- Home
- »
- Animal Health
- »
-
Veterinary Regenerative Medicine Market Size, Report, 2033GVR Report cover
![Veterinary Regenerative Medicine Market Size, Share & Trends Report]()
Veterinary Regenerative Medicine Market (2026 - 2033) Size, Share & Trends Analysis Report By Animal (Small Animals, Large Animals), By Product (Stem Cells, Platelet-Rich Plasma), By Application (Orthopedics, Trauma/Wound Care), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-043-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
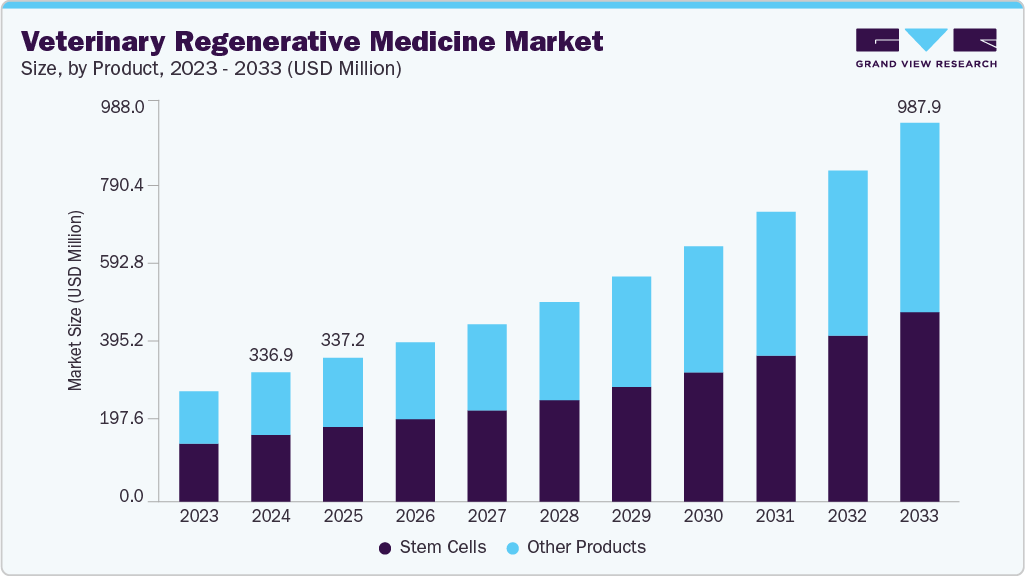
Veterinary Regenerative Medicine Market Summary
The global veterinary regenerative medicine market size was estimated at USD 1.5 billion in 2025 and is projected to reach USD 3.1 billion by 2033, growing at a CAGR of 10.4% from 2026 to 2033. The veterinary regenerative medicine industry is advancing, driven by the rising prevalence of chronic and degenerative diseases in animals,advancements in stem cell and gene therapy technologies, and the growing humanization of pets and willingness to spend on advanced care.
Key Market Trends & Insights
- North America dominated the global veterinary regenerative medicine market with the largest revenue share of 37.3% in 2025.
- By application, the orthopedics segment led the market with the largest revenue share of 41.8% in 2025
- By animal, the small animals segment accounted for the largest market revenue share in 2025.
- By product, the stem cells segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.5 Billion
- 2033 Projected Market Size: USD 3.1 Billion
- CAGR (2026-2033): 10.4%
- North America: Largest market share in 2025
- Asia Pacific: Fastest growing market
The increasing incidence of chronic, degenerative, and age-related conditions such as osteoarthritis, hip dysplasia, tendon injuries, and spinal disorders in companion and livestock animals is driving demand for veterinary regenerative medicine.Osteoarthritis (OA) causes 80% of lameness and joint disease in companion animals, with rising cases due to obesity and longer lifespans. In young dogs, 39.8% show radiographic OA and 23.6% clinical signs, emphasizing underreported prevalence and the need for early detection to optimize treatment outcomes.
