- Home
- »
- Biotechnology
- »
-
U.S. Plasmid Purification Market Size, Industry Report, 2033GVR Report cover
![U.S. Plasmid Purification Market Size, Share & Trends Report]()
U.S. Plasmid Purification Market (2025 - 2033) Size, Share & Trends Analysis Report By Product & Service (Instruments, Kits & Reagents), By Grade (Molecular Grade, Transfection Grade), By Application, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-703-7
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Plasmid Purification Market Trends
The U.S. plasmid purification market size was estimated at USD 699.4 million in 2024 and is projected to reach USD 1.84 billion by 2033, growing at a CAGR of 11.41% from 2025 to 2033. This growth is driven by increasing demand for gene therapy, vaccine development, and biopharmaceutical research, where high-quality plasmid DNA is critical.

Vaccine Development (Especially DNA Vaccines)
The rising demand for DNA-based vaccines is one of the significant market drivers. Plasmids are essential vectors in these vaccines, delivering genetic instructions to host cells to produce antigens and stimulate immune responses. DNA vaccines offer faster development, greater stability, and easier scalability than traditional vaccines. These benefits have prompted growing interest from pharmaceutical companies and research institutions in expanding DNA vaccine pipelines, increasing the demand for high-quality plasmid DNA used in preclinical studies and commercial production.

Beyond their application in infectious diseases such as Zika, HPV, and influenza, DNA vaccines are increasingly being investigated for cancer immunotherapy and veterinary uses. Continued government funding, public-private collaboration, and a favorable regulatory environment support this growth and encourage innovation in next-generation vaccine technologies. As DNA vaccine development becomes more mainstream in the U.S. biotech and pharma landscape, the need for reliable, scalable plasmid purification processes is expected to grow substantially over the forecast period.
Rise in Biopharmaceutical R&D
Increasing investment in biopharmaceutical research and development (R&D), particularly in gene therapy, cell therapy, and biologics, is a key driver of growth in the U.S. plasmid purification industry. Plasmid DNA is foundational in these advanced therapies and critical in developing viral vectors, gene editing platforms, and DNA-based therapeutics. As pharmaceutical and biotech companies expand their therapeutic pipelines, there is a rising need for scalable, high-quality plasmid DNA to support preclinical studies, investigational new drug (IND) applications, and clinical trials. This demand has led to a surge in in-house production capabilities and the outsourcing of plasmid purification services across the U.S. market.
Moreover, with the U.S. being a global hub for biotechnology innovation, significant funding from venture capital, federal agencies (such as the NIH), and private-sector partnerships continues to pour into next-generation therapies. As more gene-based and cell-based treatments move through the development pipeline, regulatory agencies like the FDA are streamlining review processes for advanced therapies, creating a more favorable environment for R&D growth. This momentum has directly translated into increased demand for plasmid purification technologies, including advanced chromatography systems and contract manufacturing services, making R&D expansion a major catalyst for market acceleration.
Industry Concentration & Characteristics
The U.S. plasmid purification industry is marked by a high degree of innovation, driven by the rapid advancement of gene and cell therapies. Emerging technologies such as membrane-based chromatography, automated purification systems, and single-use bioprocessing solutions are replacing conventional methods to achieve higher yields, improved purity, and greater scalability. For instance, in June 2024, Thermo Fisher Scientific in the United States launched the KingFisher PlasmidPro Maxi Processor, the first fully automated large‑scale plasmid DNA purification system, streamlining workflows and increasing lab efficiency. These advancements are essential to meet the biopharmaceutical industry's growing regulatory and commercial demands, positioning the U.S. as a plasmid purification technology development leader.
The U.S. plasmid purification industry is witnessing a moderate to high level of M&A activity as key players seek to expand their gene therapy and biomanufacturing capabilities. For instance, in February 2025, Thermo Fisher Scientific acquired Solventum’s Purification and Filtration business in the United States for USD 4.1 billion, enhancing its bioprocessing capabilities and expanding its biologics production portfolio, which reflects strategic efforts to secure advanced purification technologies and end-to-end plasmid DNA production capacity. Such mergers and acquisitions underscore the growing importance of integrated plasmid purification solutions amid rising demand from the gene therapy, vaccine, and biopharma sectors.
