- Home
- »
- Clinical Diagnostics
- »
-
Zika Virus Testing Market Size, Share & Trends Report, 2030GVR Report cover
![Zika Virus Testing Market Size, Share & Trends Report]()
Zika Virus Testing Market (2024 - 2030) Size, Share & Trends Analysis Report By Test (Molecular Test, Serological Test), By End Use (Hospitals/Clinics, Diagnostic Laboratory), By Region, And Segment Forecasts
- Report ID: GVR-2-68038-356-0
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2018 - 2022
- Forecast Period: 2024 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Zika Virus Testing Market Summary
The global zika virus testing market size was valued at USD 209.5 million in 2023 and is expected to reach USD 341.6 million by 2030, growing at a CAGR of 7.3% from 2024 to 2030. This growth is attributed to the increasing prevalence of Zika virus infections, particularly in endemic regions, which has heightened the demand for diagnostic testing.
Key Market Trends & Insights
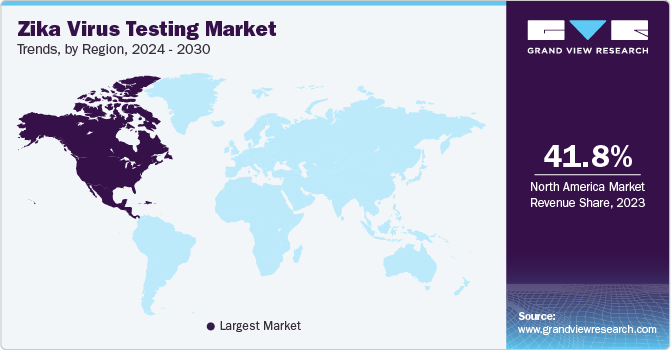
- The North America Zika virus testing market dominated the global market and accounted for the largest revenue share of 41.8% in 2023.
- The Zika virus testing market in the U.S. dominated the North American market and accounted for the largest revenue share of 93.8% in 2023.
- By test trends, the serological test segment dominated the market and accounted for the largest revenue share of 51.1% in 2023.
- By end use, clinical laboratories segment led the market and accounted for the largest revenue share of 47.4% in 2023.
Market Size & Forecast
- 2023 Market Size: USD 209.5 Million
- 2030 Projected Market Size: USD 341.6 Million
- CAGR (2024-2030): 7.3%
- North America: Largest market in 2023
In addition, technological advancements in testing methods and heightened awareness due to previous outbreaks, notably the association of Zika with severe birth defects, have further propelled market growth. Furthermore, the COVID-19 pandemic also played a role, as it intensified the focus on infectious disease testing, including Zika diagnostics. The outbreaks of this virus infection have been noted in regions worldwide, such as North America, Asia Pacific, and Europe. The virus can be transmitted from one person to another via sexual contact. People with low immune systems or newborns have been associated with severe symptoms. However, pregnant women diseased with the Zika virus have more risk of miscarriage or severe congenital disabilities in infants, such as microcephaly and other congenital abnormalities, making consciousness of Zika virus testing.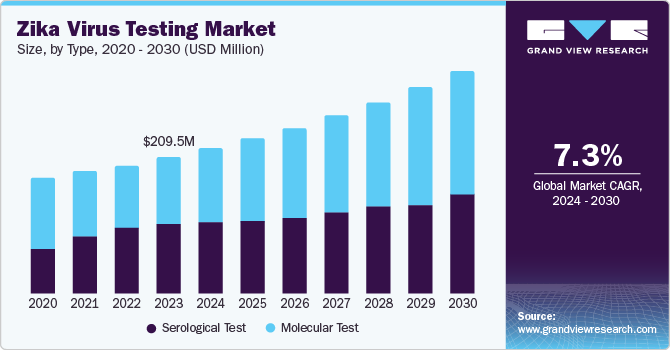
According to the WHO (World Health Organization), Zika virus (ZIKV) infection is endemic in parts of Africa and Southeast Asia. The virus has led to outbursts in developed and developing countries. Major outbreaks have also taken place in Asia-Pacific (particularly India) and South American countries. In addition, updated WHO data indicates that Zika transmission continues in several countries.
