- Home
- »
- Medical Devices
- »
-
U.S. Durable Medical Equipment Market Size Report, 2033GVR Report cover
![U.S. Durable Medical Equipment Market Size, Share & Trends Report]()
U.S. Durable Medical Equipment Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Monitoring & Therapeutic Devices, Personal Mobility Devices), By End Use (Hospitals, Specialty Clinics, Ambulatory Surgical Centers), And Segment Forecasts
- Report ID: GVR-1-68038-926-5
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
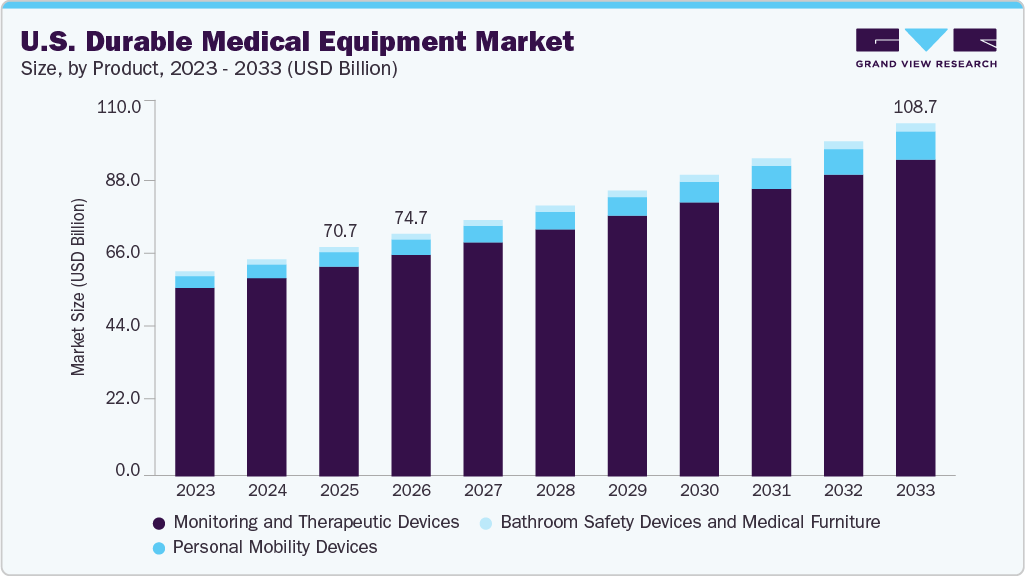
U.S. Durable Medical Equipment Market Summary
The U.S. durable medical equipment market size was estimated at USD 70.66 billion in 2025 and is projected to reach USD 108.73 billion by 2033, growing at a CAGR of 5.51% from 2026 to 2033. The prevalence of chronic diseases, such as cancer, neurological conditions, diabetes, and heart disorders, is increasing annually. According to the data published by the CDC in May 2024, chronic diseases such as diabetes, heart disease, & cancer are the leading causes of disability and death in the U.S. The increasing number of individuals suffering from chronic diseases such as cancer is anticipated to boost the demand for durable medical equipment as patients suffering from chronic conditions can experience mobility issues.
According to an article published by Lifeway Mobility in September 2023, around 25%-35% of cancer patients experience issues with walking, balance, and limb numbness. Hence, the growing cancer prevalence is anticipated to boost the demand for durable medical equipment, such as walkers, wheelchairs, and other mobility solutions in the U.S.
Table 1 Estimated new cases for selected cancers by state, 2023
State
Estimated new cases for selected cancers by state, 2023
Alabama
30,730
Alaska
3,390
Arizona
41,120
Arkansas
18,670
California
192,770
Colorado
28,920
Connecticut
23,480
Delaware
7,240
District of Columbia
3,520
Florida
162,410
Georgia
61,170
Hawaii
8,460
Idaho
10,810
Illinois
74,580
Indiana
40,270
Iowa
20,460
Kansas
16,840
Kentucky
30,270
Louisiana
28,580
Maine
10,490
Maryland
35,200
Massachusetts
42,880
Michigan
61,910
Minnesota
34,380
Mississippi
18,210
Missouri
37,910
Montana
7,100
Nebraska
11,530
Nevada
17,370
New Hampshire
9,580
New Jersey
56,150
New Mexico
11,280
New York
123,810
North Carolina
67,690
North Dakota
4,370
Ohio
74,140
Oklahoma
23,420
Oregon
26,030
Pennsylvania
88,450
Rhode Island
7,030
South Carolina
33,890
South Dakota
5,340
Tennessee
43,790
Texas
139,100
Utah
13,840
Vermont
4,370
Virginia
47,100
Washington
44,630
West Virginia
12,840
Wisconsin
37,640
Wyoming
3,170
U.S.