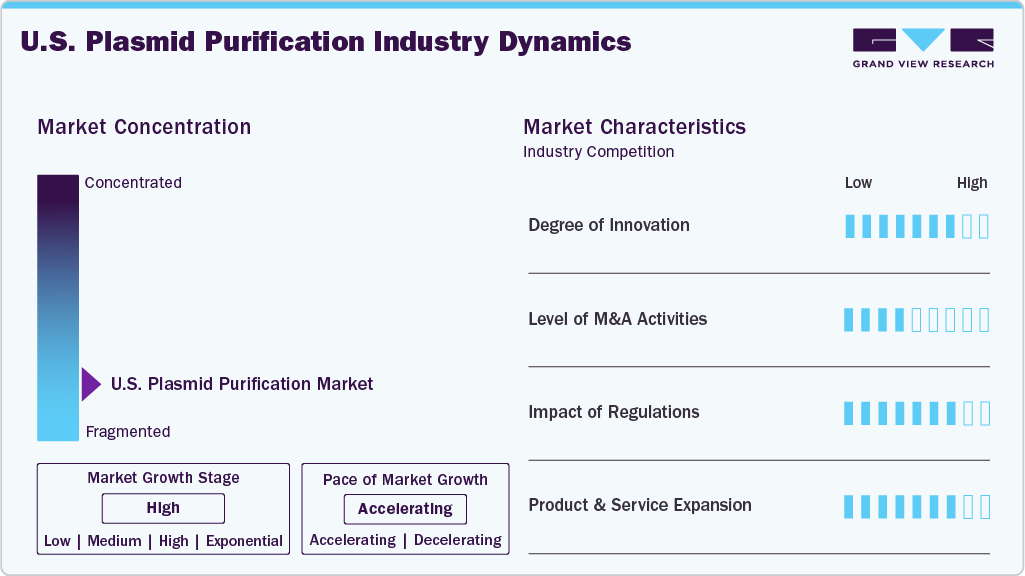
Regulatory frameworks in the U.S. play a critical role in shaping the plasmid purification industry, ensuring the safety, quality, and efficacy of plasmid DNA used in therapeutics and vaccines. The FDA’s stringent guidelines for gene therapy and biologics manufacturing require manufacturers to maintain high purity, consistency, and traceability standards throughout the purification process. Compliance with current Good Manufacturing Practices (cGMP) and evolving regulatory expectations drives purification technologies and process validation innovation. Moreover, expedited approval pathways for advanced therapies encourage investment and commercialization, further boosting demand for reliable and scalable plasmid purification solutions.
Expansion is a significant growth strategy in the U.S. plasmid purification industry, with companies continuously developing new and improved purification kits, reagents, and automated systems to meet the evolving needs of biopharmaceutical research and manufacturing. Innovations focus on enhancing yield, purity, and scalability while reducing processing time and costs. Expansion also offers customizable solutions tailored to specific plasmid types and applications, such as gene therapy, vaccine production, and synthetic biology. Moreover, collaborations between technology providers and contract manufacturing organizations (CMOs) drive broader product portfolios and integrated service offerings, helping companies capture a larger share of the growing market.
Product & Service Insights
The products segment dominated the U.S. market, accounting for the largest revenue share in 2024. This dominance can be attributed to the widespread adoption of plasmid purification kits, reagents, and consumables across research institutions, pharmaceutical companies, and contract manufacturing organizations. These products are essential for ensuring high purity and yield of plasmid DNA, which is critical for applications such as gene therapy, vaccine development, and synthetic biology. Moreover, continuous innovations in product design, including automated purification systems and scalable solutions, have further driven demand and contributed to the segment’s leading market position.
Services is expected to grow at the fastest CAGR throughout the forecast period. This rapid growth is driven by the increasing trend among biopharmaceutical companies to outsource plasmid DNA manufacturing and purification to specialized contract development and manufacturing organizations (CDMOs). Outsourcing enables companies to reduce operational costs, accelerate time-to-market, and access advanced purification technologies without significant capital investment, fueling robust expansion in this segment.
Grade Insights
The molecular-grade segment led the U.S. market with the largest revenue share in 2024, driven by the growing demand for highly pure plasmid DNA required in sensitive applications such as gene therapy, vaccine development, and advanced research. Molecular-grade plasmids meet stringent quality standards, ensuring minimal contaminants and high integrity, critical for clinical and commercial use. The increasing focus on regulatory compliance and the need for consistent, reproducible results in therapeutic development further support the preference for molecular-grade products, solidifying their dominance in the market.
