- Home
- »
- Medical Devices
- »
-
U.S. Ureteral Access Sheath Market, Industry Report 2033GVR Report cover
![U.S. Ureteral Access Sheath Market Size, Share & Trends Report]()
U.S. Ureteral Access Sheath Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Type (Standard, Reinforced, Dual-Lumen), By Clinical Indication, By Age Group, By Size, By End-use (Hospitals, Specialty Clinics, Diagnostic Centers), And Segment Forecasts
- Report ID: GVR-4-68040-874-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Ureteral Access Sheath Market Summary
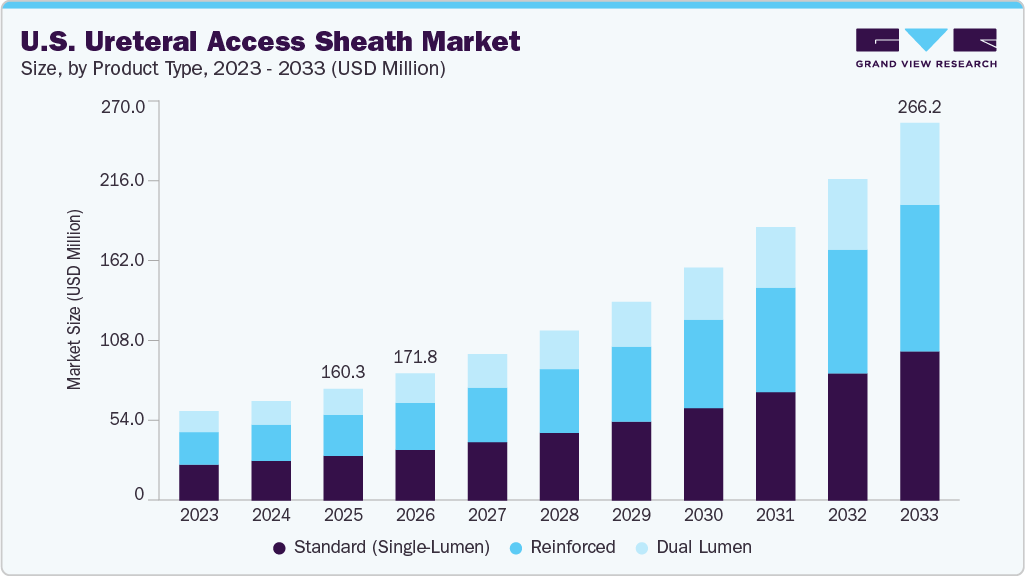
The U.S. ureteral access sheath market size was estimated at USD 160.34 million in 2025 and is projected to reach USD 266.19 million by 2033, growing at a CAGR of 6.5% from 2026 to 2033. The market is primarily driven by the rising prevalence of urolithiasis and other urological disorders, which have increased the volume of ureteroscopy procedures in the U.S.
Key Market Trends & Insights
- By product type, the standard (single lumen) segment led the market with the largest revenue share in 2025.
- By size, 10 to 12 segment led the market with the largest revenue share in 2025.
- By age group, the adult segment led the market with the largest revenue share in 2025.
- By clinical indication, the stone management segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 160.34 Million
- 2033 Projected Market Size: USD 266.19 Million
- CAGR (2026-2033): 6.5%
The growing adoption of minimally invasive surgeries, coupled with technological advancements that improve procedural efficiency, safety, and reduce operative time, is further accelerating market growth. According to the National Kidney Foundation, more than 500,000 people visit emergency rooms each year due to kidney stones, highlighting the significant clinical burden of the condition. In nearly 1 in 10 individuals, a kidney stone is expected to develop at some point during their lifetime.The U.S. ureteral access sheath market is experiencing significant growth, driven by the increasing prevalence of kidney stone disease and the widespread adoption of minimally invasive urological procedures. Ongoing innovations in sheath design and materials are improving procedural safety and efficiency, encouraging higher clinical adoption. In addition, strong healthcare infrastructure, favorable reimbursement policies, and the growing shift toward outpatient urology procedures continue to support the market expansion.