1,958,310
Source: American Cancer Society, U.S. CDC, FDA, Grand View Research
The data highlights a substantial and geographically widespread cancer patient population across U.S. states. This high disease burden indicates sustained demand for oncology diagnostics, therapeutics, and supportive care services, positioning the U.S. as a strategically attractive market for investment by key industry players seeking long-term growth opportunities.
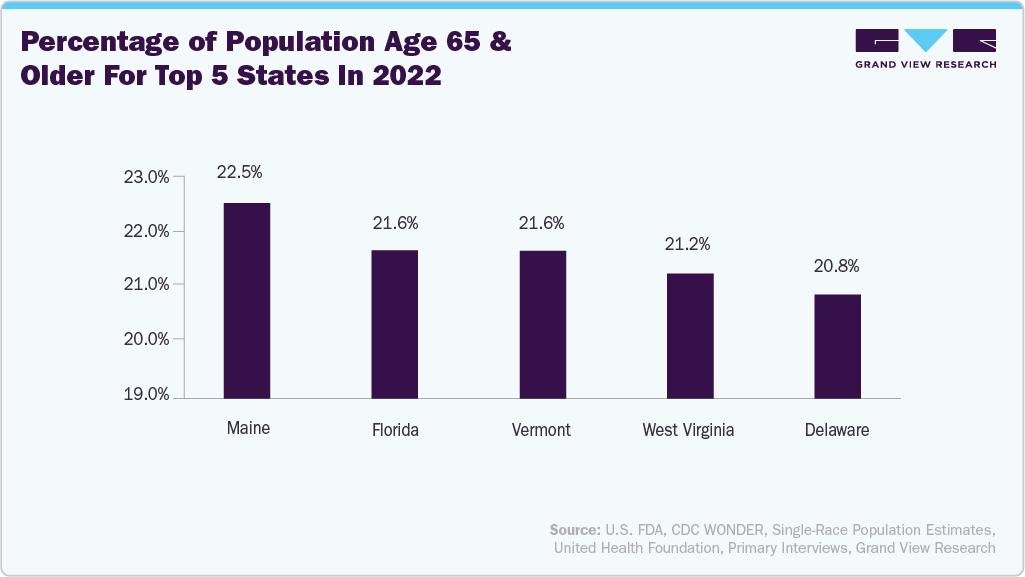
Moreover, the aging population is expected to propel the U.S. LTC and diagnostic DME market. According to data published by the Population Reference Bureau in January 2024, the number of individuals in America aged 65 and older is anticipated to rise by 47% from 58 million in 2022 to 82 million by 2050. The 65-and-older age group’s share of the overall population is estimated to increase from 17% to 23%. Aging is considered to be the greatest risk factor for chronic conditions. The rising geriatric population is expected to increase susceptibility toward chronic conditions, such as diabetes, cardiovascular disorders, and other lifestyle disorders.
According to the data published by the National Council on Aging, Inc. in July 2024,
-
Around 57.8 million individuals aged over 65 years were living in the U.S. in 2022
-
West Virginia, Florida, Maine, and Vermont had the highest percentage of people aged 65+ in 2022.
Around 20.1% of individuals aged 65+ had been diagnosed with diabetes
Durable medical equipment such as wheelchairs, canes, and crutches play a vital role in facilitating the safe & efficient movement of elderly patients within healthcare facilities, supporting their treatment and care. Hence, the demand for durable medical equipment is expected to grow in the coming decade due to the increasing geriatric population.
Key Opinion Leader (KOL) Insights And Strategic Perspectives
This section presents leadership perspectives from key industry players, highlighting strategic initiatives, investment priorities, and innovation trends shaping the medical equipment market, including acquisitions, manufacturing localization, and product innovation across homecare and mobility segments.
