- Home
- »
- Medical Devices
- »
-
Biologics Contract Development And Manufacturing Organization Market Report 2033GVR Report cover
![Biologics Contract Development And Manufacturing Organization Market Size, Share & Trends Report]()
Biologics Contract Development And Manufacturing Organization Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Monoclonal Antibodies, Recombinant Proteins & Enzymes), By Service, By Source, By Workflow, By Therapeutic Area, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-773-0
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Biologics Contract Development And Manufacturing Organization Market Summary
The global biologics contract development and manufacturing organization market size was estimated at USD 47.84 billion in 2025 and is projected to reach USD 84.91 billion by 2033, growing at a CAGR of 7.4% from 2026 to 2033. The market is driven by the rising prevalence of chronic and rare diseases, an aging global population, and the growing demand for highly targeted therapies.
Key Market Trends & Insights
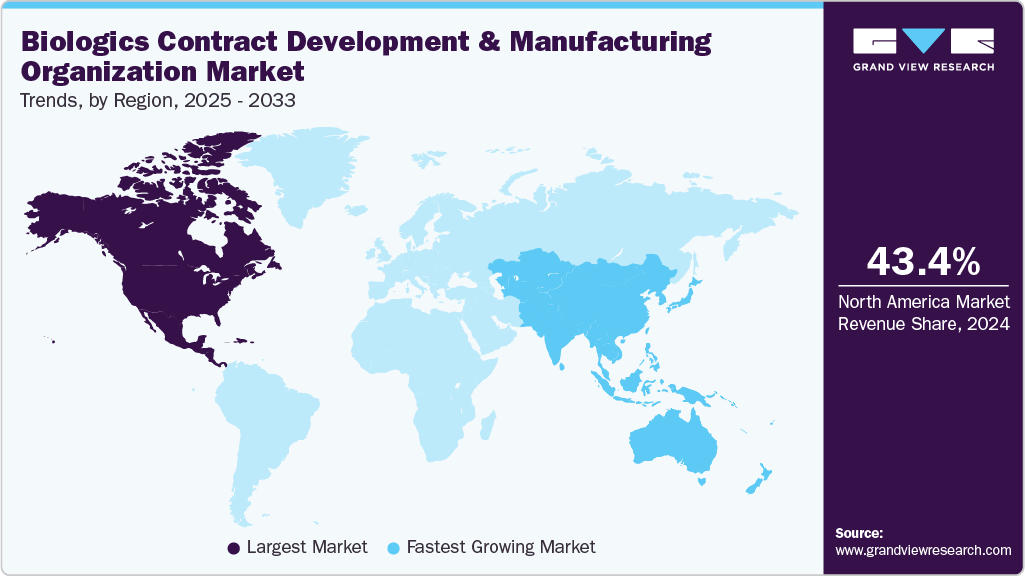
- North America biologics contract development and manufacturing organization (CDMO) market held the largest share of 43.78% of the global market in 2025.
- The biologics contract development and manufacturing organization (CDMO) in the U.S. is expected to grow significantly over the forecast period.
- By product, the monoclonal antibodies (mAbs) segment held the largest market share of 41.79% in 2025.
- By service, the contract manufacturing segment held the largest market share in 2025.
- By source, the microbial segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 47.84 Billion
- 2033 Projected Market Size: USD 84.91 Billion
- CAGR (2026-2033): 7.4%
- North America: Largest market in 2025
Besides, increasing biosimilar adoption provides cost-effective alternatives while expanding patient access. In addition, some other factors contributing to market growth are rising leadership teams that align network strategies with long-term therapeutic pipelines, next-generation manufacturing technologies, scientific breakthroughs, regulatory shifts, supply chain disruptions, and shifting patient demographics. Thus, these factors are creating a robust demand for the biologics pipeline in the pharmaceutical industry.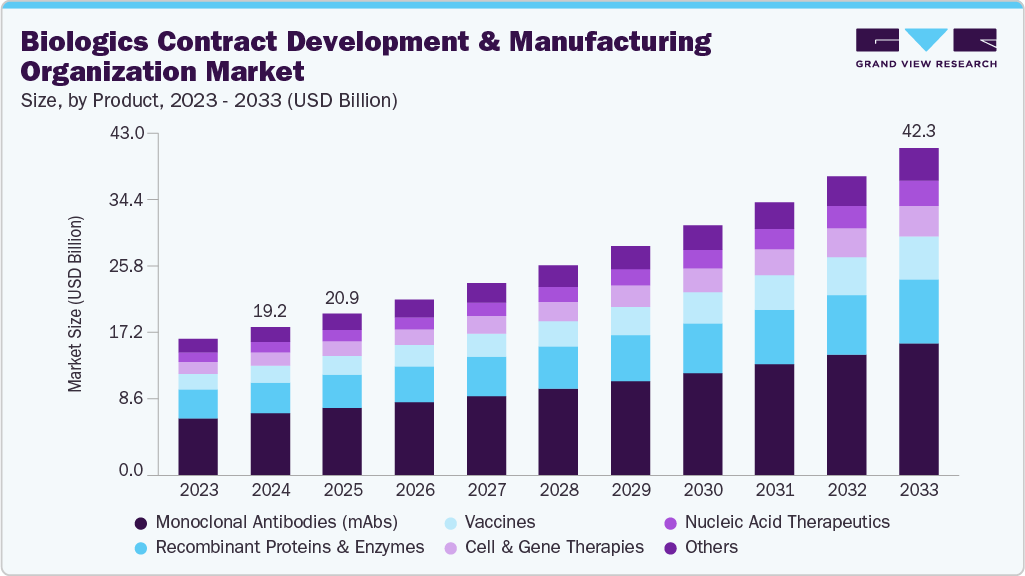
Moreover, most pharmaceutical & biopharmaceutical companies are increasingly partnering with CDMO service providers to expand capacity and access specialized technologies, while private equity and institutional investors are expanding their funds into platform companies with innovative models. Furthermore, growing investments in biologics are expected to drive venture capital, strategic collaborations, mergers, and acquisitions, further contributing to market growth across the value chain. Most pharmaceutical & biopharmaceutical companies are increasingly partnering with CDMO service providers to expand capacity and access specialized technologies.
According to the data published by Fierce Pharma in June 2025, several CDMOs are announcing significant expansions to meet rising biologics outsourcing demand: WuXi Biologics recently announced the opening of a new 95,000 m² facility in China that likely to feature a 15,000-liter fermenter and annual output exceeding 10 million vials once operational, while Aragen is preparing to begin GMP biologics manufacturing at its Bangalore facility with high-productivity fed-batch platforms later this year. These expansion initiatives highlight how capacity scaling and long-term manufacturing deals directly respond to the complex production needs of biologics developers across regions.