The AUA 2026 kidney stone surgery guideline is expected to support the growth of the U.S. ureteral access sheath market. Ureteroscopy (URS) is a key treatment that supports the use of ureteral access sheaths during URS procedures. The guideline recommends URS as a primary treatment option for many kidney and ureteral stones. It allows the use of ureteral access sheaths, including advanced suction-based designs, during URS, thereby increasing clinical adoption of UAS devices. The U.S. already performs a high and growing volume of stone surgeries, with ureteroscopy procedures rising significantly over the last decade, creating sustained procedural demand for access tools such as UAS.
AUA 2026 Kidney Stone Surgery Guideline - Key Recommendations Summary Table
Category
Recommendation
Evidence Grade
Market / Device Relevance
Flexible Ureteroscope Use
Single-use or reusable both recommended
Grade A
Supports ureteroscope market expansion
Ureteral Access Sheath (UAS)
Placement recommended during URS
Grade B
Direct clinical validation for UAS adoption
Flexible Suction UAS
Considered acceptable option
Grade C
Supports next-gen UAS innovation market
Laser Lithotripsy
Holmium:YAG or Thulium fiber laser allowed
Grade C
Supports laser + accessory ecosystem
Antibiotics Before URS & PCNL
Recommended
Grade B
Supports infection control consumables
Post-URS Stent
Not mandatory after uncomplicated URS
Grade C
Slightly reduce stent usage volume
Emerging Technologies
Suction retrograde systems, robotics, hybrid lasers
Emerging
Supports future UAS + integrated system growth
Upgrade report license to gain access to the complete analysis.
Source: American Urological Association Education and Research, Inc., 2026
The rising prevalence of urolithiasis is a key driver of growth in the ureteral access sheath market, as increasing stone cases directly increase the number of ureteroscopic procedures. Ureteral access sheaths are essential in stone management, enabling efficient scope exchanges and improved intrarenal pressure control. As kidney stone incidence continues to grow due to lifestyle and dietary changes, demand for advanced ureteral access devices is expected to increase significantly. According to the National Kidney Foundation, in 2025, approximately 1 in 10 people have experienced a kidney stone at some point in their lives. The condition is slightly more common among men, with about 11 out of every 100 affected, compared to around 9 out of every 100 women.
The growing adoption of minimally invasive urological procedures is a key driver of the ureteral access sheath market, as these techniques depend on safe and efficient ureteral access. Procedures such as ureteroscopy, retrograde intrarenal surgery (RIRS), and laser lithotripsy use access sheaths to facilitate smooth instrument exchange and improve intrarenal pressure control. The shift toward shorter hospital stays, reduced complications, and faster recovery continues to accelerate demand for ureteral access sheaths.
Technological advancements are significantly driving the ureteral access sheath market by improving procedural safety, efficiency, and clinical outcomes. Innovations such as enhanced flexible materials, hydrophilic coatings, pressure-controlled and dual-lumen designs reduce ureteral trauma and maintain optimal intrarenal pressure. These developments support wider adoption of ureteral access sheaths across complex and minimally invasive urological procedures.
For instance, in August 2025, the Journal of the Formosan Medical Association reported that urolithiasis remains a prevalent and economically burdensome condition worldwide, with a high recurrence rate. Advances in flexible ureteroscopes (fURS) and high-power holmium laser (HPHL) technologies have broadened clinical indications, positioning retrograde intrarenal surgery (RIRS) as a preferred treatment for complex upper urinary tract stones. The introduction of flexible and navigable suction (FANS) ureteral access sheaths (UAS) has further improved outcomes by enabling effective intrarenal pressure control and high stone-free rates, with minimal serious adverse events. However, alongside these clinical benefits, the rising costs of disposable fURS and HPHL systems are emerging as a significant concern in contemporary stone management.
Market Concentration & Characteristics
The ureteral access sheath market is moderately concentrated, with a few established medical device companies holding a significant share due to strong brand presence, regulatory expertise, and broad distribution networks. The market is characterized by continuous product innovation, strict regulatory compliance, and a focus on minimally invasive urological procedures.