Furthermore, the healthcare sector is dealing with numerous viral outbreaks in the present decade, including SARS, Chikungunya, Swine Flu, and Zika Virus, which led to the demand rate for advanced diagnostic tools. The rising prevalence of Zika virus illness and technological developments in Zika virus testing fuels the market. Therefore, the increasing prevalence of Zika virus infection is a key factor contributing to the market's growth during the forecasted period. Moreover, the government's initiatives to develop novel tests that can detect the Zika virus significantly and other efforts taken by governments to create awareness about the infection and associated problems among citizens are key factors boosting global Zika virus testing market growth.
Test Trends
The serological test dominated the market and accounted for the largest revenue share of 51.1% in 2023. These tests are capable of detecting acute & late-stage Zika virus in patients. These test types rely on detecting IgM and IgG antibodies through (enzyme-linked immunosorbent assay) ELISA-based tests. The accessibility of advanced serological tests for rapid detection increases segment revenue growth. Antigen-specific IgG is produced shortly after IgM becomes detectable and can be detected for years after the initial infection. However, serological analysis for ZIKV is complicated by significant cross-reactivity among antigens in ZIKV and other flaviviruses.
Molecular tests are expected to grow at a CAGR of 8.8% over the forecast period attributed to the continuous advancements in molecular diagnostic technologies, such as polymerase chain reaction (PCR) and nucleic acid amplification. The development of multiplex testing platforms that can simultaneously detect Zika virus and other pathogens has improved efficiency and reduced costs. Furthermore, implementing comprehensive surveillance systems to monitor the prevalence of the Zika virus and identify potential outbreaks has further stimulated the demand for molecular tests.
End Use Insights
Clinical laboratories led the market and accounted for the largest revenue share of 47.4% in 2023. This growth is driven by people's awareness of the diagnosis test for viruses, backed by the rising prevalence of chronic disorders among people nowadays in the fast-paced world. Moreover, the innovations and adaptations in types of diagnostics such as molecular or serological and sequencing of next-generation, the importance of early detection and treatment of diseases to prevent severe losses, broad development and growth in healthcare infrastructure in countries are some of the key factors enabling the improved patient result and enhanced services across the globe.
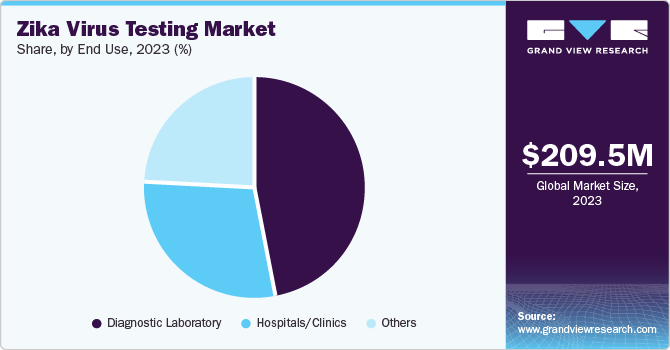
Hospitals are expected to grow at a CAGR of 7.3% over the forecast period attributed to the rising demand for diagnosis and treatment of Zika virus and the high need for accurate and quick tests. In addition, advancements in molecular diagnostics and point-of-care testing, expansion in the availability of medical services in Zika-affected regions, and implementation and adoption of testing in prenatal care are further expected to contribute toward the market’s growth worldwide.
Regional Insights & Trends
The North America Zika virus testing market dominated the global market and accounted for the largest revenue share of 41.8% in 2023 driven by the availability and accessibility of the latest diagnostic techniques, enhanced healthcare systems, and highly developed healthcare infrastructure. In addition, the initiatives by organizations, such as UNICEF, Centers for Disease Control and Prevention (CDC), and Pan American Health Organization (PAHO), to raise awareness about illness and prevention of Zika virus, enhancement in mosquito control agendas with improved laboratory, epidemiology, and surveillance capacity in at-risk areas to minimize the chances of Zika transmission are aspects for market growth.
