- Home
- »
- Healthcare IT
- »
-
Asia Pacific Clinical Trials Market Size, Industry Report, 2033GVR Report cover
![Asia Pacific Clinical Trials Market Size, Share & Trends Report]()
Asia Pacific Clinical Trials Market (2025 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Indication, By Study Design, By Service, By Indication by Study Design, By Sponsor, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-279-7
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Asia Pacific Clinical Trials Market Summary
The Asia Pacific clinical trials market size was valued at USD 16.82 billion in 2024 and is projected to reach USD 34.23 billion by 2033, growing at a CAGR of 8.46% from 2025 to 2033. The Asia Pacific clinical trials market is driven by a growing number of pharmaceutical and medical device companies, advancements in digital health, personalized medicine, and rising partnerships, which are expected to drive the market over the estimated period.
Key Market Trends & Insights
- The China clinical trials industry in the Asia Pacific is expected to grow significantly over the forecast period.
- By phase, the phase III segment led the market with the largest revenue share of 53.35% in 2024.
- Based on study design, the interventional trials segment led the market with the largest revenue share in 2024.
- By indication, the oncology segment held the highest market share in 2024.
- By sponsor, the pharmaceutical & biopharmaceutical companies segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 16.82 Billion
- 2033 Projected Market Size: USD 34.23 Billion
- CAGR (2025-2033): 8.46%
- China: Largest market in 2024
- Australia: Fastest growing market
In addition, supportive government initiatives, advanced regulatory approvals, and increased funding from pharmaceutical and biotech companies further contribute to the global market. Furthermore, collaborations, increased patient awareness, and improved trial accessibility are expected to drive the recruitment efforts, data quality, and success rates of trial populations and global regions. Furthermore, increasing collaboration between industry stakeholders, digital health innovators, and patient advocacy groups. Pharmaceutical companies are expanding their in-house R&D pipelines and actively partnering with CROs to streamline study design, site management, and regulatory submissions.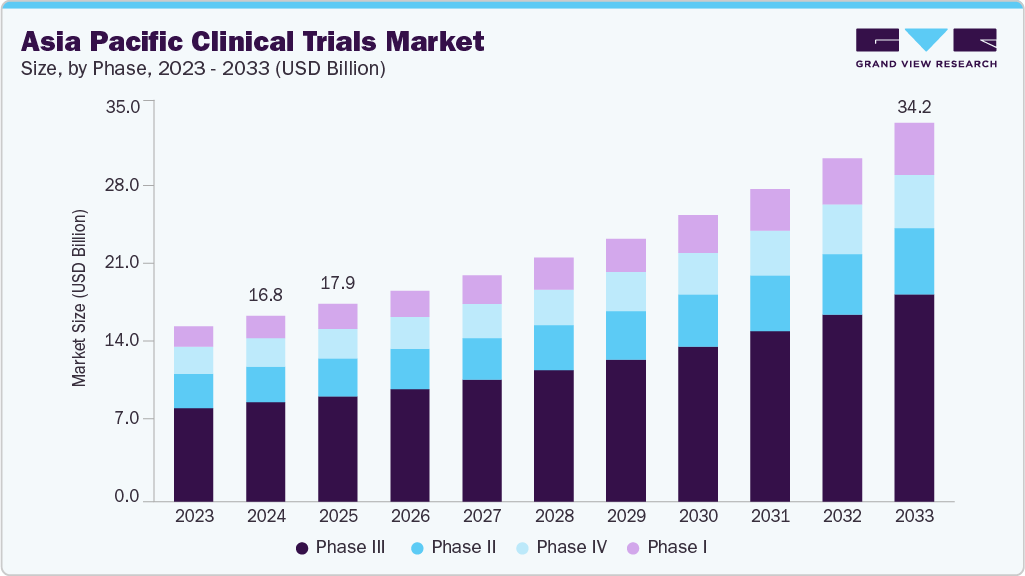
This trend allows sponsors to accelerate timelines without compromising data integrity or patient safety. Similarly, rising patient awareness and education, often supported by advocacy foundations and public health campaigns, have resulted in greater trial participation, particularly in complex areas such as oncology and rare diseases. The shift toward more inclusive enrollment strategies, supported by federal guidelines and diversity mandates, ensures clinical outcomes better reflect real-world populations.