The degree of innovation in the ureteral access sheath market is advancing rapidly, with manufacturers focusing on enhanced materials, improved flexibility, and ergonomic designs to support safer and more efficient ureteroscopic procedures. Innovations such as dual-lumen configurations, pressure-controlled systems, and coatings that reduce friction are improving clinical outcomes and expanding procedural applications. These technological advancements are driving greater adoption among urologists and fueling competitive growth within the market.

Regulatory frameworks significantly impact the ureteral access sheath market by ensuring product safety, efficacy, and quality through stringent approval processes such as FDA clearance. Compliance with evolving medical device regulations increases development costs and time-to-market and builds clinician and patient confidence in these devices. In addition, post-market surveillance and reporting requirements identify and mitigate potential risks, further shaping competitive dynamics and innovation strategies within the market.
The ureteral access sheath market has seen moderate merger and acquisition activity as larger medical device companies seek to expand their urology portfolios and gain access to innovative technologies. Strategic acquisitions allow companies broaden product offerings, enhance geographic reach, and leverage established distribution networks. This consolidation trend is expected to continue as firms aim to strengthen competitive positioning and capitalize on growing demand for advanced urological solutions.
In January 2026, Boston Scientific announced an agreement to acquire Valencia Technologies Corporation to expand its urology portfolio, particularly in overactive bladder (OAB) treatment. The Boston Scientific acquisition of Valencia Technologies reflects the broader expansion of minimally invasive urology treatments, which, in turn, supports the ureteral access sheath market. As urology procedure volumes increase and healthcare providers invest more in advanced endoscopic infrastructure, procedures such as RIRS are expected to grow. Since ureteral access sheaths are routinely used during RIRS to enable safe and repeated scope access, improving procedural efficiency and safety, the rising adoption of minimally invasive urology interventions is expected to drive demand for UAS products.
The ureteral access sheath market is experiencing rapid product expansion, with companies introducing sheaths featuring advanced materials, enhanced flexibility, and improved lumens to meet diverse clinical needs. Newer offerings focus on reducing ureteral trauma, optimizing irrigation flow, and supporting a wider range of endoscopic instruments. This continuous innovation and broadening of product portfolios are assisting manufacturers capture more market share and address evolving surgeon preferences.
Product Type Insights
The standard (single lumen) segment dominates the ureteral access sheath market in 2025, due to its widespread use in routine ureteroscopy procedures, particularly for stone management. These sheaths are preferred for their simpler design, cost-effectiveness, and proven clinical reliability across a broad patient population. Their compatibility with most ureteroscopes and ease of handling continue to support strong adoption in both hospital and ambulatory settings.
The dual-lumen segment in the ureteral access sheath market is expected to grow faster during the forecast period, driven by its ability to provide simultaneous irrigation and pressure management during complex ureteroscopy procedures. These sheaths enhance visibility, improve stone clearance efficiency, and reduce intrarenal pressure, supporting better clinical outcomes. Increasing adoption in advanced stone management and complex urological cases is further accelerating the growth of the dual-lumen segment.
Size Insights
The 10–12 Fr segment dominated in ureteral access sheath market in 2025, as this size range offers an optimal balance between adequate working channel access and patient safety. These sheaths are widely used in standard ureteroscopy stone management procedures because they are compatible with most flexible ureteroscopes. Their broad clinical acceptance and suitability for adult patients continue to support strong market dominance.
The <10 Fr segment of the ureteral access sheath market is expected to grow faster during the forecast period, driven by rising demand for minimally invasive and pediatric urological procedures. Smaller-diameter sheaths reduce ureteral trauma and postoperative complications, making them increasingly preferred in delicate and complex cases. Technological advancements that enable greater flexibility and durability in thinner sheaths are further supporting the growth of this segment.
Age Group Insights
The adult segment dominated the ureteral access sheath market in 2025, largely due to the higher prevalence of urolithiasis and other urological disorders among adults. Adults account for the majority of ureteroscopy procedures, where access sheaths are routinely used to improve procedural efficiency and safety. In addition, lifestyle factors and age-related risks continue to drive greater demand for ureteral interventions in this segment.