Pet owners and veterinarians are seeking advanced treatment options that go beyond symptom management to address the root cause of disease. Stem cell therapy, platelet-rich plasma (PRP), and tissue engineering techniques offer the potential to restore function, reduce pain, and improve quality of life. As awareness of these benefits spreads among pet owners, adoption is accelerating, creating strong growth potential in the regenerative veterinary healthcare segment.
Increasing number of innovations in stem cell isolation, culture, and delivery methods, alongside breakthroughs in gene therapy and biomaterials, is significantly enhancing the efficacy and safety of veterinary regenerative medicine. For instance,the European Commission approved Arti-Cell Forte, the first stem cell-based veterinary product, offering a ready-to-use, long-term solution for equine lameness, developed by Boehringer Ingelheim and GST to address underlying joint inflammation causes.
Improvements in cell viability, targeted delivery, and minimally invasive application techniques are expanding the range of treatable conditions in both small and large animals. These advancements are also lowering costs, making treatments more accessible to a wider population of pet owners. In addition, increasing veterinary clinical trials and peer-reviewed evidence are strengthening veterinarian confidence in regenerative approaches, leading to higher recommendation rates and fostering long-term market adoption.
The Current State Of Veterinary Regenerative Medicine
Species / Category
Key Focus Areas
Applications & Findings
Limitations / Notes
Small Animals - Dogs (PRP)
Platelet-Rich Plasma (PRP)
Used for wounds, periodontitis, joint disease, soft tissue injuries, fractures; improves wound healing; reduces pain & lameness in OA; aids tendon & ligament healing.
Product variability in platelet/WBC concentrations & growth factors affects efficacy.
Cats (MSCs)
Mesenchymal Stem/Stromal Cells (MSCs)
Applied to chronic kidney disease, refractory gingivostomatitis, chronic enteropathy, IBD, spinal cord injury; some use in acute kidney injury, asthma, cardiomyopathy.
Variability in source, dose, administration; lack of controls; limited studies but few adverse effects.
Extracellular Vehicles (EVs) from MSCs
EVs from adipose & placenta MSCs show concentrated immunomodulatory RNA; potential in tissue regeneration; differential miRNA profiles affect anti-inflammatory/immunometabolic roles.
Still experimental; mechanism understanding ongoing.
Dogs (Immunotherapy)
Genetically engineered T-cells
CAR-T cell therapy potential for autoimmune, fibrotic, senescent diseases; aligns with One Health & translational medicine.
Early-stage research; nonmalignant disease focus is new.
Equine - MSCs & PRP
Systematic review/meta-analysis
MSCs or MSC+PRP reduce reinjury rates in tendon/ligament injuries; no increase in return-to-performance.
High study heterogeneity & bias risk.
Equine MSC Preconditioning
Dual-licensed MSCs (IL-1β + TGF-β2)
Enhanced tenocyte migration, metabolism, anabolic gene expression; lower MHC-I for immune evasion.
In vitro study; needs in vivo validation.
Equine MSCs for Septic Arthritis
Immune-activated MSCs
Reduced bacteria & improved lameness vs antibiotics; altered immune gene expression in synovium.
Mechanisms still being mapped.
Equine EV Biomarkers
EV miRNA profiling
PTOA horses have altered plasma & synovial fluid EV miRNA; potential for biomarker development.
Exploratory stage.
Equine EV Therapeutics
EV production from MSCs
3-D culture increases EV yield per cell; informs scalable production methods.
No direct therapeutic testing yet.
Equine Gene Therapy
IL-1 receptor antagonist vector
Improved lameness, cartilage, subchondral bone in PTOA model; supports disease-modifying potential.
Equine model; needs clinical rollout.
Neurologic Diseases (Equine & Canine)
Cellular therapy
Investigated neuroinflammatory & neurodegenerative diseases; early but promising.
Underreported; requires more research.
Exotics - MSC Therapy
Novel cell-based treatments
Potential for joint, soft tissue, autoimmune, inflammatory, infectious diseases in exotic species; useful when repeated treatments not feasible.
Limited case studies; species-specific adaptation needed.
