- Home
- »
- Pharmaceuticals
- »
-
Actinic Keratosis Treatment Market, Industry Report, 2033GVR Report cover
![Actinic Keratosis Treatment Market Size, Share & Trends Report]()
Actinic Keratosis Treatment Market (2026 - 2033) Size, Share & Trends Analysis Report By Therapy (Topical, Surgery, Photodynamic Therapy), By Drug Class (Nucleoside Metabolic Inhibitor, Immune Response Modifiers), By End Use (Hospitals, Homecare), By Region, And Segment Forecasts
- Report ID: GVR-2-68038-940-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Actinic Keratosis Treatment Market Summary
The global actinic keratosis treatment market size was estimated at USD 7.02 billion in 2025 and is projected to reach USD 10.13 billion by 2033, growing at a CAGR of 4.86% from 2026 to 2033. This growth is driven by the increasing prevalence of actinic keratosis due to prolonged ultraviolet (UV) exposure and a growing aging population vulnerable to sun-related skin damage.
Key Market Trends & Insights
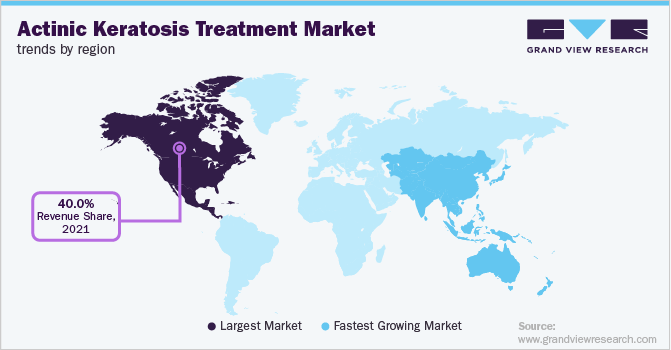
- North America actinic keratosis treatment market dominated the global market and accounted for the largest revenue share of 41.41% in 2025.
- U.S. led the North America market and held the largest revenue share in 2025.
- Based on therapy, the photodynamic therapy segment is the fastest growing during the forecast period with CAGR of 8.07%.
- On the basis of drug class, the immune response modifiers segment held the largest revenue share of 35.02% in 2025.
- On the basis of end use, the private clinics segment held the largest revenue share of 49.07% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 7.02 Billion
- 2033 Projected Market Size: USD 10.13 Billion
- CAGR (2026-2033): 4.86%
- North America: Largest Market in 2025
Rising awareness about early diagnosis of precancerous skin conditions and improved access to dermatology services are also supporting market expansion. In addition, advancements in topical therapies, photodynamic therapy technologies, and cryotherapy procedures, along with the availability of effective prescription medications, are further contributing to the market’s steady growth.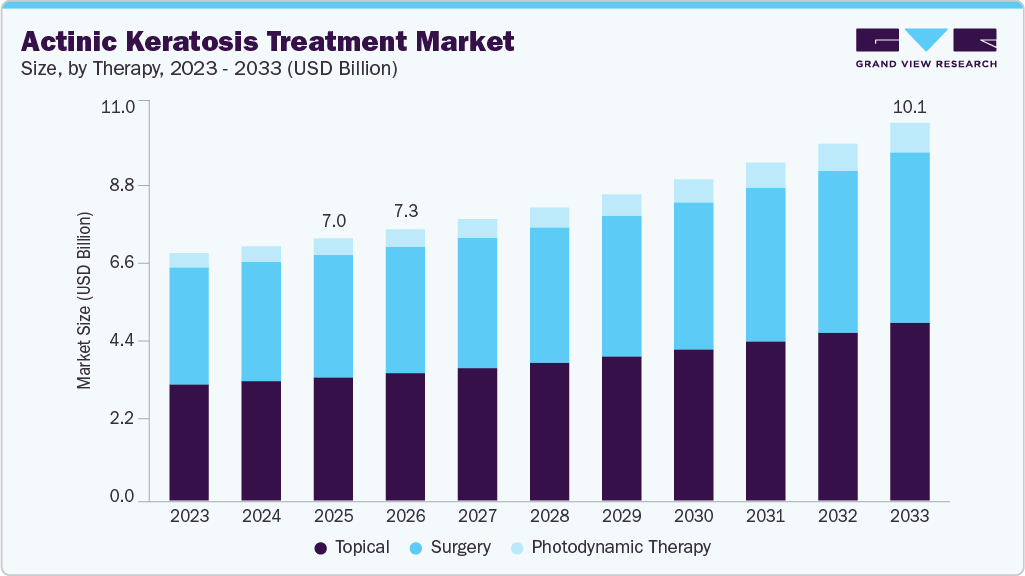
Actinic keratosis is among the most common precancerous skin conditions, largely caused by prolonged exposure to ultraviolet (UV) radiation from sunlight or artificial sources. The increasing incidence, particularly among fair-skinned and aging populations, is significantly expanding the patient pool requiring treatment. For instance, in April 2024, Oxford University Press published a meta-analysis in the British Journal of Dermatology reporting a pooled global prevalence of 14% (95% CI: 14-15), with rates reaching 18% in tropical and temperate regions and 19% among individuals aged over 60 years. This growing disease burden is directly translating into higher demand for effective therapeutic interventions.