U.S. Zika Virus Testing Market Trends
The Zika virus testing market in the U.S. dominated the North American market and accounted for the largest revenue share of 93.8% in 2023. The U.S. citizens were affected by this disease primarily due to huge numbers of travelers to and from infected regions, with the addition of sporadic locally acquired infections in Florida and Texas. Some important factors for growth include the awareness of mosquito-borne diseases, enhancements in diagnostic systems, and government support for disease control. The market has the advantage of quick but accurate testing methods diagnosed and managed promptly. In addition, the increased incidence of travel-related viral cases has lifted the demand for efficient testing solutions.
Latin America Zika Virus Testing Market Trends
The Latin American Zika virus testing market is expected to grow at a CAGR of 8.8% over the forecast period driven by the high prevalence of Zika infections in the region. In addition, the presence of the Aedes aegypti mosquito, the main vector for Zika, further exacerbates the situation. Increased public awareness, government interventions, and funding from organizations such as UNICEF enhance testing capabilities. Furthermore, the correlation between Zika infections and severe birth defects, such as microcephaly, has intensified the demand for effective diagnostics, thereby driving the growth of the market in the region.
Europe Zika Virus Testing Market Trends
The Zika virus testing market in Europe is expected to witness a significant rise over the forecast period owing to investments made by governments for manufacturing of Zika virus test kits and growing healthcare expenditure. In addition, the awareness among citizens of Europe of the Zika virus favors the country's market positively. Furthermore, the government supports the development of research tools by conducting several trials that help collect data on the Zika virus outbreak and prevent further harm.
France Zika virus testing market is expected to grow significantly over the forecast period owing to the rising transmission of the Zika virus in the country. The authorities have taken measures to minimize or prevent viral transmission, deploying major prevention and education strategies with populations, including pregnant women and their partners, health care professionals, and schools or colleges. Hence, surveys indicate that climate change may lead to the spread of the Zika virus throughout the nation, denoting a risk and propelling the market growth.
Asia Pacific Zika Virus Testing Market Trends
Asia Pacific Zika virus testing market is expected to grow with a CAGR of 8.7% over the forecast period. The rising number of cases of the Zika virus drives this growth in a few APAC countries, as well as ongoing research on the disease and rising investments in R&D activities. In past years, populous countries such as India and China have witnessed numerous cases along with severe disorders among people, which led to demand for effective testing of the disease, owing to the factors mentioned above, such as the high prevalence of Zika virus infections and the rising demand for diagnosis, the Zika virus testing market is anticipated to see growth over the forecast period in Asia Pacific.
The Zika virus testing market in Singapore is expected to grow significantly, owing to the increasing incidence of Zika infections, particularly in the region. In addition, enhanced public awareness and government initiatives aimed at controlling outbreaks further support this market expansion. Furthermore, advancements in diagnostic technologies, including rapid testing methods and serological kits, are improving detection capabilities, making testing more accessible and efficient for healthcare providers in Singapore.
Key Zika Virus Testing Company Insights
Some of the key companies in the Zika virus testing market include Abbott; DiaSorin S.p.A.; F. Hoffmann-La Roche Ltd.; Quest Diagnostics Incorporated; Siemens Healthineers AG; Chembio Diagnostic Systems, Inc.; SD Biosensors, Inc.; Biocan Diagnostics; ARUP Laboratories; Viatris Inc. in the market are focusing on development & to gain a competitive edge in the industry. Therefore, key players are taking several strategic initiatives.