Source: American Veterinary Medical Association, AVMA
The trend of pet humanization has heightened owners’ willingness to invest in sophisticated medical solutions, including regenerative therapies. Pets are increasingly viewed as family members, prompting demand for treatments that extend lifespan and enhance comfort. Higher disposable incomes, expanding pet insurance coverage, and financing options for costly procedures are further boosting market accessibility. Owners are increasingly open to novel, science-backed therapies if they offer improved recovery outcomes and reduced reliance on pharmaceuticals or invasive surgeries. This cultural shift, coupled with the emotional value placed on animal well-being, is a powerful driver shaping the growth trajectory of the veterinary regenerative medicine industry.
Market Concentration & Concentration
The veterinary regenerative medicine industry exhibits a high concentration in the industry, and the pace of the growth is accelerating. The market is dominated by global players such as Zoetis, Boehringer Ingelheim, Dechra, and VetStem, Inc. These companies leverage strong R&D, broad portfolios, and global distribution networks to maintain competitiveness.
The veterinary regenerative medicine industry is highly innovative, driven by advances in stem cell therapies, PRP applications, gene therapy, and extracellular vesicle research. Companies are focusing on disease-modifying approaches, moving beyond symptomatic relief. Novel delivery methods, preconditioned cells, and One Health translational research are accelerating breakthroughs, enhancing treatment efficacy across companion, equine, and exotic species. In April 2025, Transcend Biologics launched TropoVet PRP, an advanced one-step platelet therapy enhancing pet healing and recovery, delivering innovative, effective, and accessible regenerative solutions to the growing veterinary regenerative medicine industry.
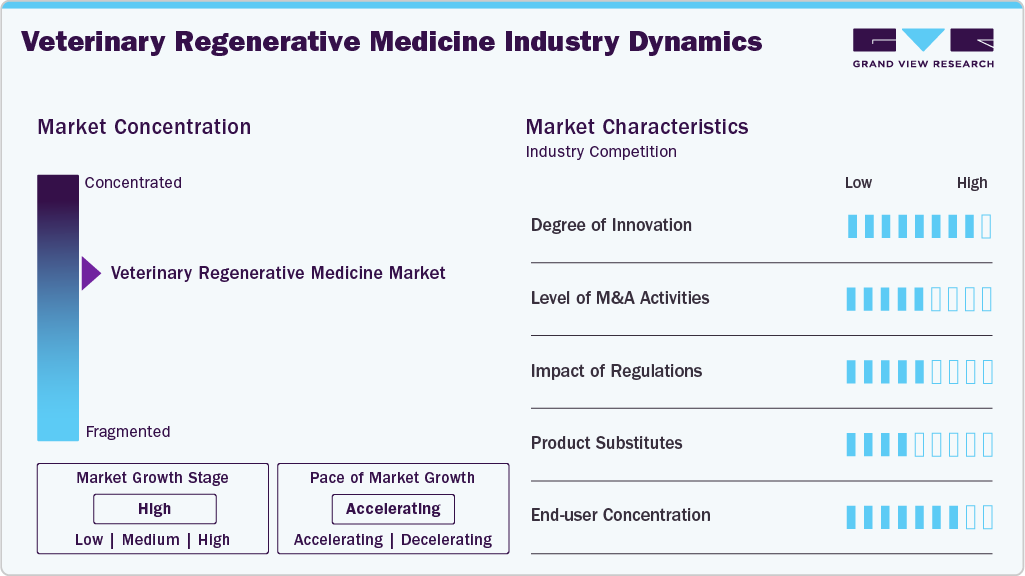
M&A activity is moderate but growing, driven by strategic collaborations among biotech firms, veterinary pharmaceutical companies, and research institutions. For instance, in December 2021. DSM Biomedical and Dechra partner to distribute ProVet regenerative systems, delivering portable, rapid cell and platelet therapies for horses and companion animals. Such deals aim to combine innovation pipelines, regulatory expertise, and distribution networks to accelerate commercialization and gain competitive advantages in emerging therapeutic segments.
