- Home
- »
- Market Trend Reports
- »
-
Ophthalmic Clinical Trials: Emerging Trends And Pipeline Outlook
Report Overview
The ophthalmic clinical trials market is witnessing rapid growth, driven by rising healthcare needs and technological advancements. Emerging trends in ophthalmology are influenced by increasing global prevalence of age-related diseases like macular degeneration, diabetic retinopathy, and glaucoma, as well as the need for new therapeutic approaches to address rare and complex conditions. This report provides a comprehensive outlook on the landscape of ophthalmic clinical trials, offering insights into market dynamics, prevalence trends, R&D investments, and the evolving regulatory environment that are determining the future of clinical research in this field. By analyzing active clinical trials by phase, study design, key indications, and region, along with emerging clinical trial models and key driving factors, this report helps stakeholders understand the opportunities and challenges in developing new treatments and technologies for ophthalmic disorders. It will serve as a strategic tool to navigate the evolving clinical trial ecosystem.
Ophthalmic Clinical Trials: Emerging Trends and Pipeline Outlook Report Coverage
Ophthalmic Clinical Trials: Emerging Trends and Pipeline Outlook Report Coverage
Market Outlook
Prevalence Trends Analysis
R&D Investment Analysis
Industry Ecosystem Analysis
Market Dynamics
Regulatory Framework
List of Top 50 Active Trials by Phase, Sponsor, and Indication
Emerging Clinical Trial Model Analysis
Global Ophthalmic Clinical Trials, by Phase & Study Design
Global Ophthalmic Clinical Trials, by Key Indications, by Region
The increasing prevalence of ocular diseases, such as age-related macular degeneration (AMD), diabetic eye diseases, and glaucoma, is driving significant growth in ophthalmic clinical trials. For instance, the increasing geriatric population and the incidence of several conditions like myopia and diabetic retinopathy across the globe bolster the need for novel and more effective treatments in ophthalmology. According to the National Institute of Aging (NIA), the geriatric population is expected to reach around 72 million by 2030. The increasing prevalence of diabetes and the incidence of diabetes-related blindness are predicted to fuel market growth. According to the World Health Organization (WHO), nearly 830 million people worldwide have diabetes, and the majority live in low- and middle-income countries. This heightened demand led to an upsurge in clinical trials aimed at addressing unmet medical needs, especially in areas with high morbidity rates. The prevalence of diseases such as dry eye disease and retinal disorders, which often require novel drug delivery methods or gene therapies, is influencing the type and design of clinical trials.
In addition, the adoption of advanced imaging techniques, biomarkers, and personalized medicine is accelerating research in these areas. For stakeholders, understanding the prevalence trends is vital in predicting market growth, optimizing trial design, and ensuring that the right therapies are developed to meet the needs of diverse patient populations. This report’s analysis of prevalence trends enables a deep dive into how these factors shape clinical trial activity. It provides stakeholders with essential insights to align their research and development efforts with growing market demand.
R&D Investment and Funding Analysis
R&D investment is a significant catalyst in ophthalmic clinical trials, with both private and public sectors prioritizing funding for novel treatments, especially in the areas of gene therapy, biologics, and drug delivery systems. Growing government initiatives to enhance research and development in ophthalmology worldwide are driving clinical trial activities. For instance, as per the American Academy of Ophthalmology data, In fiscal year 2024, U.S. government allocated USD 47 billion to the National Institutes of Health (NIH), USD 896 million to the National Eye Institute (NEI), and USD 20 million to the Department of Defense Vision Research Program (VRP). The increasing availability of venture capital and strategic partnerships between biopharma companies, research institutes, and contract research organizations (CROs) has led to a more dynamic and innovative clinical trial environment.
Furthermore, the regulatory framework plays a pivotal role in shaping trial methodologies and approval processes. Regulatory agencies, such as the FDA and EMA, improved regulatory guidelines to accelerate approval for breakthrough therapies through mechanisms like fast-track designation and orphan drug status. These regulatory pathways incentivize the development of treatments for rare and complex ophthalmic conditions, thereby driving clinical trial activity.
The increasing adoption of digital health technologies, remote monitoring, and artificial intelligence (AI) tools in clinical trials is transforming R&D workflows and enabling more efficient trials. By providing detailed insights into R&D funding trends, regulatory changes, and investment flows, this report enables stakeholders to understand the evolving financial landscape and regulatory influences that are accelerating the pace of innovation in ophthalmic clinical trials.
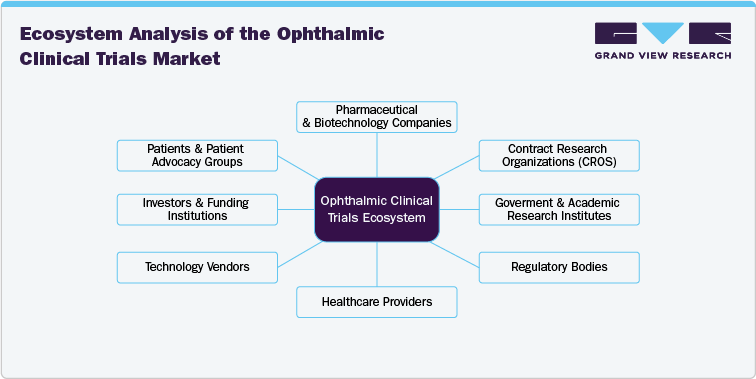
The ophthalmic clinical trials ecosystem comprises numerous stakeholders, including pharmaceutical & biotechnological companies, CROs, regulatory bodies, research institutions, healthcare providers, technology vendors, investors & funding institutions, and patients & advocacy groups. Pharmaceutical organizations and biotech firms are at the forefront of clinical trial sponsorship, investing heavily in research to develop innovative therapies for age-related eye diseases, retinal disorders, and gene therapies.