The market is also benefiting from increasing awareness and early diagnosis of skin disorders, supported by large-scale public health initiatives and screening programs. Governments and dermatology organizations are actively promoting early detection of skin lesions, encouraging individuals to seek timely medical consultation. For instance, in May 2024, the Skin Cancer Foundation reported that over 5 million skin cancer cases are diagnosed annually in the U.S., with 1 in 5 individuals expected to develop skin cancer by age 70, largely linked to UV exposure. Additionally, screening initiatives have proven effective in early identification; the American Academy of Dermatology reported in 2024 that its programs have screened over 2.9 million individuals, identifying more than 293,000 suspicious lesions. Such initiatives are significantly contributing to earlier diagnosis and treatment of actinic keratosis, thereby driving market growth.
Advancements in dermatology treatments and therapies are further strengthening market expansion by improving treatment efficacy, safety, and patient compliance. Pharmaceutical innovation has led to the development of newer topical therapies with shorter treatment durations and better tolerability, while procedural advancements such as photodynamic therapy (PDT) are gaining traction for their ability to treat multiple lesions with favorable cosmetic outcomes. For instance, in July 2024, a Phase 3 multicenter study published in the Journal of Drugs in Dermatology demonstrated that tirbanibulin ointment 1% reduced lesion count by 77.8% within 57 days in patients with actinic keratosis, highlighting the effectiveness of emerging therapies. In parallel, combination treatment approaches integrating topical and procedural methods are increasingly adopted to enhance clinical outcomes and reduce recurrence rates.
Furthermore, demographic and environmental factors such as aging populations, high UV exposure regions, and occupational risks are contributing to sustained market demand. Older individuals, who accumulate prolonged UV exposure over time are more susceptible to developing multiple lesions, increasing the need for ongoing dermatological care. For instance, in August 2025, a study published in Frontiers in Medicine reported a 5.84% prevalence of actinic keratosis among elderly residents in Shanghai, with age identified as a significant risk factor (OR 4.37). In addition, geographic regions with high UV radiation and rising public awareness are witnessing increased diagnosis rates. As a result, the demand for safe, effective, and minimally invasive treatment solutions is expected to grow steadily, positioning the actinic keratosis treatment market for continued expansion in the coming years.
Market Concentration & Characteristics
Innovation in the actinic keratosis treatment market remains at a moderate to high level, driven by continuous advancements in dermatology therapies and pharmaceutical formulations. The market is characterized by the development of topical medications, photodynamic therapy (PDT), and minimally invasive procedures that improve treatment efficacy and patient compliance. Newer agents such as tirbanibulin and improved formulations of fluorouracil and imiquimod are enhancing treatment outcomes by reducing therapy duration and minimizing adverse reactions. In addition, advancements in field-directed therapies allow clinicians to treat both visible and subclinical lesions simultaneously, improving long-term disease management. For instance, as highlighted in the report, a July 2024 Phase 3 study demonstrated that tirbanibulin ointment achieved a 77.8% reduction in lesion count within 57 days, reflecting the growing effectiveness of next-generation therapies. These innovations are expanding commercial opportunities for pharmaceutical companies and strengthening competitive positioning through differentiated product offerings.