Opportunity Analysis
The Asia Pacific region offers considerable new growth opportunities for clinical trials, attributed to its expansive and diverse patient base, reduced trial costs, and evolving regulatory environments. Countries such as China, India, South Korea, Japan, and Australia are emerging as key players in this field. Besides, the market is driven by governmental support, enhanced healthcare infrastructure, and rising participation in international trials. This region enables swift patient recruitment, particularly in oncology, neurology, and rare diseases. Furthermore, digital health innovations, real-world data integration, and decentralized trial models' adoption facilitate complex research across various locations. Regulatory bodies in countries like China and India are also simplifying approval processes, encouraging more global sponsors to engage in local trial operations. With a growing disease burden, an expanding middle class, and better access to healthcare, the Asia Pacific stands out as a strategic hub for both early- and late-phase clinical trials, solidifying its role in the international clinical research landscape.
Impact of U.S. Tariffs
U.S. tariffs have indirectly influenced clinical trials in the Asia Pacific region. Although clinical research services are generally exempt from tariffs, rising costs for pharmaceuticals, laboratory equipment, and medical devices imported from Asia Pacific have led U.S. sponsors to reevaluate their supply chain and operational strategies. Besides, some companies have relocated aspects of their trials, such as manufacturing, packaging, and data management, to the Asia Pacific to alleviate growing domestic expenses. This transition has led to new opportunities for regional CROs and trial sites, particularly in India, China, and South Korea. In addition, the need to diversify supply chains away from China due to tariff uncertainties has resulted in increased clinical trial activity in Southeast Asia and India. Besides, ongoing geopolitical tensions and regulatory challenges may present obstacles. Hence, the tariffs have disrupted established practices; however, they have also fueled greater investment and capacity development in clinical research within the Asia Pacific.
Technological Advancements
Technological innovations are transforming clinical trials in the Asia Pacific region, significantly improving efficiency, accuracy, and participant engagement. Decentralized Clinical Trials (DCTs) utilize tools such as telemedicine, electronic consent, and remote monitoring to minimize the necessity of site visits, thereby enhancing accessibility for participants in remote or underserved areas. Devices like smartwatches and biosensors enable continuous and real-time monitoring of vital signs and self-reported outcomes, facilitating thorough and precise data collection. Besides, artificial intelligence (AI) and machine learning enhance patient recruitment by scrutinizing extensive datasets, including electronic health records and genomic data, to swiftly identify suitable candidates, thereby reducing screening failures and expediting the enrollment process.

Electronic Data Capture (EDC) and eSource systems streamline data collection through digitization, promoting accurate real-time entry, which minimizes errors and accelerates monitoring and regulatory compliance. In addition, integrating Real-World Evidence (RWE) and big data enhances trial design by utilizing electronic medical records, insurance claims, and patient registries to improve patient selection and support post-market surveillance. These advancements are fostering more patient-centric, efficient, and data-driven clinical trials in the Asia Pacific region, ultimately speeding up the development of innovative therapies.
Pricing Analysis
Pricing models in clinical trials and related services are designed to accommodate various project requirements and client preferences. The Milestone-Based Pricing model links payments to achieving specific project milestones or deliverables, ensuring that clients only pay when agreed-upon results are met. This approach helps mitigate risk and promotes timely progress. Value-Based Pricing emphasizes the overall value or impact provided to the client, rather than focusing solely on costs or time. This model is particularly suitable for innovative services that can lead to significant improvements in outcomes. The Fixed-Fee Model offers a set price for the entire project, ensuring budget predictability and simplicity when the scope is clearly defined.