Regulations in the veterinary regenerative medicine industry are evolving, with stringent guidelines on product safety, cell sourcing, and clinical evidence. Variations across regions affect market entry timelines and costs. While rigorous approval processes ensure quality, they can delay innovation. Harmonization efforts in the EU and North America are fostering clarity, aiding long-term investment and global product rollout.
Conventional veterinary treatments like NSAIDs, corticosteroids, surgical interventions, and physiotherapy serve as substitutes, offering more established, lower-cost options. However, they often focus on symptom relief rather than regeneration. As awareness grows around the limitations of traditional care, regenerative medicine’s disease-modifying potential is increasingly favored, reducing the competitive threat from substitutes over the long term.
End users are primarily concentrated among veterinary clinics, specialty hospitals, and equine rehabilitation centers. High-value treatments target companion animal owners willing to invest in advanced care and performance-driven equine sectors. Growing adoption by referral veterinarians and specialist practices is driving market penetration, though broader uptake will require cost reductions and wider practitioner training.
Animal Insights
The small animals segment accounted for the largest market revenue share in 2025 and is projected to grow at the fastest CAGR during the forecast period. Rising pet ownership, increased spending on advanced treatments, and growing awareness of regenerative therapies are driving demand. Conditions such as osteoarthritis, ligament injuries, and chronic wounds in small animals are increasingly treated with platelet-rich plasma (PRP), mesenchymal stem cells (MSCs), and other biologics. Pet owners’ willingness to invest in high-quality, minimally invasive solutions is boosting adoption. Veterinary clinics are expanding regenerative offerings for small animals, supported by ongoing clinical research and technological advancements, positioning this segment as the primary growth engine of the market.
The large animals segment is anticipated to grow at a significant CAGR during the forecast period. The increasing adoption of stem cell and platelet-based therapies for conditions such as lameness, tendon injuries, and joint disorders is driving interest. Advancements in portable treatment systems and rising awareness among breeders, trainers, and farmers are accelerating demand for regenerative solutions in large animal care.
Product Insights
The stem cells segment accounted for the largest market revenue share in 2025, driven by their ability to repair, regenerate, and restore damaged tissues in companion and large animals. Widely used to treat osteoarthritis, tendon injuries, and ligament damage, stem cell therapies offer long-term benefits by targeting the root causes of conditions rather than their symptoms. Growing clinical evidence, supportive regulatory pathways, and advancements in cryopreservation and ready-to-use formulations are fueling adoption. For example, in April 2025, VetStem, Inc. secured funding to advance FDA conditional approval of StemStat Ortho, an allogeneic stem cell therapy for canine osteoarthritis, aiming to be the first FDA-approved product in a USD 1 billion market. Strategic collaborations between biotech firms and veterinary providers are enhancing accessibility and innovation.
The platelet-rich plasma (PRP) and other emerging regenerative therapies segment is anticipated to grow at the fastest CAGR during the forecast period, driven by rising adoption for treating musculoskeletal injuries, osteoarthritis, and wound healing in animals. PRP offers a minimally invasive, cost-effective, and natural treatment option that utilizes the animal’s own platelets to stimulate tissue repair and reduce inflammation. Increasing awareness among veterinarians and pet owners about its efficacy, coupled with advancements in portable PRP systems, is boosting market penetration.
Application Insights
The orthopedics segment led the market with the largest revenue share of 41.8% in 2025, driven by the high prevalence of musculoskeletal disorders such as osteoarthritis, ligament injuries, and joint degeneration in companion and performance animals. Regenerative therapies like stem cells, Platelet-Rich Plasma (PRP), and bone marrow concentrates are increasingly used to repair damaged cartilage, promote healing, and reduce inflammation. Rising pet life expectancy, obesity-related orthopedic issues, and demand for minimally invasive, long-lasting treatments are fueling segment growth. Advancements in product innovation, such as allogeneic “off-the-shelf” solutions, are further expanding accessibility. This dominance is reinforced by strong clinical adoption and increasing investment in orthopedic-focused veterinary research.