Opinion Leader (KOL) Insights
Company
Strategic Focus Area
KOL Statement
Quipt Home Medical
M&A Strategy & Healthcare Partnerships
“Acquiring this Ballad Health owned medical equipment provider and concurrently entering into a Preferred Provider Agreement with Ballad Health exemplifies our commitment to creating lasting, system-wide healthcare partnerships that enhance the delivery of home-based care,” said Greg Crawford, CEO
BD
Domestic Manufacturing & Supply Chain Resilience
"Domestic manufacturing is crucial for ensuring a resilient supply of essential health care devices, "By expanding our production capacity, we are not only meeting the critical needs of patients and providers, but we also are reinforcing our commitment to the nation's health care infrastructure. Said Eric Borin, president of Medication Delivery Solutions at BD
Airchair
Product Innovation & Regulatory Compliance
“Airchair II demonstrates not just our design and manufacturing capability but also our understanding of the issues affecting both reduced mobility passengers and the airlines. In a really challenging commercial environment airlines need to ensure that any solution meets their space and weight requirements and the Airchair II does just that.” Said Sarah McOnie of Airchai
Market Concentration & Characteristics
The market growth stage is moderate at an accelerating pace. The market is characterized by a moderate-to-high degree of growth. Key drivers include a combination of technological advancements, aging demographics, and the increasing prevalence of chronic diseases. The market is characterized by a diverse range of products, including mobility aids, respiratory equipment, and patient monitoring devices. Regulatory frameworks, such as Medicare reimbursement policies, significantly influence market dynamics, shaping the competitive landscape and market entry barriers.
The market showcases a strong trend toward home healthcare solutions, emphasizing patient convenience and cost-effectiveness. Advances in materials and design contribute to durable and user-friendly equipment, while a focus on telehealth integration further propels the market forward. As the healthcare landscape evolves, the industry continues to adapt, offering innovative solutions that cater to the evolving needs of healthcare providers and patients alike. For instance, in January 2024, Robooter, a mobility device manufacturer specializing in robotic technology for the elderly and disabled, introduced its innovative power wheelchair X40 at the Consumer Electronics Show (CES) 2024 in Las Vegas. The company expanded its product range with the launch of the E Series in 2023, emphasizing adaptability for both indoor and outdoor use. This strategic move caters to the diverse needs of elders and individuals with disabilities. Robooter's focus on the U.S. market is evident through its active efforts to strengthen partnerships with local collaborators. Participation in CES, alongside esteemed American collaborators, underscores the company's dedication to expanding its presence and advancing accessibility solutions in the U.S. market. The impact of Robooter's technological advancements is poised to contribute significantly to the evolution of mobility solutions within the industry.
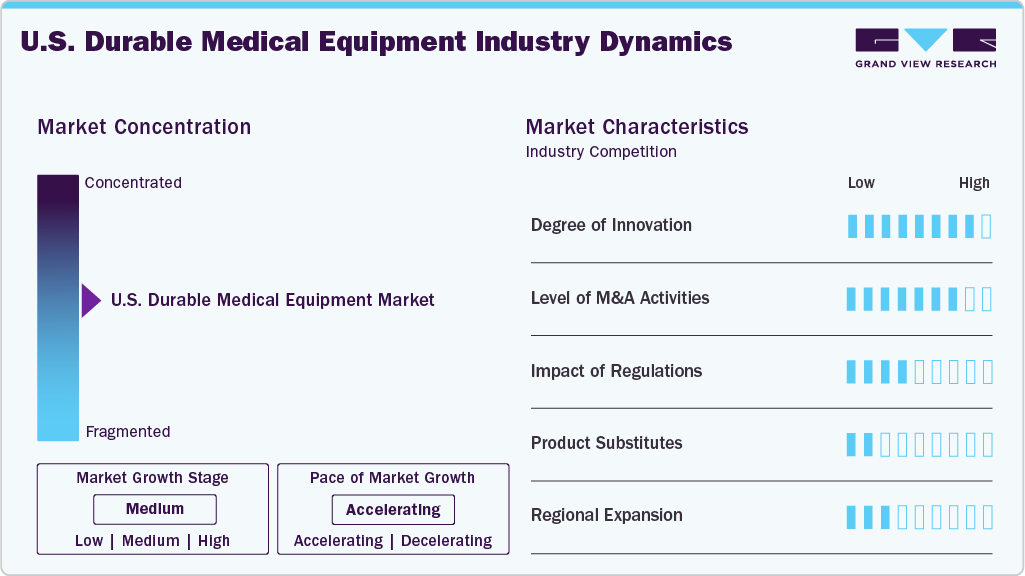
The market is marked by a significant degree of innovation, propelled by rapid technological advancements and a heightened adoption of advanced products, especially in home care settings. This innovation surge is fueled by a growing demand for home care services, driving the development and integration of cutting-edge technologies such as artificial intelligence, big data analytics, and remote monitoring within the DME sector. Companies heavily investing in these technological advancements gain a competitive edge, contributing to the market's overall growth and reflecting a dynamic landscape that continually meets evolving healthcare needs through innovation.