The actinic keratosis treatment market exhibits a moderately fragmented competitive structure, with the presence of both pharmaceutical companies and medical device manufacturers. Key players such as Almirall, Galderma, Sun Pharmaceutical, and Biofrontera focus on topical drugs and photodynamic therapy agents, while companies like Brymill Cryogenic Systems provide cryotherapy devices. As per the report, companies are increasingly adopting product launches, expansions, and collaborations as primary growth strategies rather than aggressive merger and acquisition activity. Strategic partnerships with dermatology clinics, outpatient centers, and distribution networks are helping companies expand geographic reach and improve product accessibility. This collaborative ecosystem supports steady market penetration without significant consolidation, particularly in emerging markets where dermatology infrastructure is still developing.
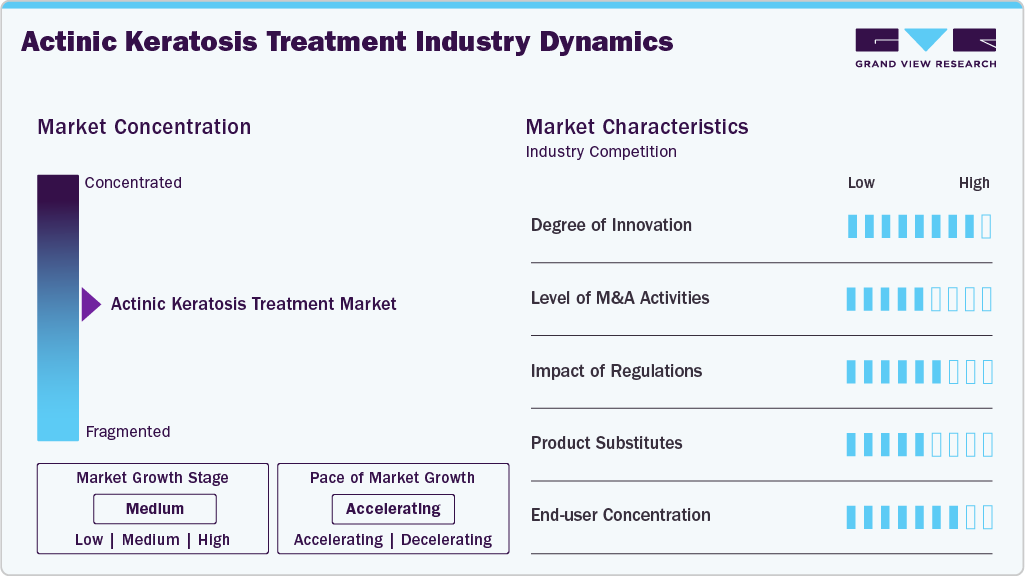
The regulatory landscape plays a critical role in shaping the market, acting as both an enabler and a barrier to entry. Approval processes for dermatological drugs and therapies require extensive clinical validation to ensure safety and efficacy, particularly for novel topical agents and photodynamic therapy products. Regulatory bodies such as the U.S. FDA and European Medicines Agency (EMA) impose stringent requirements, which can increase development timelines and costs. In addition, variations in regulatory frameworks and reimbursement policies across regions can impact product adoption and market entry strategies. The report also highlights that treatment cost variability and reimbursement differences influence patient access, particularly in emerging economies, thereby affecting overall market growth dynamics.
The market demonstrates moderate substitutability, with multiple treatment options available but limited external alternatives beyond established dermatological therapies. Internal substitution is evident between topical treatments, cryotherapy, and photodynamic therapy, depending on lesion severity, patient preference, and clinical guidelines. For instance, topical therapies dominate due to their non-invasive nature and convenience for homecare use, accounting for a significant share of treatment adoption, while photodynamic therapy is gaining traction for treating larger or cosmetically sensitive areas. Combination therapy approaches are also emerging, enabling clinicians to integrate multiple modalities for improved outcomes and reduced recurrence rates. This evolving treatment landscape continues to reshape competitive dynamics and product portfolios.
Therapy Insights
The topical treatment segment held the largest share of 46.98% in 2025, largely because it offers a convenient, non-invasive approach that can be applied directly to sun-damaged skin areas. Dermatologists often prefer topical therapies for patients with multiple lesions or “field cancerization,” where precancerous cells extend beyond visible lesions. Widely used agents such as fluorouracil, imiquimod, diclofenac, and newer molecules like tirbanibulin are designed to eliminate abnormal keratinocytes or stimulate immune responses within affected skin areas, making them highly effective in early-stage AK management.