The other segment is anticipated to grow at the fastest CAGR during the forecast period, encompassing dermatology, wound healing, neurology, dentistry, and ophthalmology. Growth is driven by expanding use of regenerative therapies beyond orthopedics, addressing chronic skin disorders, nerve injuries, oral diseases, and corneal damage in animals. Innovations like PRP, stem cell treatments, and tissue scaffolds are improving recovery outcomes. Rising awareness among veterinarians, coupled with pet owners' growing willingness to explore advanced, minimally invasive solutions, is accelerating adoption and expanding the market’s reach across diverse therapeutic areas.
End Use Insights
The veterinary hospitals and clinics segment accounted for the largest market share in 2025, serving as primary hubs for administering advanced therapies, including stem cell treatments, PRP, and tissue engineering solutions. Their dominance stems from widespread accessibility, specialized equipment, and trained veterinary professionals capable of delivering complex regenerative procedures. These facilities cater to both companion and large animals, driving adoption through integrated diagnostics, treatment, and follow-up care. Increasing partnerships with biotech firms, along with rising demand for minimally invasive and long-term solutions for conditions like osteoarthritis, injuries, and chronic diseases, further solidify their central role in market growth.
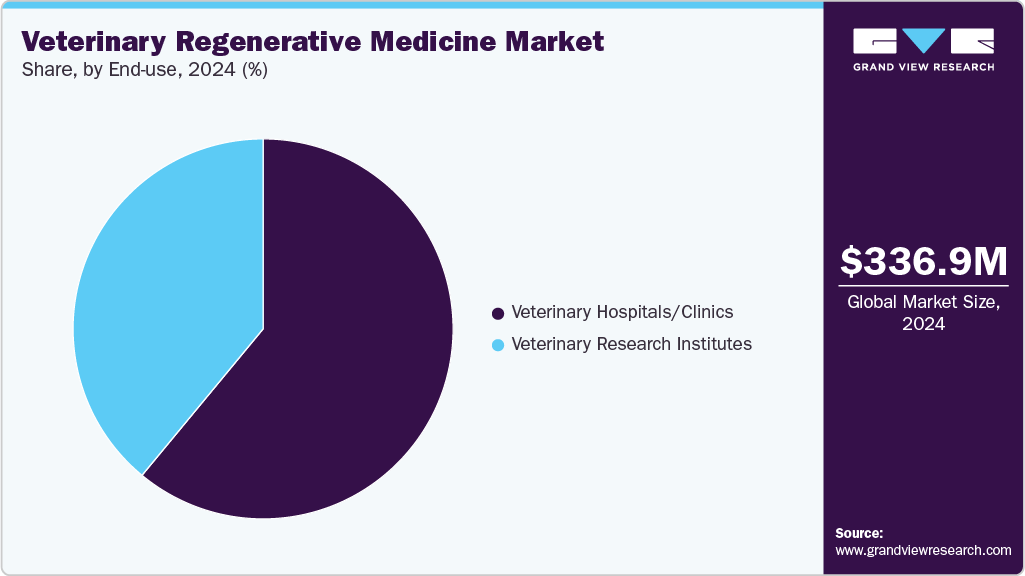
The veterinary research institutes segment is anticipated to grow at the fastest CAGR over the forecast period. These institutes conduct advanced studies in stem cell therapy, tissue engineering, and gene editing, generating scientific evidence that supports the safety and efficacy of regenerative treatments for both companion and livestock animals. Through collaborations with biotechnology companies, veterinary hospitals, and academic institutions, they accelerate the commercialization of novel therapies and facilitate technology transfer. In addition, research institutes often receive government and private funding, enabling large-scale clinical trials and infrastructure development, which further strengthens market growth. Their role in training veterinarians and raising awareness about regenerative treatment options also boosts adoption, ultimately expanding the market’s reach and credibility.
