- Home
- »
- Medical Devices
- »
-
U.S. Clinical Trials Market Size & Share Analysis Report, 2030GVR Report cover
![U.S. Clinical Trials Market Size, Share & Trends Report]()
U.S. Clinical Trials Market Size, Share & Trends Analysis Report By Phase (Phase I, II, III, IV), By Study Design (Interventional Trials, Observational Trials, Expanded Access Trials), By Indication, And Segment Forecasts, 2022 - 2030
- Report ID: GVR-4-68039-580-4
- Number of Report Pages: 204
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2020
- Forecast Period: 2022 - 2030
- Industry: Healthcare
Report Overview
The U.S. clinical trials market was valued at USD 22.6 billion in 2021 and is expected to expand at a compound annual growth rate (CAGR) of 5.4% from 2022 to 2030. The growth is majorly driven by a rise in R&D activities, increasing vaccine trials, the need for personalized medicine, and the growing use of new technologies across clinical research. The ongoing COVID-19 pandemic is driving the industry to change the way of conducting existing and upcoming clinical trials. As the COVID-19 pandemic has disrupted clinical trials, adopting direct-to-patient (DTP)/direct-from-patient (DFP) services can help overcome increasing challenges of performing clinical trials such as site closings, travel bans, quarantines, and protecting site staff and patients.
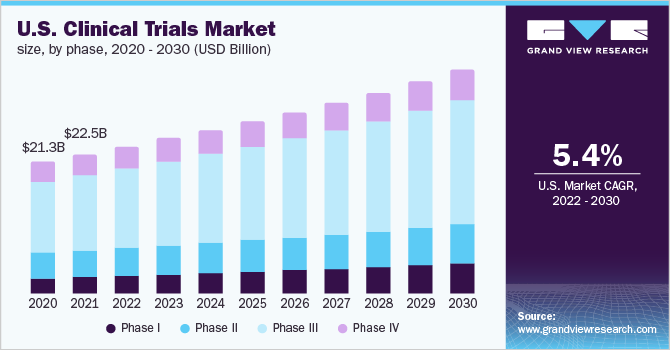
The outbreak of the COVID-19 pandemic has significantly hampered growth. The market witnessed a major drop in its revenues in the year 2020. However, the adoption of virtual technologies such as decentralized clinical trials, remote monitoring, etc. has significantly supported the market to rebound its revenues from 2021 till the forecast years.
Regulatory agencies such as the U.S. FDA, the European Medicines Agency (EMA), the National Institutes of Health (NIH), and China’s National Medical Products Administration, and several other countries have issued guidelines related to the conduct of clinical trials during the outbreak of coronavirus, and are in complete support of incorporating virtual services.
A virtual trial follows a patient-centered approach, allowing for a more conventional trial experience for the participant together with enhanced data collection, increased diversity of patients, and time savings from the perspective of the trial duration.
With the help of this approach, a large population can be considered for the trial, which improves the study, engagement, recruitment, and retention. Virtual trials can remarkably decrease the effort, time, and burden on participants, investigators, and clinical research coordinators. Thus, making it a very effective and efficient approach for the U.S. clinical trials market.
Phase Insights
The phase III segment held the lion’s share of 53.6% in 2021. This is primarily because of the fact that the phase III clinical trials are most crucial as they involve 300 to 3000 participants and have a longer treatment period. Thus, it is the most expensive phase. Although the number of drugs in this phase is relatively low, the complexity associated with this phase is the highest.
The failure rate in this phase is the highest as the sample size and study design require complex dosing at an optimum level. The loss associated with failure is both human and financial and the majority of the failures occur due to non-compliance with safety and efficacy standards. Only one-third of drugs that enter clinical trials make it to phase III trials.
Phase I studies are anticipated to register the fastest CAGR growth of 7.1% during the forecast period. These studies assess the safety of a device or drug and involve the evaluation of the tolerability and pharmacokinetics of molecules. It determines the effect of a device or drug on humans including the way it is absorbed, metabolized, and excreted.
It also examines the side effects of the drug in case of increased dosage levels. The stage includes 20 to 100 healthy volunteers or people with the disease. The high growth of the segment is majorly due to the increasing drug pipeline of pharmaceutical as well as biopharmaceutical companies.
Study Design Insights
The interventional study design segment accounted for the largest market share of 46.0% in 2021. The segment is also anticipated to register the second-fastest growth during the forecast period. Interventional studies are categorized based on the intervention that is to be studied which includes drug or biologic, behavioral, surgical procedure, and devices.
There has been a significant rise in the number of interventional studies carried out over time. The interventional studies comprise 83.4% of total registered studies in the U.S. as of September 2021, out of which the majority of studies are on drug or biologics, followed by behavioral, clinical procedure, and device intervention studies.
The expanded access trials segment is projected to grow at a CAGR of 5.6% during the forecasted period in the U.S. clinical trials market. It would be a prospective approach for patients having a serious disease or condition, which is life-threatening. In this, a patient is allowed to carry out treatment outside of a clinical trial when no satisfactory therapies are available.
The main factor driving the growth of this segment is increasing innovation. For instance, HUTCHMED, in collaboration with Surufatinib, has started an Expanded Access Program (EAP) in the U.S. for Neuroendocrine Tumors for patients having life-threatening conditions that can’t be treated with available medications or clinical trials.
Indication Insights
The oncology segment accounted for the maximum revenue share of 25.2% & is also anticipated to register the fastest CAGR over the forecast period. The increasing prevalence of cancer and the need for effective treatments against various types of cancer is the key factor driving the growth of the segment. Investment in R&D is expected to grow owing to the disease prevalence across the U.S. thereby fueling the market’s growth.
