials market in 2024, with a market share of 54.35%. Major factors boosting the demand for cell and gene therapy clinical trials services include an increasing number of late-stage trials aiming to validate the safety, efficacy, and durability of treatments in larger patient populations, further fueling the segment's growth.
b. Some key players operating in the U.S. cell and gene therapy clinical trials market include IQVIA, ICON Plc, LabCorp, Charles River Laboratories, PAREXEL International Corp., Syneos Health, Medpace, Thermo Fisher Scientific, Inc., Novotech, and Veristat, LLC. among others.
b. The increase in R&D funding, increasing disease burden in oncology, rare genetic disorders, and neurodegenerative conditions, growing interest in cell and gene therapies for cancer treatment, and a favorable regulatory environment are key growth drivers for this market. Moreover, surging CGT pipeline candidates at various preclinical and clinical stages further support the market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