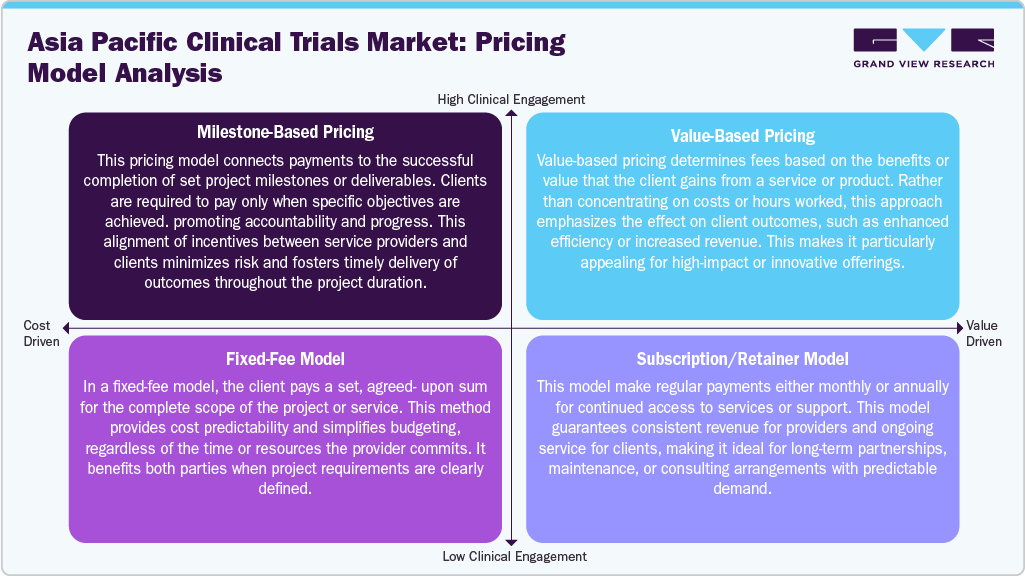
In addition, the Subscription or Retainer Model involves regular payments over a specified period, allowing clients continuous access to services or support. This model is effective for ongoing partnerships and maintenance needs. Thus, these pricing strategies provide unique flexibility and align incentives in different ways, enabling organizations to select the models that align with their strategic and financial objectives.
Phase Insights
On the basis of the phase segment, the phase III segment accounted for the largest revenue share in the Asia Pacific clinical trials industry of 53.35% in 2024. The segment growth is driven by increasing drug pipelines and rising unmet needs therapies and medical devices. These trials are designed to assess a treatment's safety and effectiveness over an extended period, typically ranging from one to four years. In addition, phase III trials tend to have more funding, leading to increased research activities, which allow for results and data comparisons of the drug’s effectiveness against existing treatments on the market.
The phase I segment is anticipated to grow at the fastest CAGR during the forecast period. The clinical research industry across the globe is experiencing a major transformation, driven by rapid technological advancements, changing regulatory environments, and the increasing need for more effective and inclusive clinical trials. As we move towards 2025 and beyond, various emerging trends are fueling the demand for phase I clinical trials, enhancing their efficiency, patient focus, and data utilization. These trials serve as the cornerstone for drug development and medical device innovations, focusing on safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) in a small group of healthy volunteers or patients, which is expected to support the market growth.
Study Design Insights
On the basis of the study design segment, the interventional trials segment held the largest market share in 2024. Interventional trials in the Asia Pacific region are rapidly growing due to increasing disease burden, enhanced clinical research infrastructure, and supportive government policies. Besides, the presence of countries such as China, India, South Korea, Japan, and Australia is leading these efforts, offering cost-effective operations and quick patient recruitment from diverse populations, which is appealing to global sponsors. Moreover, advances in technology, such as digital data capture and decentralized trial models, are optimizing these studies. As regulatory frameworks evolve, Asia Pacific is emerging as a key hub for Phase I-IV interventional clinical research.