Transfection-grade is expected to grow at the fastest CAGR over the forecast period in the U.S. plasmid purification industry. This rapid growth is driven by the increasing use of transfection-grade plasmid DNA in research applications, including gene expression studies, functional genomics, and drug discovery. As more academic and pharmaceutical research institutes adopt advanced transfection techniques to accelerate early-stage development, the demand for high-quality, reliable transfection-grade plasmids is rising. Moreover, ongoing advancements in transfection reagents and protocols are expected to further propel market growth.
Application Insights
The cloning and protein expression segment led the U.S. market with the largest revenue share of 46.46% in 2024. This leadership is attributed to the widespread use of plasmids in molecular cloning and recombinant protein production, which are foundational techniques in biotechnology and pharmaceutical research. The demand for high-purity plasmid DNA in these applications ensures accurate gene expression and protein synthesis, driving the need for efficient purification products and services.
Transfection and gene editing is expected to grow at the fastest CAGR over the forecast period, owing to the increasing adoption of advanced gene editing technologies such as CRISPR and the rising demand for efficient, non-viral transfection methods. These technologies revolutionize therapeutic development, allowing precise genetic modifications and personalized treatments. Moreover, improvements in transfection reagents and delivery systems make gene editing more accessible and scalable, which drives greater use in research and clinical applications.
End Use Insights
The academic and research institutes segment led the U.S. plasmid purification industry with the largest market share of 40.68% in 2024. This leadership is primarily driven by the extensive use of plasmid purification products and services in basic research, molecular biology studies, and early-stage drug discovery conducted within universities and research centers. These institutions demand high-purity plasmid DNA for applications such as gene cloning, transfection, and gene expression analysis, which further sustains the market demand.

The contract research organizations (CROs) segment is projected to grow at the fastest CAGR from 2024 to 2033. This rapid growth is driven by the increasing trend among pharmaceutical and biotechnology companies to outsource plasmid purification and related services to CROs to reduce costs and accelerate development timelines. CROs offer specialized expertise, advanced purification technologies, and scalable manufacturing capabilities, making them attractive partners for gene therapy, vaccine development, and other plasmid-dependent applications. As demand for plasmid DNA continues to rise, especially in gene and cell therapy pipelines, CROs are poised to capture a growing market share. For instance, in June 2024, Bionova Scientific, a U.S.-based biologics CDMO under Asahi Kasei, announced plans to establish a new facility in Texas dedicated to plasmid DNA services, aligning with the company's growth strategy.
Key U.S. Plasmid Purification Company Insights
The U.S. market is dominated by several established players who leverage robust product portfolios, strategic collaborations, and sustained investments in research and development. Leading companies such as Merck KGaA, QIAGEN, Thermo Fisher Scientific, Inc., and Takara Bio hold significant market shares by offering a comprehensive range of purification kits, reagents, and automated systems tailored to diverse research and biomanufacturing needs.
Industry leaders, including Promega Corporation, Zymo Research, MP BIOMEDICALS, and New England Biolabs, continue strengthening their position through innovative product development and expanding service capabilities. These organizations address the growing demand for high-purity plasmid DNA essential for gene therapy, vaccine development, synthetic biology, and molecular biology research. Their end-to-end offerings, from small-scale purification kits to scalable automated platforms, enable seamless workflows for academic, clinical, and industrial users.