Regulations play a pivotal role in shaping the market. Stringent regulatory frameworks, particularly related to reimbursement policies such as those governed by Medicare, significantly influence market dynamics. Compliance with these regulations poses challenges but is crucial for market entry. The regulatory environment fosters a standardized approach, ensuring the safety and efficacy of DME. It also acts as a key determinant in the competitive landscape, affecting product development and market access for companies operating in the U.S. DME sector. Overall, regulations have a profound impact, guiding industry practices and contributing to the market's stability and credibility.
The market is witnessing a notable level of merger and acquisition activities. Companies within the sector are strategically consolidating to enhance their market presence, broaden product portfolios, and achieve economies of scale. M&A transactions are driven by the pursuit of synergies, technological advancements, and the need for strategic partnerships to navigate evolving healthcare landscapes. This trend indicates a dynamic market where businesses are aligning resources to capitalize on growth opportunities and address challenges, ultimately influencing the competitive landscape and shaping the future trajectory of the U.S. DME market. For instance, in November 2023, Humana Inc., a health and well-being company, partnered with national organizations Rotech Healthcare Inc. and AdaptHealth Corp. to provide DME services. This collaboration is set to commence on July 1 under a value-based structure that aims to enhance health support for Humana Medicare Advantage HMO members in their homes. Each national DME provider will serve specific regions of the country, marking a strategic move in the evolving DME market.
Product substitutes vary depending on the specific equipment category and the intended use. In mobility aids, lightweight manual wheelchairs or powered exoskeletons may substitute traditional motorized wheelchairs. For respiratory equipment, portable oxygen concentrators and wearable CPAP devices are gaining traction as substitutes for bulkier traditional machines due to their convenience and ease of use.
Regional expansion initiatives are strengthening distribution efficiency and improving product accessibility within the homecare DME segment. Strategic partnerships enable faster delivery, improved patient outcomes, and broader geographic coverage, contributing to market growth and supplier penetration. For instance, in December 2025, Medline signed a supplier agreement with Texas-based Coastal Bend DME, providing access to Medline’s extensive product portfolio and next-day delivery capabilities across South Texas, enhancing supply availability, logistics efficiency, and pricing competitiveness. Leadership insights from both organizations further illustrate how the partnership supports regional expansion, supply chain optimization, and improved service delivery.
“Partnering with Medline allows us to expand our product offerings and better serve our clients with reliable and innovative medical supplies, We’re excited about this formalized agreement, which brings competitive pricing and streamlined logistics to our operations, ultimately benefiting the communities we serve.”
said Raymond Salloum, owner of Coastal Bend DME.
“We are thrilled to solidify our relationship through this new agreement, “This deal formalizes our commercial relationship, enables drop shipping and leverages our Medline Home Direct program to deliver single-patient orders directly to residences.”
said Britt Murray, post-acute care sales representative, Medline.
End Use Insights
The hospital segment dominated the market in 2025, driven by a surge in patients with rising chronic diseases in the country. Hospitals, equipped with advanced medical technology and skilled healthcare professionals, are the primary facilities for addressing these disorders. Moreover, the growing number of hospitals is expected to drive the demand for durable medical equipment. For instance, in September 2023, Orlando Health introduced Florida’s first orthopedic hospital to provide physician training, orthopedic care, and rehabilitation space. Thus, the increasing number of hospitals is expected to boost the demand for durable medical equipment, including mattresses, bedding devices, and patient positioning devices, thus helping drive segment growth in the coming years.