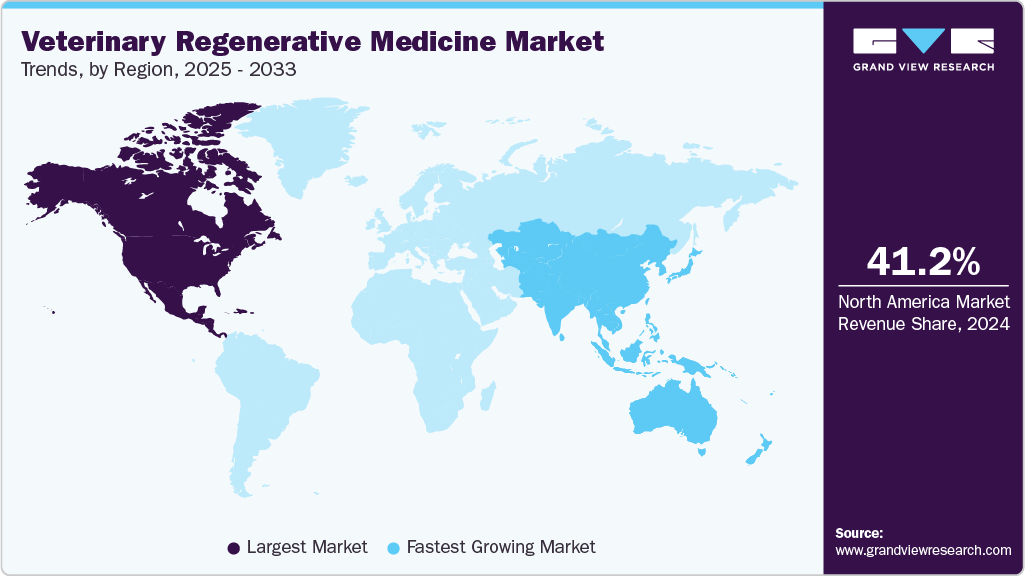
Regional Insights
North America dominated the global veterinary regenerative medicine market with the largest revenue share of 37.3% in 2025, driven by rising pet ownership, increasing prevalence of osteoarthritis, and demand for minimally invasive treatments. Some of the companies include VetStem, Boehringer Ingelheim, Dechra, and Transcend Biologics. In June 2025, Gallant secured USD 18 million Series B funding, led by Digitalis Ventures, to advance FDA conditional approval of its first ready-to-use allogeneic stem cell therapy for pets, targeting refractory Feline Chronic Gingivostomatitis by 2026.
U.S. Veterinary Regenerative Medicine Market Trends
The veterinary regenerative medicine market in the U.S. is expanding rapidly, driven by rising pet healthcare spending, increasing osteoarthritis prevalence, and demand for minimally invasive treatments. Advancements in allogeneic stem cell therapies, PRP systems, and FDA-regulated products are fueling innovation and strengthening market competitiveness. In April 2025, Vetirus Pharmaceuticals acquired Enso Discoveries, enhancing its regenerative medicine portfolio with patented products like Rebound PRP and Rebound PRF, strengthening global capabilities in delivering innovative, cost-effective healing solutions for veterinary and human health.
The Canada veterinary regenerative medicine marketis growing rapidly, driven by increasing pet healthcare spending, strong availability of advanced veterinary services, and the presence of companies developing innovative stem cell therapies, including off-the-shelf regenerative solutions that improve treatment efficiency. In addition, rising cases of orthopedic conditions and injuries in animals are accelerating the adoption of regenerative treatments across both companion and equine segments.
Europe Veterinary Regenerative Medicine Market Trends
The veterinary regenerative medicine market in Europe is growing, driven by rising companion animal ownership, equine sports industry demand, and early adoption of advanced therapies. Some of the players include Boehringer Ingelheim, Dechra, and MSD Animal Health. Innovations such as Arti-Cell Forte, portable PRP/BMC systems, and expanding stem cell applications are enhancing treatment outcomes and boosting regional competitiveness.
