- Home
- »
- Clinical Diagnostics
- »
-
U.S. Point of Care Molecular Diagnostics Market Report, 2033GVR Report cover
![U.S. Point of Care Molecular Diagnostics Market Size, Share & Trends Report]()
U.S. Point of Care Molecular Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Test Location (OTC, POC), By Application (Infectious Diseases, Oncology, Prenatal Testing), By Technology, By End-use (Decentralized Labs, Hospitals), And Segment Forecasts
- Report ID: GVR-4-68040-203-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
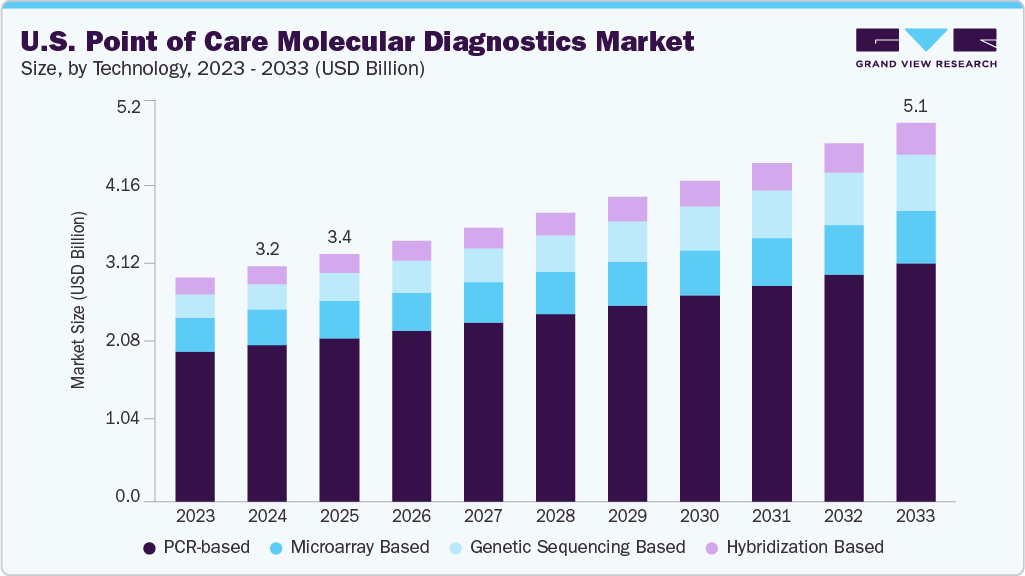
The U.S. point of care molecular diagnostics market size was estimated at USD 3.23 billion in 2024 and is expected to grow at a CAGR of 5.45% from 2025 to 2033. The market is centered around rapid and decentralized molecular testing that supports quicker clinical decisions at or near the site of patient care. These diagnostic platforms are increasingly adopted for applications such as infectious disease screening, respiratory tract infections, and sexually transmitted disease detection, driven by the need for immediate and accurate results in outpatient and emergency settings.
Market growth is supported by the rising demand for quick and easy testing outside traditional labs, especially in emergency rooms and clinics. The growing number of infectious disease cases and the need for fast results are pushing the use of point-of-care molecular tests. New technologies, such as portable PCR machines and simple sample processing tools, make these tests more accurate and easier to use. In addition, the focus on faster testing after the COVID-19 pandemic is helping the market grow further.
Moreover, point-of-care molecular diagnostics in primary care settings cover a wide range of uses, from basic glucose tests to more complex blood clotting checks. Many clinics across the U.S. are moving away from traditional lab-based testing to point-of-care methods, which help reduce delays linked to sample handling and transport. This switch allows for quicker decisions during patient visits, leading to faster results, lower costs, and improved care.
In addition, the growing elderly population is expected to support the growth of the U.S. point of care molecular diagnostics industry. Based on the U.S. National Cancer Institute’s SEER Database, 38% of women and 43% of men are likely to develop cancer during their lifetime. Nearly two-thirds of all new cancer cases are found in people aged 65 and older, showing that aging increases vulnerability to cancer. Molecular diagnostics play a key role in managing cancer, infectious diseases, and heart conditions, making them an essential part of timely diagnosis and treatment in older adults.