One of the key factors driving the growth of the topical segment is its suitability for field-directed therapy, allowing treatment across larger skin areas rather than targeting individual lesions. This capability aligns with modern dermatology practices, where clinicians aim not only to remove visible lesions but also to treat the broader field of sun-damaged skin to prevent recurrence or progression to squamous cell carcinoma. As dermatology clinics increasingly focus on preventive skin cancer management, topical therapies are becoming an essential part of long-term treatment strategies.
The photodynamic therapy segment is expected to grow at the CAGR of 8.07% during the forecast period, due to its ability to selectively destroy abnormal skin cells while preserving surrounding healthy tissue. PDT involves the application of a photosensitizing agent to the affected skin area, followed by exposure to a specific wavelength of light that activates the drug and destroys precancerous cells. This targeted approach is particularly valuable for patients with multiple lesions or large areas of sun-damaged skin, where field-directed treatment is often preferred over lesion-by-lesion procedures. As dermatology continues to emphasize both therapeutic efficacy and cosmetic outcomes, PDT has gained strong acceptance in clinical practice.
Drug Class Insights
The immune response modifiers segment held the largest revenue share of 35.02% in 2025, as it utilizes the body’s own immune system to identify and eliminate abnormal precancerous cells. Unlike cytotoxic therapies that directly destroy cells, immune response modifiers stimulate local immune activity within the skin, triggering inflammatory and immune-mediated pathways that attack dysplastic keratinocytes. Topical agents such as imiquimod are widely used in dermatology practice for treating AK lesions on sun-exposed areas, particularly the face and scalp, where field-directed therapy is often preferred.
The mechanism of action behind this drug class is centered on immune activation through Toll-like receptors (TLRs). Imiquimod acts as a TLR-7 agonist, activating immune cells such as dendritic cells, macrophages, and natural killer cells in the skin. This activation stimulates the release of cytokines including interferon-alpha, tumor necrosis factor, and interleukins, which collectively trigger an immune attack against abnormal keratinocytes and promote apoptosis of precancerous cells.
The photoenhancer drug class segment is expected to grow at the fastest CAGR over the forecast period and is closely associated with photodynamic therapy, where specialized compounds enhance the sensitivity of abnormal skin cells to light-based treatment. Photoenhancers, also known as photosensitizers, are applied topically and selectively accumulate in precancerous keratinocytes. Once activated by a specific wavelength of light, these agents trigger a photochemical reaction that produces reactive oxygen species capable of destroying abnormal cells while preserving surrounding healthy tissue. This selective mechanism has made photoenhancers an important component in modern dermatologic oncology and a key driver of innovation in AK therapy.
End Use Insights
The private clinics held the largest share in the market in 2025, as many patients prefer accessible outpatient dermatology services for early diagnosis and treatment of precancerous skin lesions. Private dermatology clinics provide specialized care, shorter appointment waiting times, and personalized treatment approaches, making them a convenient option for patients seeking routine skin examinations and lesion management. In many healthcare systems, dermatology consultations in private settings account for a substantial portion of skin disease management, reflecting the growing importance of outpatient care models in dermatology.
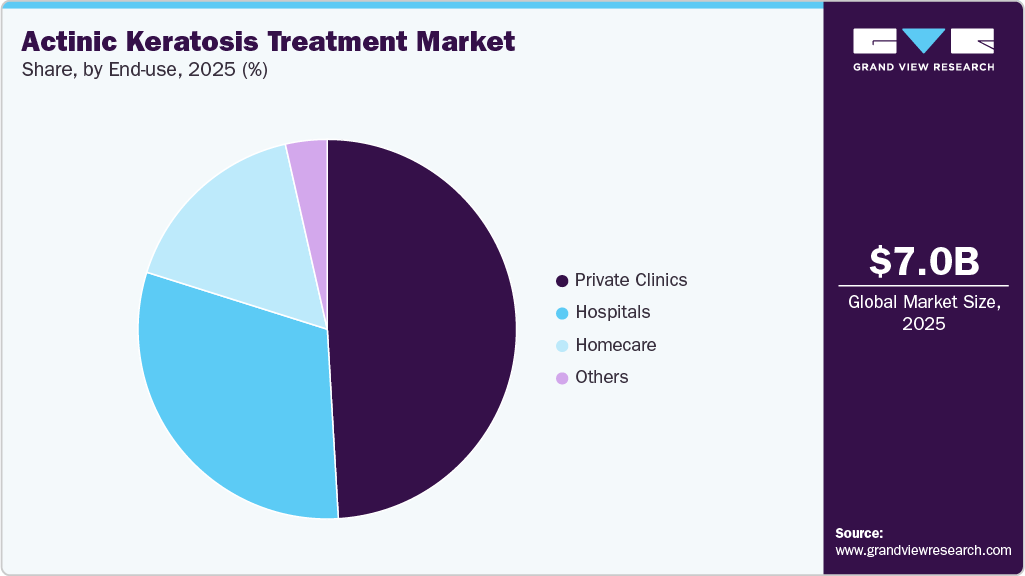
The homecare segment is projected to witness a lucrative growth rate over the forecast period, as more therapies are designed for self-administration outside clinical settings. Home-based treatment typically involves prescription topical medications that patients apply directly to affected skin areas under dermatological guidance. These therapies allow individuals to treat multiple lesions without frequent clinic visits, which is particularly beneficial for patients with widespread sun-damaged skin. As healthcare systems increasingly emphasize patient-centered and convenient care models, home-based management has become an important component of the AK treatment landscape.