The observational trials segment is anticipated to grow at the fastest CAGR over the forecast period. These studies are essential to outline robust protocols focused on the objectives, methods, and procedures for conducting a clinical trial. It serves as a roadmap for researchers to gain insights into the natural progression of diseases, identify potential risk factors, and assess the effectiveness of treatments in real-world data (RWD), patient-reported outcomes, and epidemiological endpoints contexts. Besides, these studies address ethical considerations, regulatory requirements, and potential challenges that may arise during the trial. Such factors are expected to witness rising demand over the estimated time period.
Indication Insights
On the basis of the indication segment, the oncology segment held the largest market share in 2024. Increasing cases of cancer globally have led to substantial investment in oncological drug development, positioning it as the largest revenue-generating segment for clinical trials. The most common cancers are breast, lung, prostate, and colorectal cancers. Moreover, recent trends have shown an immense migration in pharmaceutical and medical device products, with significant advancements made in the treatment of cancer. In addition, the outsourcing services such as CROs have experienced significant growth due to the rising demand for oncology drugs & therapies, fueled by innovative targeted treatments, immunotherapies, and personalized medicine approaches among pharmaceutical and medical device companies.
The autoimmune/inflammation segment is anticipated to grow at the second fastest CAGR over the forecast period. The clinical trials in autoimmune/inflammation represent a key milestone for innovating new devices and drug developments for autoimmune diseases. Besides, the ongoing innovative approaches for autoimmune diseases drug development are expected to drive the segment growth. For instance, as of April 2025, new immunotherapies are being developed at the University of Queensland that could allow individuals with RA and diabetes to live without the need for daily medication. In addition, the country is backing early-stage research into anti-inflammatory drugs, involving healthy volunteers in studies to assess safety and the body’s response. Such factors are expected to drive market growth.
Service Insights
On the basis of service segment, the laboratory services segment held the largest market share in 2024. Laboratory services are essential for supporting clinical trials across the Asia Pacific region, providing crucial functions such as sample analysis, biomarker testing, pharmacokinetics, and central lab operations. The increasing demand for precision medicine, biologics, and complex trial designs has heightened the need for specialized laboratory capabilities. Recent advancements in molecular diagnostics, automation, and data integration have significantly improved lab services' accuracy and efficiency. Furthermore, strategic partnerships between CROs and laboratory networks are optimizing operations. As clinical trial activities continue to grow, robust and scalable laboratory services are becoming vital for ensuring data quality and regulatory compliance.
The patient recruitment segment is anticipated to grow at the fastest CAGR over the forecast period. Patient recruitment for clinical trials in the Asia Pacific region is gaining momentum, driven by the area's large and diverse population along with a rising disease burden. Enhanced healthcare access, increased awareness, and the use of digital outreach tools are contributing to improved enrollment rates. Technologies such as AI-driven matching algorithms and mobile health platforms are streamlining the recruitment process. Furthermore, the growing clinical research and supportive regulatory frameworks are positioning the region as a strategic option for efficient and timely patient enrollment in global trials.
Sponsor Insights
On the basis of the sponsor segment, the pharmaceutical & biopharmaceutical companies segment held the largest market share in 2024. The pharmaceutical & biopharmaceutical industry is subject to strict regulatory requirements, and clinical trials are critical to ensure compliance with regulatory standards. This has led many companies to outsource their clinical trials to improve efficiency, reduce costs, and shorten timelines. Besides, increased patient populations, regulatory expertise, and the availability of advanced technologies are fueling the demand for outsourcing services among pharmaceutical & biopharmaceutical companies.