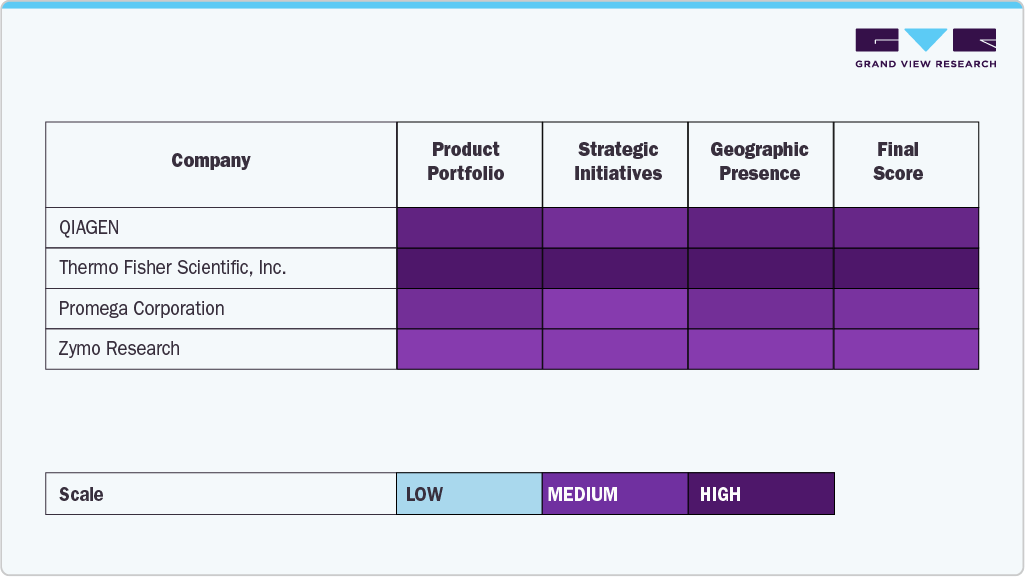
The U.S. market is also witnessing a vibrant interplay between established firms and emerging companies like MCLAB and Applied Biological Materials Inc., fostering innovation and competitive differentiation. Mergers and acquisitions, strategic partnerships, and technological breakthroughs are accelerating market evolution, intensifying competition across the sector. As demand for precision medicine and advanced biotherapeutics rises, market participants emphasizing product quality, scalability, and customer-centric solutions will be best positioned to drive sustained growth and shape the future of plasmid purification in the U.S.
Key U.S. Plasmid Purification Companies:
- Merck KGaA
- QIAGEN
- Thermo Fisher Scientific, Inc.
- Takara Bio
- Promega Corporation
- Zymo Research
- MP BIOMEDICALS
- New England Biolabs
- MCLAB
- Applied Biological Materials Inc.
Recent Developments
-
In May 2025, Thermo Fisher Scientific introduced the Applied Biosystems MagMAX Pro HT NoSpin Plasmid MiniPrep Kit in the United States, designed to streamline small-scale plasmid DNA purification. This kit enables the processing of 96 samples from just 1 milliliter of culture in under 35 minutes, eliminating manual steps like centrifugation and offline lysis. Utilizing magnetic bead technology and eco-friendly chemistry, it enhances scalability and reduces reagent waste, making it suitable for high-throughput systems and accelerating research workflows.
-
In February 2025, Omega Bio-tek introduced its Mag-Bind Endo-Free Plasmid Mini and Midi Kits in the United States. These kits utilize magnetic bead-based technology for endotoxin-free plasmid DNA purification, achieving levels below 0.1 EU/µg. Designed for automation, they eliminate manual centrifugation steps, streamlining workflows for applications such as gene editing and molecular diagnostics.
U.S. Plasmid Purification Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 776.9 million
Revenue forecast in 2033
USD 1.84 billion
Growth rate
CAGR of 11.41% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product & service, grade, application, end use
Key companies profiled
Merck KGaA; QIAGEN; Thermo Fisher Scientific, Inc.; Takara Bio; Promega Corporation; Zymo Research; MP BIOMEDICALS; New England Biolabs; MCLAB; Applied Biological Materials Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
U.S. Plasmid Purification Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the U.S. plasmid purification market based on product & service, grade, application, and end use:
-
Product & Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Products
-
Instruments
-
Kits & Reagents
-
-
Services
-
-
Grade Outlook (Revenue, USD Million, 2021 - 2033)
-
Molecular Grade
-
Transfection Grade
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Cloning & Protein Expression
-
Transfection & Gene Editing
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biotechnology Companies
-
Academic & Research Institutes
-
Contract Research Organizations
-
Frequently Asked Questions About This Report
b. The U.S. plasmid purification market size was estimated at USD 699.4 million in 2024 and is expected to reach USD 776.9 million in 2025.
b. The U.S. plasmid purification market is expected to grow at a compound annual growth rate of 11.41% from 2025 to 2033 to reach USD 1.84 billion by 2033.
b. The cloning & protein expression segment dominated the U.S. plasmid purification market with a share of 46.46% in 2024. This is attributable to its widespread use in research, biopharmaceutical production, and the development of recombinant proteins and therapeutic enzymes.
b. Some key players operating in the U.S. plasmid purification market include Merck KGaA; QIAGEN; Thermo Fisher Scientific, Inc.; Takara Bio; Promega Corporation; Zymo Research; MP BIOMEDICALS; New England Biolabs; MCLAB; Applied Biological Materials Inc.
b. Key factors driving the market growth include rising demand for gene therapies, DNA vaccines, biologics development, and increased research in molecular biology, genetic engineering, and personalized medicine.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