Home healthcare is expected to witness the highest growth rate over the forecast period due to the cost-saving approach for the patient to get remote healthcare services. The rising preference for home-based care aligns with a broader trend in the healthcare industry, emphasizing convenience, reduced healthcare costs, and the comfort of receiving medical services within the familiar environment of one's home. This surge in demand for durable medical equipment within the home healthcare setting reflects a transformative shift in patient care delivery. As the industry continues to adapt to these changing dynamics, the market stands ready to play a pivotal role in shaping the future landscape of patient-centric healthcare.
Product Insights
The monitoring and therapeutic devices segment held the largest revenue share of over 91.38% in 2025 due to the increasing prevalence of chronic diseases requiring long-term care. In addition, increasing patient preference for at-home medical treatment or remote medical services over an in-patient setting is contributing to the segment growth. In monitoring and therapeutic devices, neuromonitoring techniques are expected to witness growth over the forecast period as they are becoming a part of standard medical practices and are routinely used during many surgical procedures. According to the data published by the CDC in May 2024, about 38 million individuals in the U.S. have diabetes, and approximately 98 million American adults are prediabetic. Moreover, as per the data published by the National Heart, Lung, and Blood Institute in October 2023, COPD affects around 16 million adults in the U.S. Thus, the growing patient population suffering from chronic diseases across the U.S. is anticipated to support the segment growth in the coming years.
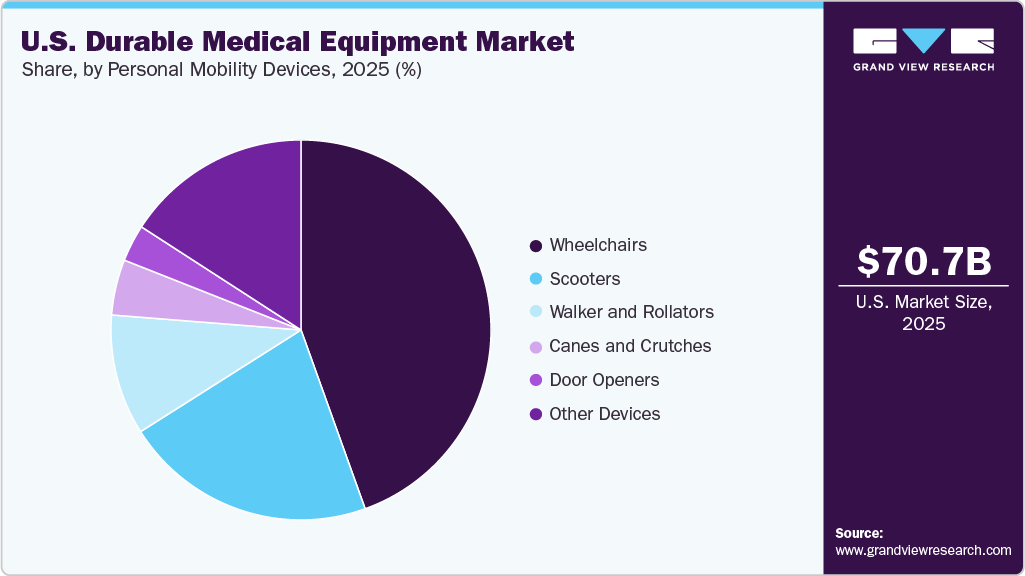
The personal mobility devices segment is anticipated to register the fastest growth rate during the forecast period. The growing geriatric population and rising chronic illnesses are expected to create more need and demand for mobility products over the forecast period as these factors generally reduce the ability to move and perform physical tasks to maintain independent functioning. Moreover, aged people are more prone to falls, which can cause fatal or non-fatal injuries.
Key U.S. Durable Medical Equipment Company Insights
Key companies in the U.S. DME market are proactively driving industry evolution through strategic initiatives such as mergers & acquisitions, regional expansion, and innovative product development. With a focus on capturing a substantial market share, these players introduce cutting-edge technologies and diversified products. The market landscape is characterized by a mix of major players, local distributors, and suppliers. The key emphasis is on providing cost-efficient products with remote usability. Notably, trends such as strategic home healthcare solutions, artificial intelligence integration, and expanded telehealth capabilities are pivotal, addressing evolving healthcare needs and propelling overall market growth. This dynamic landscape sets new industry standards, fosters healthy competition, and enhances the overall quality of products and services in the industry.