The UK veterinary regenerative medicine market is expanding, fueled by increasing pet healthcare spending, strong equine sports demand, and awareness of stem cell and PRP therapies. The Roslin Institute and iC-biosolutions have partnered to develop ready-to-use stem cell preparations, aiming to deliver more effective, affordable regenerative therapies for horses, enhancing tissue repair and advancing veterinary medicine efficiency. Advancements like portable ProVet systems and novel stem cell applications are enhancing accessibility, driving innovation, and positioning the UK as a leader in regenerative veterinary care.
The veterinary regenerative medicine market in Germanyis expanding steadily, driven by its advanced veterinary infrastructure, strong regulatory framework, and high adoption of innovative therapies such as stem cells and platelet-rich plasma. The presence of leading animal health companies, continuous product innovation, and the increasing incidence of degenerative conditions such as arthritis and ligament injuries are further boosting demand, while collaborations between research institutions and clinics are accelerating clinical adoption.
Asia Pacific Veterinary Regenerative Medicine Market Trends
The veterinary regenerative medicine market in Asia Pacific is anticipated to grow at the fastest CAGR over the forecast period, driven by rising pet ownership, increasing livestock care investments, and growing awareness of advanced treatments. Some of the players include VetStem, Medrego, and VETBIO. Advancements in stem cell therapy, PRP, and portable regenerative systems enhance accessibility, while supportive regulations and collaborations with research institutes foster innovation and market penetration.
The India veterinary regenerative medicine market is expanding, driven by rising companion animal care, advances in stem cell research, and government-backed biotech initiatives. Key players include NIAB, StemRx, and Vetbiotech. Innovations like India’s first Animal Stem Cell Biobank in August 2025 and growing collaborations are enhancing therapeutic options, positioning India as a regional hub for advanced, cost-effective regenerative veterinary solutions.
The veterinary regenerative medicine market in China is expecting a steady growth fueled by rapid advancements in stem cell research and increasing clinical trials. Rising pet ownership, urbanization, and a growing middle-class population willing to spend on advanced pet care are key drivers, along with expanding veterinary infrastructure and improving access to specialized treatments. In addition, increasing awareness and ongoing research in tissue engineering and regenerative therapies are significantly accelerating market expansion.
Latin America Veterinary Regenerative Medicine Market Trends
The veterinary regenerative medicine market in Latin America is growing, fueled by increasing pet ownership, demand for advanced treatments, and rising awareness of regenerative therapies. Some of the companies include VetBR, BioCell, and Regenvet. Advancements in stem cell and PRP therapies, along with expanding veterinary hospital capabilities, are driving adoption, offering cost-effective solutions for orthopedic, wound healing, and chronic disease management in companion animals.
The Brazil veterinary regenerative medicine market is expanding, driven by rising pet healthcare spending, advanced therapies for chronic conditions, and growing awareness. Key players like Ourofino Saúde Animal lead innovations by launching a stem cell treatment for dogs. Technological advancements, supportive regulations, and increasing adoption of regenerative solutions position Brazil as a key growth hub in Latin America.
Middle East & Africa Veterinary Regenerative Medicine Market Trends
The veterinary regenerative medicine market in Middle East & Africa is gaining momentum, driven by rising pet ownership, equine sports demand, and advancing biotech infrastructure. Key innovators like Pet Pavilion (UAE) are offering stem cell and PRP therapies, while regional markets such as Saudi Arabia and UAE are leading adoption. For instance, Qatar Foundation’s EVMC launched Inara, a biobank for preserving horse DNA, stem cells, and tissues, enabling rapid regenerative treatments for injuries by ensuring timely access to stored genetic and cellular materials. Collaboration with international biotech firms and regulatory support are accelerating accessibility and innovation.
The South Africa veterinary regenerative medicine market is fast emerging, driven by growing pet ownership and demand for advanced orthopedic care. Stem cell therapies represent the largest segment, while other products are the fastest growing. Key providers such as Valley Farm Animal Hospital (in partnership with VetRenew) and Blue Hills Veterinary Hospital are advancing accessibility, signaling a strengthening and innovation-driven regional market.