CROs, acting as intermediaries between sponsors and clinical trial sites, play a critical role in ensuring trial efficiency, regulatory compliance, and data management. Research institutions and universities often collaborate with industry players to advance novel treatment approaches, particularly in niche areas like gene editing and stem cell therapy.
Regulatory bodies like the U.S. FDA and EMA establish the framework within which clinical trials operate, influencing study design, recruitment strategies, and approval processes. The ecosystem also includes technology vendors providing clinical trial management solutions, data analytics tools, and advanced diagnostic technologies like OCT and AI-driven imaging. The convergence of these entities is fostering innovation, making the ophthalmic clinical trials market highly collaborative and competitive.
Emerging Clinical Trial Models
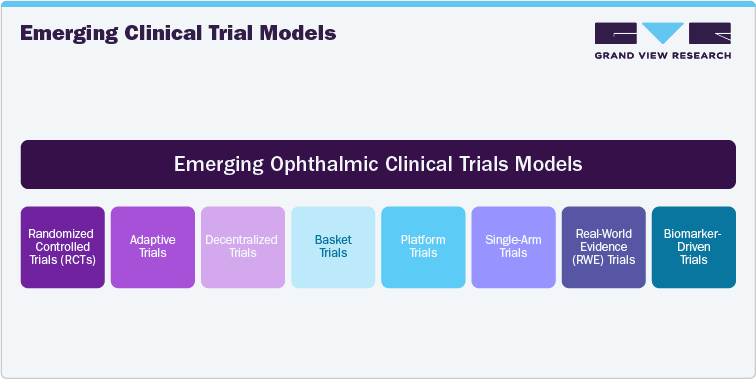
Emerging clinical trial models are transforming the way ophthalmic trials are designed and conducted. With the advent of decentralized trials, remote patient monitoring, and telemedicine, clinical trials are becoming more patient-centric, reducing geographic and logistical barriers. Virtual visits, wearables, and mobile health apps enable real-time data collection, making it easier for patients to participate from their homes, especially in rare or geographically dispersed conditions. Adaptive trial designs are also gaining traction, allowing modifications to the trial protocol based on interim results, thereby improving efficiency and reducing costs. Moreover, artificial intelligence (AI) and machine learning are enhancing recruitment, predictive modeling, and the identification of patient populations that are most likely to benefit from specific therapies. These advancements are making clinical trials more efficient and allowing the collection of more granular data, thus providing deeper insights into treatment efficacy and safety. This report explores these emerging trial models, offering key insights into how these innovative approaches are reshaping ophthalmic research.
U.S. Ophthalmology Active Clinical Trials, by Phase (2024)
Phase
Number of Trials
Key Sponsors
Top Indications
Phase I
68
Biogen, Novartis, Roche
Gene therapy (Retinal diseases), AMD
Phase II
99
Bayer, Allergan, Regeneron
Diabetic Retinopathy, Dry Eye Disease
Phase III
69
Pfizer, Sanofi, Regeneron
Glaucoma, Cataract, Retinitis Pigmentosa
Phase IV
24
Merck, Santen, AbbVie
Post-surgical Inflammation, Uveitis
Source: Clinicaltrials.gov, Grand View Research
This report’s detailed analysis of active clinical trials across phases provides stakeholders with a global view of ongoing research, helping identify trends, key players, and the most promising areas for investment. By offering insights into the number of trials, sponsors, and indications at each phase, this table serves as a vital resource for those looking to track the progress of the ophthalmic pipeline and make informed decisions about future investments and trial strategies.
List of Major Active Trials by Phase, Sponsor, and Indication, A Key Example:
Clinical Trial Study Title
Effectiveness of Intravitreal Injection of Aflibercept 8 mg in Resistant Diabetic Macular Edema, Retinal Vein Occlusion, and Myopic Choroidal Neovascularisation Patients
Study Status
Not Yet Recruiting
Phase
Early Phase I
Study Type
Interventional
Type of Ophthalmic Tests Required
- Best Corrected Visual Acuity (BCVA)
- Optical Coherence Tomography (OCT)
Study Design
- Allocation: NA
- Intervention Model: Single Group
- Masking: None
- Primary Purpose: Treatment
Conditions
- Diabetic Macular Edema (DME)
- Retinal Vein Occlusion (RVO)
- Myopic Choroidal Neovascularisation
Interventions
DRUG: Intravitreal injection of aflibercept 8 mg
Sponsor
Tanta University
Number of Patients (Enrollment)
60
Start Date
19-10-2025
Primary Completion Date
19-11-2025
Completion Date
19-12-2025
Locations
Faculty of Medicine, Tanta University, Tanta, 1335, Egypt
Other aspects that shall be analyzed will include the market overview, clinical trials by study design, by key indications, by region, and a list of key clinical trials, sponsors, among several other factors.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