The pediatric segment of the ureteral access sheath market is expected to grow faster during the forecast period, driven by rising diagnoses of pediatric urolithiasis and congenital urological disorders. Growing awareness among clinicians, along with advancements in smaller-diameter and more flexible access sheaths designed specifically for children, is supporting adoption. In addition, rising preference for minimally invasive pediatric urological procedures is further accelerating growth in this segment.
Clinical Indication Insights
The stone management segment dominates the ureteral access sheath market in 2025 and is also expected to grow fastest during the forecast period. The semgne growth is driven by the high burden of kidney and ureteral stones and the rising volume of ureteroscopic procedures. Ureteral access sheaths are widely used in stone removal and laser lithotripsy, as they facilitate smooth instrument exchange and maintain low intrarenal pressure. Their ability to enhance procedural efficiency, reduce operative time, and lower complication risks continues to drive strong adoption in stone management applications.
The other segment in the ureteral access sheath market is witnessing significant growth, supported by expanding applications beyond stone management, including tumor biopsies, ureteral strictures, and diagnostic ureteroscopy. Increasing adoption of minimally invasive urological procedures and improved sheath designs that enhance flexibility and access are contributing to this growth. In addition, rising clinician preference for versatile devices across multiple indications is accelerating the segment's expansion.
End-use Insights
Hospitals segment dominate the ureteral access sheath market in 2025, due to their role as the primary centers for managing complex urological conditions and performing high-volume endourological procedures such as ureteroscopy. These settings are equipped with advanced surgical infrastructure, imaging systems, and skilled urologists, enabling safe and efficient use of ureteral access sheaths. In addition, the rising number of inpatient and outpatient kidney stone procedures, along with the preference for minimally invasive surgery, further strengthens hospital demand for these devices.
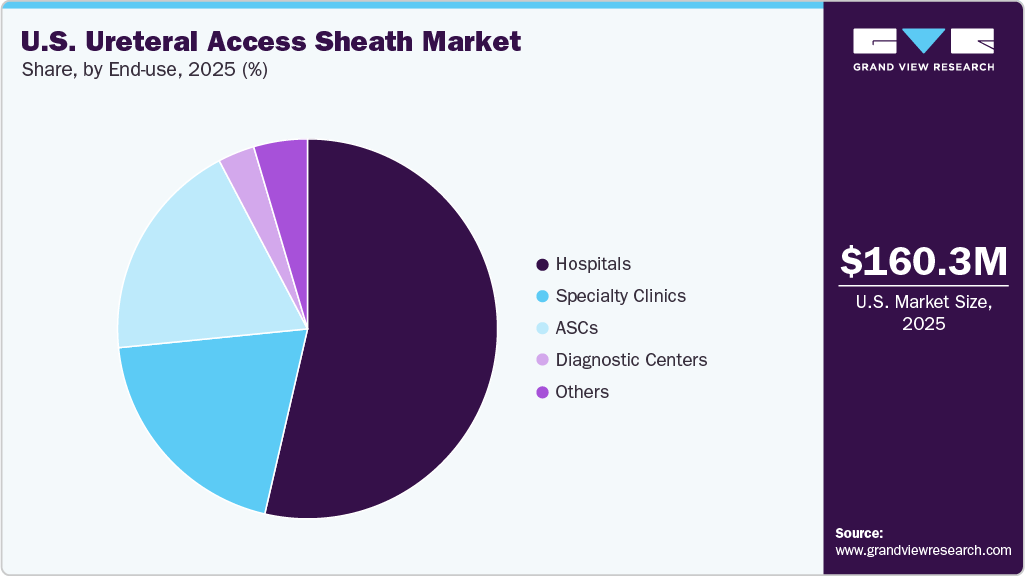
The ambulatory surgery centers (ASCs) segment is expected to grow at the fastest CAGR during the forecast period, driven by the increasing shift of urological procedures from hospitals to outpatient settings. ASCs offer cost-effective treatment, shorter procedure times, and faster patient recovery, making them an attractive option for minimally invasive ureteroscopic interventions. The growing adoption of advanced endourology technologies and favorable reimbursement policies further support the rapid expansion of this segment.