Furthermore, market players are actively developing new point-of-care molecular testing products to address growing diagnostic needs. In April 2023, Curative, Inc., based in Los Angeles, announced the spin-off of Sensible Diagnostics to commercialize a desktop PCR testing platform. This system is designed to deliver lab-quality results in approximately 10 minutes, at a cost similar to lateral flow antigen tests. It is intended for use in retail clinics, urgent care centers, and other near-patient environments.
In August 2023, Sensible Diagnostics received a NIH RADx Tech award of approximately USD 1 million to support the development of a multiplex respiratory viral panel for point-of-care use. These efforts reflect growing investment in rapid, accurate molecular testing solutions that support timely diagnosis and improve clinical workflows across decentralized care settings.
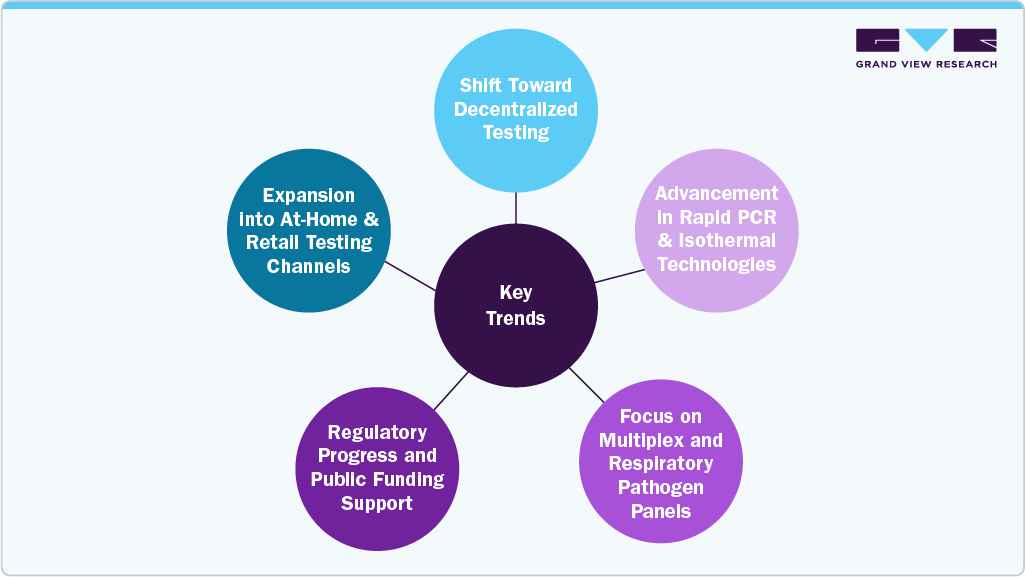
Key Trends Shaping the U.S. Point of Care Molecular Diagnostics Market
1. Shift Toward Decentralized Testing: There is a growing demand for diagnostics that can be performed at or near the patient site. Point-of-care molecular platforms are increasingly used in outpatient clinics, urgent care centers, and even home settings to reduce delays, enable immediate decisions, and minimize lab dependency.
2. Advancement in Rapid PCR and Isothermal Technologies: Improved molecular platforms such as portable PCR devices and isothermal amplification systems are making high-sensitivity testing possible with shorter turnaround times. These innovations are helping replace traditional, slower lab-based methods in many frontline care environments.
3. Focus on Multiplex and Respiratory Pathogen Panels: Post-pandemic healthcare has highlighted the need for rapid testing of multiple respiratory pathogens. Companies are developing multiplex molecular assays that detect infections like COVID-19, influenza, and RSV simultaneously, supporting efficient diagnosis and treatment decisions.
4. Regulatory Progress and Public Funding Support: Recent developments such as the U.S. FDA’s evolving oversight of diagnostic tools and the NIH RADx Tech program are encouraging product innovation. Startups and established players alike are receiving support for novel platforms targeting respiratory, STI, and infectious disease detection.