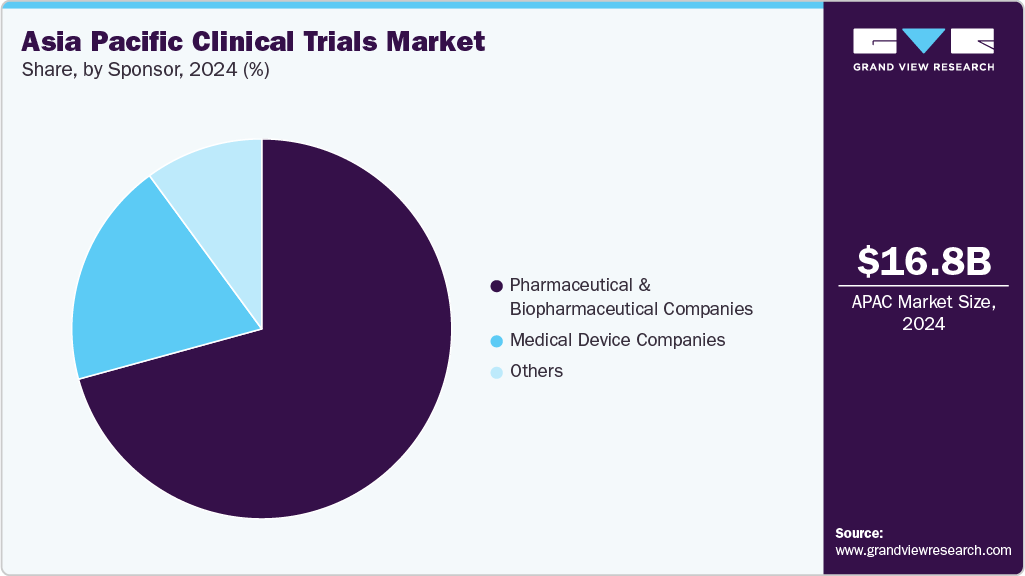
The medical device companies segment is anticipated to grow at the second fastest CAGR over the forecast period. Currently, in the healthcare industry, the necessity of embracing industry innovation while optimizing resource allocation has created a robust requirement for outsourcing services among medical device companies across multiple functional areas along the product development value chain. Several medical device companies prefer to outsource clinical trials to CROs to lower development costs, accelerate the time-to-market, and access specialized knowledge in regulatory compliance, trial design, and data management. Besides, ongoing technological advancements, a changing regulatory environment, and strategic shifts in medical devices are anticipated to drive the market growth.In addition, OEMs rely on outsourcing for clinical trials and regulatory and quality support, contributing to market growth. The clinical trials market provides medical device companies with the advantages of faster market entry and cost reduction while optimizing internal operations. Such factors are expected to drive the market over the estimated time period.
Country Insights
Japan Clinical Trials Market Trends
The clinical trials market in Japan is driven by its advanced healthcare infrastructure, robust regulatory framework, and commitment to high standards of clinical data quality. The Pharmaceuticals and Medical Devices Agency (PMDA) playsa pivotal role in enhancing compliance and trial efficiency. The country is increasingly participating in global multicenter trials, particularly in fields such as oncology, neurology, and rare diseases. With an aging population and a rising demand for personalized medicine, the country is also supportive of early-phase studies. Thus, the adoption of electronic data systems, decentralized trial models, and the use of real-world evidence are expected to drive the country as a key destination for clinical research.
China Clinical Trials Market Trends
China's clinical trials market is experiencing rapid growth, driven by its vast patient population, regulatory reforms, and increasing investments in healthcare. The National Medical Products Administration (NMPA) streamlined the approval processes, significantly reducing trial initiation timelines. The rising demand for treatments in oncology, chronic diseases, and rare disorders contributes to the escalating volume of clinical trials. In addition, advancements in digital health and decentralized trial models enhance data quality and patient accessibility. Moreover, robust government support and partnerships with global pharmaceutical companies further strengthen the clinical research ecosystem. Thus, the country is emerging as a preferred destination for both early- and late-phase clinical trials, efficiently addressing the growing local and international demand.
India Clinical Trials Market Trends
The clinical trials market in India is driven by a large and diverse population, cost-efficient operations, and an evolving regulatory landscape. Recent reforms have simplified the approval processes, making it quicker and easier to initiate trials. Besides, trends include the rise of decentralized clinical trials that leverage digital tools, such as telemedicine and e-consent, to enhance patient reach, particularly in rural areas. Furthermore, integrating AI and machine learning enhances patient recruitment and data analysis efficiency. There is an increasing emphasis on addressing conditions such as cancer, diabetes, and rare disorders, aligning with the shifting healthcare needs in India.