Regional Insights
The North American actinic keratosis treatment market held the largest revenue share of 41.41% in 2025, driven by the high prevalence of skin cancer, skin damage from ultraviolet (UV) radiation of the sun, and the large established healthcare infrastructure that supports advanced dermatological treatments, particularly in the U.S. The region's robust research and development ability has led to readily available innovative therapies and drugs, further boosting the market growth.
The actinic keratosis is one of the most common precancerous skin conditions, affecting more than 58 million Americans. This creates a rising demand for medical treatment for actinic keratosis. Furthermore, actinic keratosis is the most frequent diagnosis and generates millions of physician visits annually, driving the demand for topical drug therapies in North America. The market is further supported by the growing adoption of topical field-directed treatments for improving treatment convenience and adherence. For example, the tirbanibulin ointment for actinic keratosis lesions can now be used on larger areas of the face and scalp of the patient, after approval from the FDA.
U.S. Actinic Keratosis Treatment Market Trends
U.S. actinic keratosis treatment market is driven by the growing prevalence of UV-induced skin lesions, strong dermatology care access, and increasing adoption of topical field therapies that treat multiple lesions simultaneously. The presence of major dermatologically focused pharmaceutical companies and continuous clinical development further strengthens market competition. For instance, in June 2024, the U.S. regulatory authority, FDA, approved an expanded indication for tirbanibulin ointment 1% (Klisyri) by Almirall. This ointment allows treatment of actinic keratosis on facial or scalp areas up to 100 cm², improving treatment flexibility and expanding its clinical use.
In addition, many companies, including Biofrontera, are working on expanding their products, investing in research and development, and running tests and trials for actinic keratosis treatments to cover more types of cases. In February 2026, Biofrontera reported strong results from its phase 3 clinical trial of Ameluz PDT for treating actinic keratoses on the arms, legs, neck, and trunk, showing clear and significant benefits. These activities show that companies are constantly innovating and bringing new treatments to the U.S. actinic keratosis market.
Europe Actinic Keratosis Treatment Market Trends
The Europe actinic keratosis drugs market in 2025 is driven primarily by an aging population, increased UV radiation exposure, and changing lifestyle behaviors. Actinic keratosis is a common condition diagnosed by dermatologists, with a prevalence of about 13.3% prevalence in the European population. Actinic keratosis is common among older adults, especially in countries with aging populations such as Germany, France, and the UK, where the risk increases due to repeated ultraviolet exposure. This large patient population continues to drive demand for topical treatments, including fluorouracil, imiquimod, and diclofenac. Leading dermatological companies are expanding their products through innovative label extensions and strategic regional partnerships. Biofrontera has significantly strengthened its European presence. For instance, in November 2025, Biofrontera partnered with Propharma d.o.o. to distribute Ameluz in Croatia, accelerating the company’s global expansion.
The UK actinic keratosis drugs market is fueled by an aging population, increased UV exposure, and the urgent need to address growing patient backlogs. Major pharmaceutical companies are strengthening their market positions through strategic label extensions and continuous commercial rollouts. Rising awareness of skin treatment and its benefits has led to higher treatment rates and increased demand for effective therapies options. Biofrontera significantly expanded its UK market potential. For instance, in February 2024, the Medicines and Healthcare Products Regulatory Agency (MHRA) approved a label extension for Ameluz to be used with artificial daylight. This low-pain photodynamic therapy option improves patient convenience by removing weather and time-of-day constraints.