5. Expansion into At-Home and Retail Testing Channels: Molecular diagnostics are moving beyond traditional clinical settings. The launch of FDA EUA-approved at-home molecular tests and efforts to bring devices into pharmacies and retail clinics reflect the market’s push toward convenience, self-testing, and wider access.
Market Concentration & Characteristics
The U.S. point of care molecular diagnostics market is defined by rapid innovation in portable testing technologies, miniaturized platforms, and integrated sample-to-answer systems. Developments in isothermal amplification, microfluidic cartridges, and handheld PCR devices are improving test speed and accuracy. Companies are also introducing multiplex assays that detect multiple pathogens from a single sample. These advances are making molecular diagnostics more accessible across primary care, urgent care, and at-home settings, aligning with the broader goals of decentralized and patient-centered healthcare.
Mergers and acquisitions in this space are focused on strengthening molecular testing capabilities and expanding presence in non-traditional care settings. U.S. companies are acquiring startups with proprietary rapid PCR platforms, multiplex respiratory panels, and digital integration tools. These deals aim to accelerate product pipelines, enter high-demand segments like respiratory and STI testing, and broaden access through retail clinics and telehealth-enabled diagnostics. Strategic investments are also directed toward enhancing throughput and developing platforms suitable for emergency and field-based use.
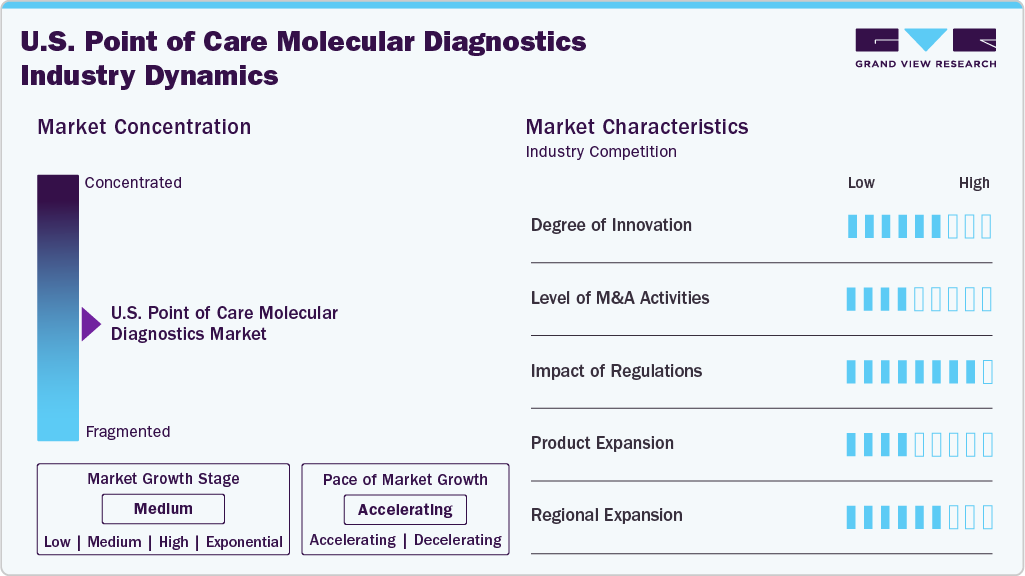
Regulatory oversight has a significant impact on market dynamics. The U.S. FDA plays a central role through 510(k) clearances and Emergency Use Authorizations (EUAs), particularly for infectious disease panels. The market is also shaped by recent efforts to expand FDA regulation over lab-developed tests (LDTs), which are frequently used in decentralized settings. Reimbursement frameworks under CMS and private payers influence test accessibility and adoption. Companies must navigate these regulatory landscapes to ensure compliance while supporting innovation and timely market entry.
Diagnostic providers are expanding their offerings to cover a wider range of conditions using point-of-care molecular platforms. Beyond COVID-19, test menus now span influenza, RSV, chlamydia, gonorrhea, and other infections. Platforms are being adapted for multiplex testing and use in pharmacy clinics, employer health programs, and ambulatory care. Companies are also investing in mobile-friendly result delivery systems and clinical decision support tools to improve test utility and integration into care workflows.