In addition, technological advancements are reshaping the development of medications, including biologics with complex modalities such as bispecific antibodies, antibody-drug conjugates, CAR-T therapies, and mRNA-based treatments, further fueling strategic investments and drug innovation. Besides, there is a growing breakthrough in bioprocessing, including the use of single-use bioreactors, continuous manufacturing, and advanced process modeling, which have significantly improved efficiency & scalability. Moreover, growing integration of digitalization, automation, and artificial intelligence into drug discovery, development, and manufacturing processes further enables faster timelines and increased product success rates. Thus, these innovations enhance therapeutic precision and boost new growth opportunities for personalized and small-batch production to meet niche patient needs.
In addition, most large pharmaceutical companies are focusing on acquisitions to strengthen their biologics pipelines, diversify therapeutic portfolios, and secure advanced manufacturing capabilities. Thus, these capital investments and strategic innovations are accelerating commercialization and reshaping the competitive landscape, and are expected to drive the market over the estimated period.
In addition, amid growing regulatory scrutiny, the U.S. FDA, EMA, and other global authorities are refining approval pathways for cell therapies, biosimilars, and gene-based treatments, issuing guidelines to encourage innovation while safeguarding patient safety. Besides, harmonized standards across regions support streamlining submissions and reducing delays, while post-market surveillance & evidence requirements remain stringent in the market. The evolving regulatory environment enables faster approvals for novel therapies while increasing the compliance burden on developers to meet rigorous chemistry, manufacturing, and control requirements. Such factors are expected to drive the market over the estimated time period.
Opportunity Analysis
The biologics CDMO market is experiencing significant growth due to the rapid expansion of biologics pipelines, such as monoclonal antibodies, bi-specifics, and gene therapies. Besides, increased investment in R&D and a rising demand for outsourced manufacturing further contribute to allowing access to advanced production capabilities without incurring heavy capital costs. In addition, growing technological innovations such as continuous bioprocessing, single-use bioreactors, and automated cell culture systems are contributing to increased efficiency, cost reductions, and new service offerings. Moreover, growing emerging markets and the rising adoption of biologics for treating chronic and rare diseases are expanding the requirement for CDMO service providers and pharmaceutical and biotechnology companies. Furthermore, leading CDMOs are pursuing strategic collaborations and developing their capacities to secure early-stage development contracts and scale up commercial production. Besides, most service providers offer flexible, high-quality, and technologically advanced solutions, positioned to benefit from the increasing trend toward outsourcing, catering to niche and large-scale manufacturing needs. Thus, the biologics CDMO market is anticipated to witness new growth opportunities.