Key U.S. Durable Medical Equipment Companies:
- Invacare Corporation
- Sunrise Medical
- Arjo
- Medline Industries, Inc.
- GE HealthCare
- GF Healthcare Products, Inc.
- Carex Health Brands, Inc.
- Cardinal Health
- Drive DeVilbiss Healthcare
- NOVA Medical Products
- Kaye Products, Inc.
- Karman Healthcare, Inc.
- Numotion
- Pride Mobility Products Corporation
- Inogen, Inc.
- CAIRE, Inc.
- Portable Oxygen Solutions
- Rhythm Healthcare, LLC
- MedaCure Inc.
- Compass Health Brands
- Thermo Fisher Scientific, Inc.
- Abbott
- F. Hoffmann-La Roche Ltd.
- Medtronic
- Ascensia Diabetes Care Holdings AG
- Byram Healthcare Centers, Inc.
- Novo Nordisk
- Insulet Corporation
- Baxter
- B. Braun SE
- Micrel Medical Devices SA
- Boston Scientific Corporation
- Tandem Diabetes Care, Inc.
- BD
- Moog, Inc.
- IRadimed Corporation
- Koninklijke Philips N.V.
- Siemens Medical Solutions USA, Inc.
- Essity
- Coloplast Corporation
- Paul Hartmann AG
- Hollister Incorporated
- Convatec Group PLC
- Enovis Corporation
- Zynex, Inc.
- ZOLL Medical Corporation
- Össur
- Breg, Inc.
- Bauerfeind USA, Inc.
- Biotronik
- AliMed
Recent Developments
-
In August 2025, Airchair has introduced the Airchair II, a lightweight onboard wheelchair designed to help airlines meet updated U.S. Department of Transportation requirements under Regulation 14 CFR Part 382.65, part of the Air Carrier Access Act. Airchair II weighs just 6.8 kg but supports up to 250 kg, making it easy for cabin crew to stow, deploy, and maneuver while improving accessibility for passengers with reduced mobility. The product aims to ensure compliance with new accessibility standards by October 2026.
-
In July 2025, Quipt Home Medical has acquired a Ballad Health-owned durable medical equipment (DME) provider with USD 6.6 million in annual revenue, serving over 12,500 patients across four branches. The deal includes a preferred provider agreement covering 20 hospitals in four states, strengthening Quipt’s health system partnerships, expanding its regional footprint, and enhancing care coordination and referral channels.
-
In January 2025, Becton, Dickinson and Company (BD) announced additional investments in its U.S. manufacturing network to expand capacity for critical medical devices, including syringes, needles, and IV catheters, to support the nation’s healthcare needs. New production lines installed in Connecticut and Nebraska will significantly increase output, with further investments planned for Utah. The expansion enhances domestic supply resilience, supports healthcare delivery, and reinforces BD’s long-term commitment to local manufacturing of essential medical products.
-
In August 2024, Medline successfully completed the acquisition of the surgical solutions business from Ecolab Inc. This acquisition strengthens Medline's position in the surgical products sector, expanding its portfolio of offerings to healthcare providers.
-
In June 2024, Abbott received U.S. FDA clearance for two new over-the-counter CGM systems. These advanced CGM systems are designed to offer individuals greater accessibility and convenience in managing their glucose levels without requiring a prescription.
-
In June 2024, Boston Scientific Corporation announced that it entered into a definitive agreement to acquire Silk Road Medical, Inc. This medical device company has developed a pioneering platform for preventing stroke in patients with carotid artery disease through a minimally invasive procedure known as Transcarotid Artery Revascularization (TCAR).
-
In March 2024, Sunrise Medical introduced the QUICKIE Q50 R Carbon, the lightest folding power wheelchair in the QUICKIE lineup, weighing just 32 lbs. This compact and easily transportable wheelchair is designed for effortless storage and maneuverability, offering an ideal solution for individuals who wish to stay active & independent without the hassle of a bulky and heavy mobility device.