Efforts to expand reach are focused on rural, underserved, and retail-based care locations. Companies are deploying compact molecular systems in areas with limited lab access, supported by mail-in sample kits, mobile diagnostics units, and collaborations with local health networks. These strategies aim to reduce diagnostic delays and improve care access in geographically dispersed populations. Retail chains and telemedicine platforms are also emerging as important distribution channels for point-of-care molecular testing in the U.S. market.
Application Insights
The infectious diseases segment accounted for a notable share of the U.S. point of care molecular diagnostics industry in 2024, supported by ongoing public health efforts to manage conditions such as respiratory infections, sexually transmitted diseases, and viral outbreaks. Despite advancements in centralized testing, there remains a strong demand for accurate, fast, and cost-effective molecular diagnostics at the point of care. The continued focus on early detection and control of infectious diseases across outpatient and decentralized settings is expected to support segment growth.
The other applications segment held the largest market share in 2024. This segment includes a broad range of use cases such as fertility and pregnancy testing, nephrology, autoimmune disorders, and cardiac markers. The expanding use of point-of-care molecular tools in non-infectious disease areas is driven by growing awareness among patients and providers regarding the convenience and speed these diagnostics offer. As these tests often require minimal clinical intervention, the segment is expected to maintain strong momentum throughout the forecast period.
The oncology segment is anticipated to witness the fastest growth over the forecast period. Molecular testing tools are being explored for early cancer detection in decentralized settings such as community clinics and outreach programs. These efforts aim to reduce turnaround times and improve access to screening for populations with limited access to centralized laboratories. Growth potential is further supported by federal initiatives such as the National Breast and Cervical Cancer Early Detection Program, which promotes access to timely screening services. As interest in early intervention and decentralized cancer diagnostics expands, the adoption of point-of-care molecular platforms in oncology is expected to increase.
Test Location Insights
In 2024, the over-the-counter (OTC) diagnostics segment held the highest share of the U.S. point of care molecular diagnostics market. These products support early detection and ongoing health monitoring without requiring visits to healthcare facilities, helping reduce overall medical costs. As demand for point-of-care molecular testing continues to rise, there is a strong need for user-friendly platforms that allow individuals to perform tests independently.
In response, companies are launching innovative at-home molecular testing kits for conditions such as COVID-19, influenza, and other infections. For example, in March 2023, Lucira Health introduced the first FDA-authorized at-home test that detects both COVID-19 and flu in the U.S., improving access to timely diagnosis. Such developments are boosting the growth of the OTC diagnostics segment by making self-testing more convenient and widely accessible.
The point-of-care test location segment is projected to register the fastest CAGR over the forecast period. These molecular tests detect viruses and other infectious agents through their genetic material, offering rapid results in portable, near-patient formats. While most oncology diagnostics remain centralized, early efforts are underway to develop point-of-care molecular tests for select cancers, such as cervical and colorectal cancer, particularly through HPV and DNA-based screening approaches.
A key example of support for this trend is the funding awarded in October 2023, when the NIH’s National Institute of Biomedical Imaging and Bioengineering (NIBIB) granted USD 7.8 million over five years to the Atlanta Center for Microsystems Engineered Point-of-Care Technologies (ACME POCT). This investment aims to accelerate the development and clinical translation of microsystems-based point-of-care diagnostic platforms across the U.S.
Technology Insights
PCR-based technologies held the largest share of the U.S. point of care molecular diagnostics industry in 2024, driven by their high sensitivity, proven reliability, and vast clinical applicability. As the gold standard for DNA amplification, PCR remains the most commonly used method for detecting infectious diseases such as COVID-19, influenza, and HIV. Its applications are expanding in cancer monitoring, drug response testing, and pharmacogenomics, although these remain more common in centralized laboratory environments. The development of compact PCR platforms and advancements such as multiplex PCR have improved portability and efficiency, making these tools well-suited for near-patient use. These factors continue to reinforce the dominance of PCR-based diagnostics in decentralized settings.