Germany is a major market for actinic keratosis treatment in Europe, due to the high disease prevalence and robust dermatology care infrastructure. According to a study by PubMed, dermatologists diagnose over 1.7 million cases of actinic keratosis each year in Germany, indicating a notable patient pool requiring treatment. The prevalence increases with age, with about 11-12% of individuals aged 60-70 affected, reflecting the impact of the country’s aging population. Germany has strong skin cancer screening programs that detect early precancerous lesions like actinic keratosis. As a result, there is a growing demand for topical drugs such as 5-fluorouracil, imiquimod, diclofenac gel, and tirbanibulin. These trends are expected to drive market growth in Germany.
Asia-Pacific Actinic Keratosis Treatment Market Trends
The Asia Pacific region is seeing rapid growth in the actinic keratosis treatment market mainly because of improving healthcare infrastructure and the growing availability of advanced dermatological treatments. Many countries in the region are expanding hospital networks, dermatology clinics, and specialized skin treatment centers, which enhances access to early diagnosis and care. For example, countries like China, Japan, and South Korea have greatly expanded dermatology services in urban hospitals and private clinics, leading to increased use of treatments such as topical therapies, cryotherapy, and photodynamic therapy. Moreover, according to the International Trade Administration, China is the world’s second-largest healthcare market, valued at over USD 1 trillion in 2024 and expected to surpass USD 1.5 trillion by 2029. Government healthcare reforms and increased healthcare investments are also enhancing the availability of dermatological care.
The actinic keratosis treatment market in Japan is experiencing significant growth, primarily driven by increasing awareness of skin health, an aging population, and continuous advancements in dermatological technologies. Japan has one of the world’s oldest populations, and since actinic keratosis is more common in older adults with long-term sun exposure, the demand for effective treatments is rising. Technological advancements, including innovations in topical agents such as 5-fluorouracil and imiquimod, as well as photodynamic therapy (PDT), cryotherapy, and laser ablation, have improved both efficacy and safety profiles.
There is rapid expansion of dermatology services and specialized skin care clinics across major cities in China. Hospitals and private dermatology centers have significantly increased their capacity to diagnose and treat precancerous skin conditions, leading to greater adoption of modern actinic keratosis therapies. Large tertiary hospitals and dermatology-focused clinics are increasingly offering advanced treatments such as cryotherapy, photodynamic therapy, and prescription topical medications as part of routine dermatological care. For instance, Peking University Shenzhen Hospital has a well-established dermatology department that provides specialized management of precancerous skin lesions.
Latin America Actinic Keratosis Treatment Market Trends
The actinic keratosis treatment market in Central and South America is steadily growing, fueled by increased awareness of skin health, high UV exposure, and an aging population in the region. Many areas in Central and South America receive strong sunlight year-round due to their proximity to the equator, which results in more cases of sun-related skin issues such as actinic keratosis. Because of this, more people are being diagnosed and treated early for precancerous skin lesions at dermatology clinics and hospitals. Pharmaceutical companies such as Almirall S.A., Sun Pharmaceutical Industries, and Bausch Health Companies have grown in the region by working with local distributors and dermatology networks. This has made prescription topical medicines and field-directed therapies more widely available.
Middle East and Africa Actinic Keratosis Treatment Market Trends
High ultraviolet (UV) radiation exposure and the rising number of precancerous skin lesions are driving consistent demand for dermatological treatments in MEA actinic keratosis treatment market. In South Africa, extended sun exposure and a large outdoor workforce contribute to a significant burden of sun-related skin conditions, resulting in increased use of cryotherapy, topical medications, and photodynamic therapy in dermatology clinics. For instance, in January 2026, South Africa has the highest rate of non-melanoma skin cancer in Africa, with cases expected to increase by 28% by 2030 and 73% by 2040. Universal Healthcare, a leading medical scheme administrator in the country, also reported a 130% increase in members diagnosed with non-melanoma skin cancer over the past eight years.
Key Actinic Keratosis Treatment Company Insights
Some of the leading players operating in the actinic keratosis treatment market include 3M, Sun Pharmaceutical Industries Ltd., Novartis AG, Almirall S.A. Companies focus on capturing the market by increasing their presence using various business initiatives, such as partnerships & collaborations with government. Moreover, these companies have well established product portfolio, which helps capture major market share.