Key U.S. Ureteral Access Sheath Company Insights
The U.S. ureteral access sheath market is highly competitive, with key players holding significant positions. The major companies are undertaking various organic as well as inorganic strategies such as new product development, collaborations, acquisitions, mergers, and regional expansion for serving the unmet needs of their customers.
Key U.S. Ureteral Access Sheath Companies:
- Boston Scientific Corporation
- BD
- Olympus
- Coloplast
- Cook Medical
- Advin Health Care
- ACE Medical Devices
- Blue Neem Medical Devices Private Limited
- AMECATH
- Applied Medical Resources Corporation
- Volkmann Medizintechnik Gmbh.
- Urovision-Urotech
- Rocamed SAM
- Well Lead Medical Co.,Ltd.
- Richard Wolf GmbH
- SCW Medicath Ltd
Recent Developments
-
In September 2025, Olympus Corporation announced an exclusive global distribution partnership with MacroLux Medical Technology to expand its single-use urology product portfolio. The agreement covers single-use cystoscopes, ureteroscopes, and suction access sheaths used to diagnose and treat urinary tract conditions, such as kidney stones.
-
In September 2025, Dornier MedTech announced the full U.S. commercial launch of two single-use urology devices: The Hoover Flexible and Navigable Suction Ureteral Access Sheath and the Axis II Slim ureteroscope. The Hoover sheath enables continuous irrigation, suction, and instrument exchange during stone procedures, improving visibility and reducing procedure time.
“Dornier MedTech America has a deep understanding of urological workflows and urologists’ needs. The Dornier Hoover delivers best-in-class suction performance and Dornier Axis II Slim offers best-in-class deflection-giving urologists a proven competitive edge in the operating theater,” CEO Brock Faulkner
U.S. Ureteral Access Sheath Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 171.83 million
Revenue forecast in 2033
USD 266.19 million
Growth rate
CAGR of 6.5% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product type, size, age group, clinical indication, end-use
Country scope
U.S.
Key companies profiled
Boston Scientific Corporation; BD (Becton, Dickinson and Company); Olympus; Coloplast; Cook Medical; Advin Health Care; ACE Medical Devices; Blue Neem Medical Devices Private Limited; AMECATH; Applied Medical Resources Corporation; Volkmann Medizintechnik GmbH; Urovision-Urotech; Rocamed SAM; Well Lead Medical Co., Ltd.; Richard Wolf GmbH; SCW Medicath Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Ureteral Access Sheath Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis on the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the U.S. ureteral access sheath market report on the basis of product type, size, age group, clinical indication, and end-use:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Standard (Single-Lumen)
-
Reinforced
-
Dual‑Lumen
-
-
Size Outlook (Revenue, USD Million, 2021 - 2033)
-
<10
-
10 to 12
-
More than or equal to 13 - 15
-
-
Age Group Outlook (Revenue, USD Million, 2021 - 2033)
-
Adult
-
Pediatric
-
-
Clinical Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Stone Management
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Clinics
-
Diagnostic Centers
-
ASCs
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. ureteral access sheath market size was estimated at USD 160.34 million in 2025.
b. The U.S. ureteral access sheath market is expected to grow at a compound annual growth rate of 6.5% from 2026 to 2033 to reach USD 266.19 million by 2033.
b. By product type, the standard (single lumen) segment led the market with the largest revenue share in 2025.
b. Some key players operating in the U.S. ureteral access sheath market include Boston Scientific Corporation, BD, Olympus Corporation, Coloplast, Cook Medical, Advin Health Care, ACE Medical Devices, Blue Neem Medical Devices Private Limited, AMECATH, Applied Medical Resources Corporation, Volkmann Medizintechnik GmbH, Urovision-Urotech, Rocamed SAM, Well Lead Medical Co., Ltd., Richard Wolf GmbH, and SCW Medicath Ltd.
b. The U.S. ureteral access sheath market is primarily driven by the rising prevalence of kidney stones and other urological disorders, which increases the volume of ureteroscopy procedures requiring access sheaths.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