The genetic sequencing-based point-of-care diagnostics segment is expected to grow at the fastest CAGR during the forecast period. This technology supports real-time analysis of genetic material, allowing for early disease detection and more tailored treatment decisions. The rising demand for rapid and accurate molecular testing and the increasing burden of infectious and chronic diseases support this segment’s growth. Recent improvements are making sequencing platforms more adaptable to smaller healthcare environments, though widespread use in point-of-care settings is still developing. The broader shift toward personalized medicine and growing interest in genomic screening further contribute to the expanding potential of sequencing-based diagnostics at the point of care.
End-use Insights
The decentralized segment held the largest share of the U.S. point of care molecular diagnostics market in 2024. These platforms support faster clinical decision-making, reduce turnaround times, and eliminate delays associated with centralized laboratory testing. Their widespread use in urgent care centers, retail clinics, and community health settings reflects a broader shift toward near-patient diagnostics that reduce costs and improve access to timely care.
A notable development occurred in July 2024, when Roche completed the acquisition of LumiraDx’s point-of-care technology for up to USD 350 million. LumiraDx’s platforms are already in use across various decentralized healthcare settings in the U.S., including CLIA-waived locations and retail clinics. Many of these systems had previously received U.S. FDA Emergency Use Authorization for rapid testing of conditions such as COVID-19. Roche aims to build on this foundation by expanding decentralized testing options for outpatient, pharmacy, and mobile care environments. This move supports the segment’s continued growth and relevance in the U.S. market.
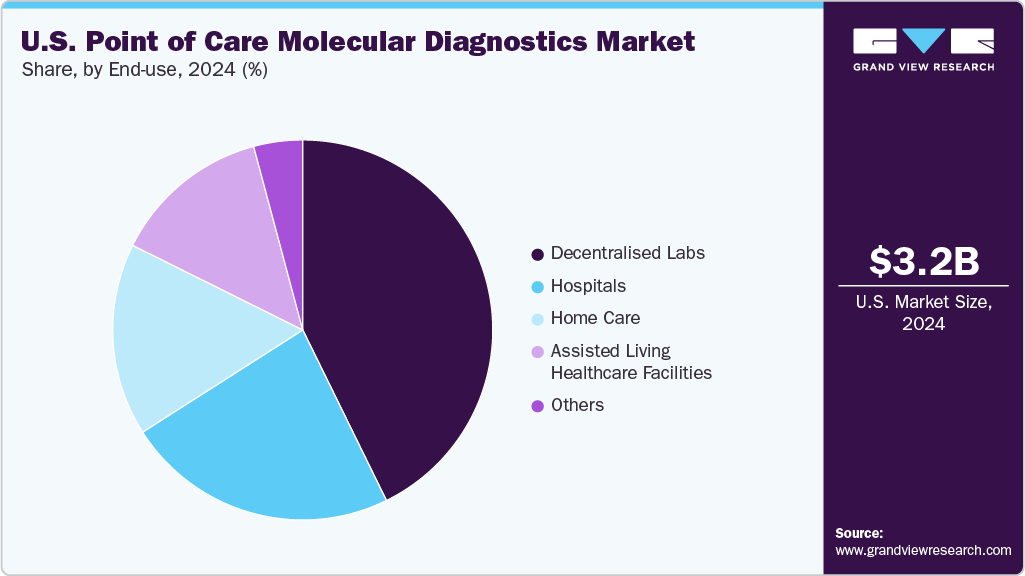
The home care segment is expected to grow at the fastest CAGR over the forecast period, driven by the ease and affordability of point-of-care molecular diagnostics at home. These tests allow individuals to monitor their health, receive quick results, and make informed decisions without needing to visit a clinic or hospital. Most POC devices designed for home use are simple to operate and do not require any specialized equipment, making them well-suited for self-testing.
This segment is particularly valuable for conditions that may carry social stigma, such as sexually transmitted infections, where privacy and convenience are key concerns. As the U.S. healthcare landscape continues to emphasize early detection and preventive care, home-based POC molecular diagnostics are expected to see growing adoption. These solutions offer patients greater control over their health while reducing the burden on traditional healthcare facilities.