Impact of U.S. Tariffs on the Global Biologics Contract Development And Manufacturing Organization (CDMO) Market
The impact of tariffs and international trade policies on the biologics CDMO market is a critical factor influencing operational costs, supply chain strategies, and client pricing. Biologics manufacturing relies heavily on the import of high-value raw materials, specialized equipment, and advanced single-use technologies, many of which are sourced from global suppliers. Increases in import duties or the imposition of trade tariffs can significantly raise production costs, especially for CDMOs that operate on thin margins during early-stage development projects. Tariff fluctuations create uncertainty in budgeting and can affect decisions regarding the location of manufacturing facilities, sourcing of critical reagents, and allocation of resources across regions.
Tariffs also influence geographical client strategies. For instance, North American and European CDMOs are often affected by tariffs on imported stainless steel, bioreactor components, or chromatography resins, which may prompt clients to negotiate higher fees to offset the increased costs. Similarly, CDMOs in Asia-Pacific or Latin America may adjust pricing models in response to regional import duties, making these markets more attractive for clients seeking cost-efficient options. However, reliance on lower-cost regions comes with regulatory and logistical considerations that may offset savings. On the other hand, some companies view these challenges as an opportunity to support manufacturing capabilities, diversify their supply chains, and invest in automation to mitigate the impacts of tariffs. Thus, U.S. tariffs significantly transform operational strategies and competitive positioning within the global biologics CDMO landscape.
Technological Advancements
Technological advancements such as upstream bioprocessing, downstream processing, digital and data-driven and integrated end-to-end manufacturing platforms are significantly transforming the landscape of the biologics CDMO market. In the market, technological advancements in biologics CDMOs are transforming biomanufacturing toward higher efficiency, flexibility, and data integration.

In the market, upstream bioprocessing technologies have evolved through high-yield cell line engineering, perfusion systems, and single-use bioreactors. Process intensification and optimized media improve productivity while reducing costs and contamination risks. Continuous upstream processing further enhances scalability and consistency. Besides, downstream processing technologies are advancing with high-capacity chromatography resins, membrane filtration, and continuous purification systems. Innovations such as single-use technologies and inline buffer dilution reduce processing time and operational costs. Continuous downstream integration minimizes bottlenecks and improves overall yield and efficiency.
Moreover, digital and data-driven technologies are becoming central to CDMO operations. Artificial intelligence (AI), machine learning (ML), and advanced analytics enable process optimization, predictive maintenance, and real-time monitoring. Digital twins and connected systems allow simulation and control of manufacturing processes, improving quality and reducing deviations.
Furthermore, integrated end-to-end manufacturing platforms are emerging as a competitive advantage. CDMOs now offer seamless services from cell line development to fill-finish, supported by co-located facilities and unified data systems. This integration reduces technology transfer risks, accelerates timelines, and enhances product quality consistency. Besides, emerging trends and future outlook highlight a shift toward modular facilities, continuous biomanufacturing, and personalized medicine production. “Scale-out” models for cell and gene therapies, along with flexible manufacturing platforms, are gaining traction. Increasing adoption of automation and digitalization is expected to further drive efficiency, while integrated CDMOs are expected to become strategic partners in drug development. Such factors are expected to drive the market growth.