BIOFRONTERA Inc., Hill Dermaceuticals Inc, Ortho Dermatologics (Bausch Health Companies Inc.) are some of the emerging market participants in the actinic keratosis treatment market. Developing & launching new and improved products that offer faster results, higher accuracy, and easier usability are observed as prevalent operating strategies by these companies. The players may face challenges in penetrating the market due to competition and regulatory hurdles.
Key Actinic Keratosis Treatment Companies:
The following key companies have been profiled for this study on the actinic keratosis treatment market.
- Almirall, S.A.
- Brymill Cryogenic Systems
- Sun Pharmaceutical Industries Ltd.
- Cortex Technology
- Galderma
- Ortho Dermatologics (Bausch Health Companies Inc.)
- BIOFRONTERA Inc.
- Hill Dermaceuticals, Inc
- Teva Pharmaceutical
- Viatris Inc.
Recent Development
-
In April 2025, SKNV, a pioneer in dermatologic innovation and customized prescription medications, announced the launch of Kefunova Cream (Fluorouracil 5% / Calcipotriene 0.005%). This new prescription-only topical therapy was specifically developed to address the needs of dermatology providers in treating actinic keratosis (AK) and superficial basal cell carcinoma (sBCC).
-
In February 2026, Biofrontera Inc. announced positive and statistically significant top-line results from its Phase 3 clinical trial. The study evaluated Ameluz PDT in combination with the RhodoLED red-light platform for the treatment of mild to moderate actinic keratoses (AKs) on the extremities, neck, and trunk.
-
In September 2025, Almirall presented new clinical/real‑world data on tirbanibulin (Klisyri) at EADV 2025 confirming efficacy for larger AK fields.
Actinic Keratosis Treatment Market Report Scope
Report Attribute
Details
Market size in 2026
USD 7.27 billion
Revenue forecast in 2033
USD 10.13 billion
Growth rate
CAGR 4.86% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, volume in thousands and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Therapy, drug class, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Denmark, Sweden, Norway, China; Japan; India; Australia; Thailand, South Korea; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Almirall, S.A.; Brymill Cryogenic Systems; Sun Pharmaceutical Industries Ltd.; Cortex Technology; Galderma; Ortho Dermatologics (Bausch Health Companies Inc.); BIOFRONTERA Inc.; Hill Dermaceuticals, Inc; Teva Pharmaceutical; Viatris Inc.
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Actinic Keratosis Treatment Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the actinic keratosis treatment market on the basis of therapy, drug class, end use, and region:
-
Therapy Outlook (USD Million, 2021 - 2033)
-
Topical
-
Surgery
-
Photodynamic Therapy
-
-
Drug Class Outlook (USD Million; 2021 - 2033)
-
Nucleoside Metabolic Inhibitor
-
NSAIDs
-
Immune Response Modifiers
-
Photoenhancers
-
Others
-
-
End Use Outlook (USD Million,2021 - 2033)
-
Hospitals
-
Private Clinics
-
Homecare
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global actinic keratosis treatment market size was valued at USD 7.02 billion in 2025 and is projected to reach USD 7.27 billion in 2026.
b. The global actinic keratosis treatment market is expected to grow at a compound annual growth rate (CAGR) of 4.86% from 2026 to 2033, reaching approximately USD 10.13 billion by 2033.
b. Based on therapy, the topical treatment segment dominated the market with the largest revenue share of 46.98% in 2025, driven by its non-invasive nature, ease of application, and effectiveness in treating multiple lesions through field-directed therapy approaches.
b. Key players operating in the market include Almirall, S.A., Brymill Cryogenic Systems, Sun Pharmaceutical Industries Ltd., Cortex Technology, Galderma, Ortho Dermatologics (Bausch Health Companies Inc.), BIOFRONTERA Inc., Hill Dermaceuticals, Inc., Teva Pharmaceutical, and Viatris Inc.
b. Key factors driving market growth include the rising prevalence of actinic keratosis due to prolonged ultraviolet (UV) exposure, increasing aging population, and growing awareness regarding early diagnosis of precancerous skin conditions. Advancements in topical therapies, photodynamic therapy technologies, and minimally invasive procedures are further improving treatment outcomes and supporting market expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