-
In January 2024, Novo Nordisk entered research collaborations with Omega Therapeutics and Cellarity to explore novel treatment approaches for cardiometabolic diseases.
U.S. Durable Medical Equipment Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 74.69 billion
Revenue forecast in 2033
USD 108.73 billion
Growth Rate
CAGR of 5.51% from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends, product outlook
Segments covered
Product, end use
Key companies profiled
Invacare Corporation; Sunrise Medical; Arjo; Medline Industries, Inc.; GE HealthCare; GF Healthcare Products, Inc.; Carex Health Brands, Inc.; Cardinal Health; Drive DeVilbiss Healthcare; NOVA Medical Products; Kaye Products, Inc.; Karman Healthcare, Inc.; Numotion; Pride Mobility Products Corporation; Inogen, Inc.; CAIRE, Inc.; Portable Oxygen Solutions; Rhythm Healthcare, LLC; MedaCure Inc.; Compass Health Brands; Thermo Fisher; Scientific, Inc.; Abbott; F. Hoffmann-La Roche Ltd.; Medtronic; Ascensia Diabetes Care Holdings AG; Byram Healthcare Centers, Inc.; Novo Nordisk; Insulet Corporation; Baxter; B. Braun SE; Micrel Medical Devices SA; Boston Scientific Corporation; Tandem Diabetes Care, Inc.; BD; Moog, Inc.; IRadimed Corporation; Koninklijke Philips N.V.; Siemens Medical Solutions USA, Inc.; Essity; Coloplast Corporation; Paul Hartmann AG; Hollister Incorporated; Convatec Group PLC; Enovis Corporation; Zynex, Inc.; ZOLL Medical Corporation; Össur; Breg, Inc.; Bauerfeind USA, Inc.; Biotronik; AliMed
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Durable Medical Equipment Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. durable medical equipment market report based on product, and end use.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Personal Mobility Devices
-
Wheelchairs
-
Electric Wheelchairs
-
Manual Wheelchairs
-
-
Scooters
-
Walker and Rollators
-
Cranes and Crutches
-
Door Openers
-
Other Devices
-
-
Bathroom Safety Devices and Medical Furniture
-
Commodes and Toilets
-
Mattress & Bedding Devices
-
-
Monitoring and Therapeutic Devices
-
Blood Glucose Monitors
-
Continuous Passive Motion (CPM)
-
Infusion Pumps
-
Nebulizers
-
Oxygen equipment
-
Continuous Positive Airway Pressure (CPAP)
-
Suction Pumps
-
Traction Equipment
-
Others
-
Insulin Pumps
-
Ostomy Bags & Accessories
-
Wound Care Products
-
Cardiology Devices
-
Vital Signs Monitor
-
Respiratory Supplies
-
Urinary Supplies
-
Diabetic Supplies
-
Incontinence Products
-
Orthopedic Braces & Support
-
Muscle Stimulators
-
Others
-
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Clinics
-
Ambulatory Surgical Centers
-
Diagnostic Centers
-
Home Healthcare
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. durable medical equipment market size was estimated at USD 70.66 billion in 2025 and is expected to reach USD 74.69 billion in 2026.
b. The U.S. durable medical equipment market is projected to grow at a CAGR of 5.51% from 2026 to 2033 to reach USD 108.73 billion by 2033.
b. Based on product, monitoring and therapeutic devices segment held the largest revenue share of over 91.38% in 2025 due to the increasing prevalence of chronic diseases requiring long-term care.
b. Key players operating in the global U.S. durable medical equipment market include Invacare Corporation, Sunrise Medical, Arjo, Medline Industries, Inc., GE HealthCare, GF Healthcare Products, Inc., Carex Health Brands, Inc., Cardinal Health, Drive DeVilbiss Healthcare, NOVA Medical Products, Kaye Products, Inc., Karman Healthcare, Inc.
b. The U.S. durable medical equipment market is driven by an aging population, rising chronic disease burden, and growing demand for home-based care. Increased hospital-to-home transitions and post-acute care needs support equipment use. Medicare and private insurance reimbursement enable access. Demand is further supported by mobility assistance needs, long-term care requirements, and patient preference for treatment at home, alongside gradual technology integration in home-use medical devices.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