Key Asia Pacific Clinical Trials Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures merger, partnership & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key Asia Pacific Clinical Trials Companies:
- IQVIA Inc.
- Parexel International Corporation
- Thermo Fisher Scientific Inc.
- ICON Plc.
- LabCorp
- Syneos Health
- Novotech
- Avance Clinical
- AbbVie
- Merck Sharp & Dohme LLC
Recent Developments
-
In February 2025, LabConnect entered into a strategic alliance with Australian Clinical Labs' (ACL) Specialised Trials to enhance support for clinical trials in Australia and the Asia-Pacific region. This multi-year agreement strengthens their collaboration, allowing both organizations to utilize their complementary strengths to address the increasing regional demand for clinical trial support services for pharmaceutical, biotechnology, and clinical research institutions, ultimately helping patients access the medications they need.
-
In December 2024, Sunway Healthcare Group collaborated with Syneos Health to enhance clinical trial capabilities across various therapeutic areas in Malaysia. The partnership focuses on improving patient access, increasing operational efficiencies, providing specialized expertise, and engaging with diverse patient populations throughout all therapeutic areas and phases.
-
In January 2024, Parexel International Corporation mentioned its partnership with the Japanese Foundation for Cancer Research, dedicated to cancer research. This strategic alliance aimed to enhance the accessibility of oncology clinical trials in Japan.
Asia Pacific Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 17.87 billion
Revenue forecast in 2033
USD 34.23 billion
Growth rate
CAGR of 8.46% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication, indication by study design, service, sponsor, country
Country scope
Japan; China; India; Thailand; South Korea; Australia
Key companies profiled
IQVIA Inc.; Parexel International Corporation; Thermo Fisher Scientific Inc.; ICON Plc.; LabCorp; Syneos Health; Novotech; Avance Clinical; AbbVie; Merck Sharp & Dohme LLC
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Asia Pacific Clinical Trials Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Asia Pacific clinical trials market report based on phase, study design, indication, indication by study design, service, sponsor, and country:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Others
-
-
CNS Conditions
-
Epilepsy
-
Parkinson's Disease (PD)
-
Huntington's Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular Diseases
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Pain Management
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Oncology
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
CNS Conditions
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Diabetes
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Obesity
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Cardiovascular Diseases
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Others
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Laboratory Services
-
Analytical Testing Services
-
Clinical Trial Data Management Services
-
Others
-
-
Sponsor Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
South Korea
-
Australia
-
-
Frequently Asked Questions About This Report
b. The Asia Pacific clinical trials market size was estimated at USD 16.82 billion in 2024 and is expected to reach USD 17.87 billion in 2025.
b. The Asia Pacific clinical trials market is expected to grow at a compound annual growth rate of 8.46% from 2025 to 2033 to reach USD 34.23 billion by 2030.
b. The phase III segment dominated the market, with a share of 53.35% in 2024. The segment growth is driven by increasing drug pipelines and rising unmet needs therapies and medical devices. These trials are designed to assess a treatment's safety and effectiveness over an extended period, typically ranging from one to four years. Besides, the trials tend to have more funding, leading to increased research activities, which allow for results and data comparisons of the drug’s effectiveness against existing treatments on the market.
b. Some key players operating in the Asia Pacific clinical trials market include IQVIA Inc., Parexel International Corporation, Thermo Fisher Scientific Inc., ICON Plc., LabCorp, Syneos Health, Novotech, Avance Clinical, AbbVie, and Merck Sharp & Dohme LLC., among others.
b. Factors such as the growing number of pharmaceutical and medical device companies, advancements in digital health, personalized medicine, increasing prevalence of chronic diseases, and the rising number of partnerships are driving the growth of the Asia Pacific clinical trials market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