Theveterinary regenerative medicine market in Saudi Arabia is gaining traction, propelled by rising pet ownership, expanding veterinary infrastructure, and government investment in biotech research. Biologics and regenerative therapies are the fastest-growing segments, backed by collaborations with global firms. Institutions such as VetStem, Arti-Cell Forte and emerging regional biotech innovators are advancing stem cell and PRP application in both companion and livestock care.
Key Veterinary Regenerative Medicine Company Insights
Major players including Zoetis, Dechra, Boehringer Ingelheim, and VetStem, Inc. command significant market share, leveraging broad product portfolios, global distribution, and strong regulatory compliance. They are advancing through innovations in recombinant and vector-based vaccines, expansion into emerging regions, and strategic M&A activities.
Nonetheless, regulatory hurdles, high R&D costs, and tighter antibiotic restrictions are influencing competition; while emerging and regional companies gain momentum with cost-effective, targeted solutions tailored to local disease challenges and resource constraints.
Key veterinary regenerative medicine Companies:
The following key companies have been profiled for this study on the veterinary regenerative medicine market.
- Boehringer Ingelheim International GmbH
- Zoetis Services LLC
- Dechra
- Integra Lifesciences
- Vetherapy
- Vetstem, Inc.
- Ardent
- Enso Discoveries
- Animal Cell Therapies, Inc.
- Vetbiologics
- Medrego
- Celavet Inc
Recent Developments
-
In July 2025, Gallant raised USD 18 million in Series B funding to advance off-the-shelf stem cell therapies, including treatment for refractory feline chronic gingivostomatitis, using mesenchymal stem cells from the uterus.
-
In April 2025, Vetirus Pharmaceuticals acquired Kansas-based Enso Discoveries, enhancing its regenerative medicine portfolio and expanding capabilities to deliver innovative, cost-effective biologic therapies for animals and humans globally.
-
In April 2025, Transcend Biologics launched TropoVet PRP, a one-step veterinary platelet purification system, enabling on-site extraction of concentrated PRP to accelerate healing, reduce pain, and enhance recovery in animals.
-
In March 2025, VetStem secured funding to complete regulatory submissions for StemStat Ortho, aiming for FDA conditional approval as the first allogeneic stem cell therapy for canine osteoarthritis.
Veterinary Regenerative Medicine Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.6 billion
Revenue forecast in 2033
USD 3.1 billion
Growth rate
CAGR of 10.4% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Animal, product, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Qatar; Oman
Key companies profiled
Boehringer Ingelheim International GmbH; Zoetis Services LLC;Dechra; Integra Lifesciences; Vetherapy; Vetstem, Inc.; Ardent; Enso Discoveries; Animal Cell Therapies, Inc.; Vetbiologics; Medrego; Celavet Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Veterinary Regenerative Medicine Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global veterinary regenerative medicine market report based on animal, product, application, end use, and region.
-
Animal Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Animals
-
Large Animals
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Stem Cells
-
Platelet-Rich Plasma
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Orthopedics
-
Trauma/Wound Care
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Veterinary Hospitals/Clinics
-
Veterinary Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
India
-
China
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global veterinary regenerative medicine market size was estimated at USD 1.5 billion in 2025 and is expected to reach USD 1.6 billion in 2026.
b. Some key players operating in the veterinary regenerative medicine market include VetStem, Inc.; Vetherapy; Boehringer Ingelheim International GmbH; Vetbiologics; Animal Cell Therapies, Inc.; Ardent; Zoetis; Dechra; ACell Inc. (Integra LifeSciences); Enso Discoveries.
b. Key factors that are driving the veterinary regenerative medicine market growth include increasing pet industry spending, greater access to veterinary care, increasing prevalence of soft tissue injuries and osteoarthritis in animals, as well as various benefits of animal stem cell therapy.
b. The global veterinary regenerative medicine market is expected to grow at a compound annual growth rate of 10.4% from 2026 to 2033 to reach USD 3.1 billion by 2033.
b. North America dominated the veterinary regenerative medicine market with a share of 37.3% in 2025. This is attributable to the greater awareness of the therapy and presence of huge number of facilities that provide stem cell therapy services.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