In 2018, there were 1,120 oncology medicines & vaccines in development by America’s biopharmaceutical firms. These include 137 for various types of leukemia, 135 for lymphoma, 132 for lung cancer, 108 for breast cancer & other medicines for ovarian cancer, prostate cancer, colorectal cancer, childhood cancer, and solid tumors.
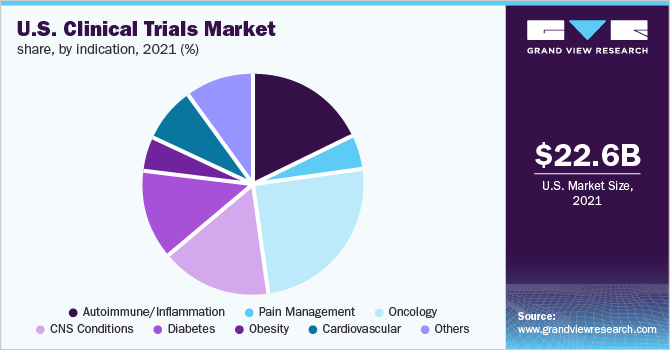
The autoimmune segment of the U.S. clinical trials market is expected to be the second most lucrative segment with a CAGR of 5.6% in 2021 due to significant growth in the prevalence of the autoimmune disease, owing to the changing lifestyle and aging population. The autoimmune diseases affect one in 15 Americans including 18 million women & 5 million men.
They are among the top 10 causes of death in women & female children in all age groups up to 64 years of age. There has been a notable increase in R&D spending on autoimmune diseases. As per the NIH’s estimate autoimmune costs approximately USD 100 billion a year in medical care.
Key Companies and Market Share Insights
The market is highly competitive due to the presence of several international companies. The most important factors that are affecting the competitive nature are the quick adoption of advanced technology for improved healthcare. Moreover, players are acquiring, partnering, and collaborating with other firms to gain a higher market share.
For instance, in May 2020, Concave formed an alliance with Medable to offer decentralized and hybrid trials. This alliance is helpful in the current scenario as it will reduce the risk of infection and the patient’s progress can be monitored virtually without hampering the clinical trial. Some prominent players in the U.S. clinical trials market include:
-
Parexel International Corp.
-
IQVIA Holdings Inc.
-
LabCorp
-
Thermo Fisher Scientific (Pharmaceutical Product Development)
-
Charles River Laboratory
-
ICON plc
-
Wuxi AppTec
-
Eli Lilly and Company
-
SGS SA
-
Novo Nordisk A/S
-
Clinipace
-
Syneos Health Inc.
-
Pfizer Inc.
U.S. Clinical Trials Market Report Scope
Report Attribute
Details
Market Size value in 2022
USD 23.8 billion
Revenue forecast in 2030
USD 36.3 billion
Growth rate
CAGR 5.4% from 2022 to 2030
Base year for estimation
2021
Historical data
2018 - 2020
Forecast period
2022 - 2030
Quantitative units
Revenue in USD million and CAGR from 2022 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication
Report coverage
Revenue forecast, company share, competitive landscape, growth factors and trends
Key companies profiled
Parexel International Corp.: IQVIA Holdings Inc.; LabCorp; Thermo Fisher Scientific (Pharmaceutical Product Development); Charles River Laboratory; ICON plc; Wuxi AppTec; Eli Lilly and Company; SGS SA; Novo Nordisk A/S; Clinipace; Syneos Health Inc.; Pfizer Inc.
Customization scope
If you need specific market information, which is not currently within the scope of the report, we will provide it to you as a part of the customization.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
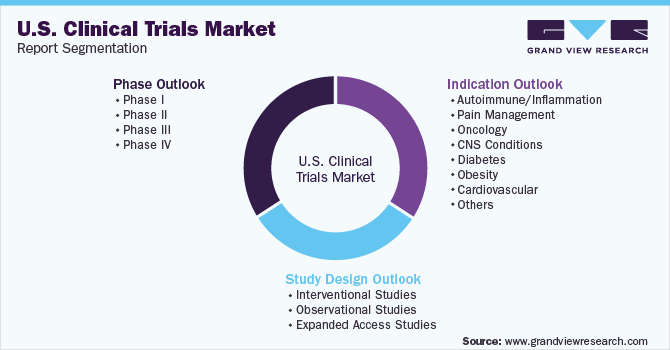
U.S. Clinical Trials Market Segmentation
This report forecasts revenue growth at a country level and provides an analysis on the industry trends in each of the sub-segments from 2018-2030. For this study, Grand View Research has segmented the U.S. clinical trials market based on phase, study design, and indication:
-
Phase Outlook (Revenue, USD Million, 2018 - 2030)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2018 - 2030)
-
Interventional Studies
-
Observational Studies
-
Expanded Access Studies
-
-
Indication Outlook (Revenue, USD Million, 2018 - 2030)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Other
-
-
CNS Conditions
-
Epilepsy
-
Parkinson’s Disease (PD)
-
Huntington’s Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. clinical trials market size was estimated at USD 22.6 billion in 2021 and is expected to reach USD 23.8 billion in 2022.
b. The U.S. clinical trials market is expected to grow at a compound annual growth rate of 5.4% from 2022 to 2030 to reach USD 36.3 billion by 2030.
b. Phase III dominated the U.S. clinical trials market with a share of 53.6% in 2021. Patent expirations of blockbuster drugs and the global financial crisis are factors expected to propel the market growth over the forecast period.
b. Some key players operating in the U.S. clinical trials market include IQVIA; PAREXEL International Corporation; Pharmaceutical Product Development, LLC; PAREXEL International Corporation; Charles River Laboratory; ICON Plc; Wuxi AppTec; and PRA Health Sciences.
b. Key factors that are driving the U.S. clinical trials market growth include the increasing prevalence of chronic disease and the growing demand for clinical trials in developing countries.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