-
Abbott Laboratories is a global healthcare company known for its innovative diagnostics, medical devices, nutrition products, and branded generic pharmaceuticals. In the context of Zika virus testing, Abbott has developed the RealTime ZIKA test, which received FDA Emergency Use Authorization. This molecular test is designed to detect Zika virus RNA in various sample types, providing rapid and accurate results.
-
DiaSorin SpA is a medical diagnostics company that develops, manufactures, and markets in vitro diagnostic reagent kits and systems. The company develops reagent kits based on Enzyme-Linked Immunosorbent Assays (ELISA) and Chemiluminescence (CLIA) techniques to enable automatic diagnostic investigation.
Key Zika Virus Testing Companies:
The following are the leading companies in the zika virus testing market. These companies collectively hold the largest market share and dictate industry trends.
- Abbott
- DiaSorin S.p.A.
- F. Hoffmann-La Roche Ltd.
- Quest Diagnostics Incorporated
- Siemens Healthineers AG
- Chembio Diagnostic Systems, Inc.
- SD Biosensors, Inc.
- Biocan Diagnostics
- ARUP Laboratories
- Viatris Inc.
Recent Developments
-
In May 2024, The FDA provided comprehensive updates on Zika virus response efforts, detailing the virus's transmission, symptoms, and associated risks, particularly for pregnant women. Key highlights included the lack of FDA-approved vaccines and treatments, though several diagnostic tests have received Emergency Use Authorization. Further, recent determinations indicated that Zika is no longer a relevant transfusion-transmitted infection, allowing blood establishments to adjust screening protocols. The FDA emphasizes ongoing collaboration with public health authorities to ensure safety and effective responses to Zika outbreaks.
Zika Virus Testing Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 223.5 million
Revenue forecast in 2030
USD 341.6 million
Growth rate
CAGR of 7.3% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Quantitative units
Revenue in USD million and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test, end use, region
Regional scope
North America, Europe, Asia Pacific, Latin America, MEA
Country scope
U.S. states, U.S. Territories, Continental Europe, Caribbean (EU), Singapore, Vietnam, Indonesia, Thailand, Brazil, Colombia, South Africa
Key companies profiled
Abbott; DiaSorin S.p.A.; F. Hoffmann-La Roche Ltd.; Quest Diagnostics Incorporated; Siemens Healthineers AG; Chembio Diagnostic Systems, Inc.; SD Biosensors, Inc.; Biocan Diagnostics; ARUP Laboratories; Viatris Inc.
Customization scope
Free report customization (equivalent to up to 8 analysts' working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
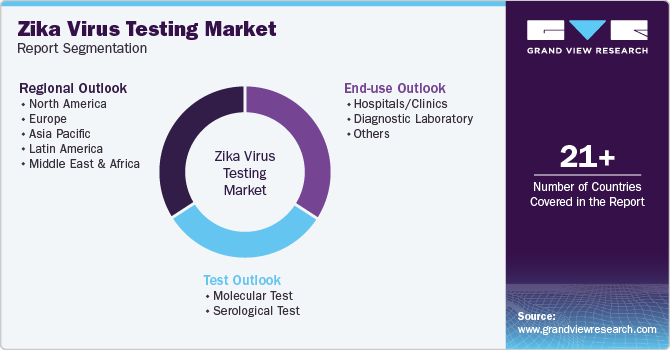
Global Zika Virus Testing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and analyzes the latest industry trends in each sub-segment from 2018 to 2030. For this study, Grand View Research has segmented the global Zika virus testing market report based on test, end use, and region:
-
Test Outlook (Revenue, USD Million, 2018 - 2030)
-
Molecular Test
-
Serological Test
-
-
End use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals/Clinics
-
Diagnostic Laboratory
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S. States
-
U.S. Territories
-
-
Europe
-
Continental Europe
-
Caribbean(EU)
-
-
Asia Pacific
-
Singapore
-
Vietnam
-
Indonesia
-
Thailand
-
-
Latin America
-
Brazil
-
Colombia
-
-
Middle East and Africa (MEA)
-
South Africa
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