Key U.S. Point of Care Molecular Diagnostics Company Insights
Key players in the U.S. point of care molecular diagnostics market are actively focusing on new product development and obtaining regulatory approvals to broaden their diagnostic portfolios. In addition, companies are pursuing strategic initiatives such as collaborations, partnerships, and acquisitions to reinforce their market position. These efforts are directed at enhancing technological capabilities, expanding access to decentralized testing, and meeting growing demand for rapid diagnostic solutions across outpatient clinics, retail health centers, and home care environments.
Key U.S. Point of Care Molecular Diagnostics Companies:
- QIAGEN
- Danaher
- Thermo Fisher Scientific, Inc.
- BD
- F. Hoffman-La Roche AG
- Charles River Laboratories
- Quest Diagnostics Incorporated
- Bio-Rad Laboratories, Inc.
- Hologic Inc.
- Agilent Technologies, Inc.
Recent Developments
-
In February 2024, BD, a U.S.-based medical technology company, announced that they have begun clinical trials for a rapid Point-of Care Molecular Dx instrument and an assay to detect STIs.
-
In November 2023, binx health, a healthcare technology and diagnostics company, announced its agreement with Fisher Healthcare, a part of Thermo Fisher Scientific, to expand the nationwide distribution of its molecular POC platform, bimx io, in the U.S.
-
In March 2023, Lucira Health, a U.S.-based company that Pfizer acquired in April 2023, announced the launch of the first and only at-home COVID-19 and flu tests in the U.S., a test approved by the FDA under Emergency Use Authorization (EUA) for OTC usage at home and other nonlaboratory locations.
U.S. Point of Care Molecular Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 3.40 billion
Revenue forecast in 2033
USD 5.19 billion
Growth rate
CAGR of 5.45% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test location, application, technology, end-use
Key companies profiled
QIAGEN; Danaher; Thermo Fisher Scientific, Inc.; BD; F. Hoffman-La Roche AG; Charles River Laboratories; Quest Diagnostics Incorporated; Bio-Rad Laboratories, Inc.; Hologic Inc.; Agilent Technologies, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
U.S. Point of Care Molecular Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the U.S. point of care molecular diagnostics market report based on application, technology, test location, and end-use:
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
PCR-based
-
Genetic Sequencing-based
-
Hybridization-based
-
Microarray-based
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Infectious Diseases
-
HIV POC
-
Clostridium difficile POC
-
HBV POC
-
Pneumonia or Streptococcus associated infections
-
Respiratory syncytial virus (RSV) POC
-
HPV POC
-
Influenza/Flu POC
-
HCV POC
-
MRSA POC
-
TB and drug-resistant TB POC
-
HSV POC
-
Other Infectious Diseases
-
-
Oncology
-
Hematology
-
Complete blood count (CBC)
-
Prothrombin time (PT)
-
Partial Thromboplastin Time (PTT)
-
Others
-
-
Prenatal Testing
-
Endocrinology
-
Other Applications
-
-
Test Location Outlook (Revenue, USD Million, 2021 - 2033)
-
OTC
-
POC
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Decentralized Labs
-
Hospitals
-
Home-care
-
Assisted Living Healthcare Facilities
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. point of care molecular diagnostics market is estimated at USD 3.23 billion in 2024 and is expected to reach USD 3.40 billion in 2025.
b. The U.S. point of care molecular diagnostics market is expected to grow at a CAGR of 5.45% from 2025 to 2033 to reach USD 5.19 billion in 2033.
b. The OTC segment held a marginally higher share of 52.59% in the U.S. and is anticipated to register a significant growth rate from 2025 to 2033.
b. Some of the key players operating in the U.S. point-of-care molecular diagnostics market include Abbott Laboratories; QIAGEN; F. Hoffman-La Roche Ltd; bioMerieux; and BD.
b. The growth of this market is driven by a rise in end-user awareness, along with the increased efficiency of the testing methods and favorability for non-laboratory testing.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