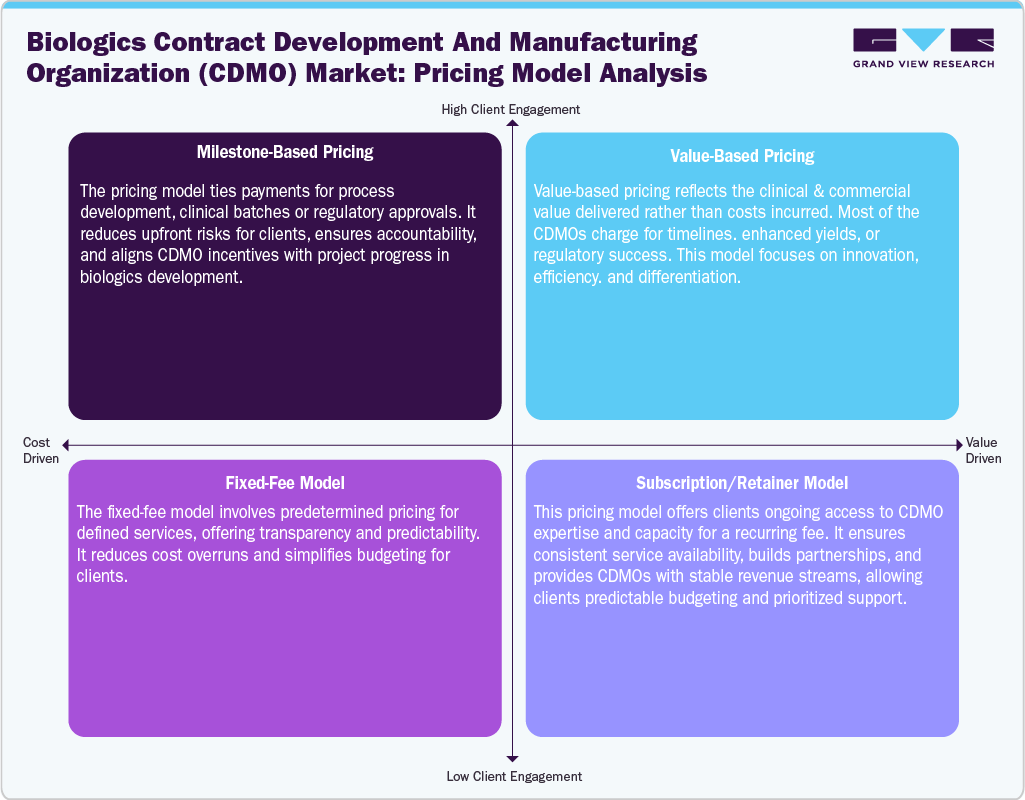
Pricing Model Analysis
The biologics CDMOs market employs various pricing models to balance client requirements, project risks, and value delivery. Biologics CDMOs use diverse pricing models to align with client needs, project complexity, and risk allocation. The cost-plus pricing model is widely adopted, where clients pay the actual production cost plus a predefined margin. It offers transparency and is suitable for early-stage or uncertain projects but may limit cost efficiency incentives.The milestone-based pricing model links payments to specific development or manufacturing achievements, such as cell line development or regulatory approval. This approach improves cash flow management for clients and aligns progress with payments. The fixed-fee pricing model involves a predetermined price for defined services. It provides budget certainly and is used for standardized or late-stage manufacturing, though it may lack flexibility for scope changes. The risk-sharing pricing model distributes financial risk between the CDMO and client, often tying compensation to product success or performance. This model strengthens long-term partnerships and is increasingly used for innovative biologics and advanced therapies.
Market Concentration & Characteristics
The biologics contract development and manufacturing organization (CDMO) market growth stage is high, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
Innovation drives competitiveness as CDMOs adopt advanced bioprocessing, single-use technologies & next-generation biologics such as bispecific antibodies and ADCs. These advancements improve efficiency, accelerate timelines, and enable adaptability, making CDMOs critical partners for complex biologics development and manufacturing projects.

M&A activities consolidate expertise, expand capabilities, and enhance geographic presence. In addition, strategic acquisitions provide access to advanced technologies and biologics-focused facilities. This integration strengthens competitiveness enriches service offerings and accelerates entry into growing biologics market.
Regulatory compliance shapes biologics CDMO operations through stringent GMP, biosafety, and therapeutic guidelines. Besides, meeting these requirements impacts costs, approvals, and timelines. CDMOs with strong regulatory expertise manage risks effectively, achieve faster approvals, and deliver reliable, compliant solutions for global biologics clients.
Biologics CDMOs expand services from early development to large-scale manufacturing, covering analytics, cell line development, and commercialization. Comprehensive portfolios attract major biopharma clients, reduce dependency on single offerings, and drive long-term partnerships, enabling CDMOs to create more substantial value throughout the biologic’s lifecycle.
Regional expansion supports the market to access, boost the revenue and create cost-effective manufacturing hubs. Besides, establishing presence in emerging regions enhances supply chain resilience, regulatory compliance, and scalability, empowering CDMOs to serve multinational clients and strengthens competitiveness in the global biologics market.
Product Insights
The monoclonal antibodies (mAbs) segment held the largest revenue share of 41.79% in 2025. The segment growth is attributed to superior physicochemical properties, including high expression yields in mammalian cell cultures, excellent solubility, and robust stability during processing, facilitate efficient scale-up and cost-effective production. These factors are expected to make mAbs advantageous for outsourced development and manufacturing models, driving growth for the biologics CDMO market.
In addition, currently, mAbs is expected to be cornerstone of the biopharmaceutical industry with strong approval rates and sales performance over the estimated time period. Besides, as a drug class, mAbs are among the most profitable, with four of the top ten medications across the globe. For instance, according to bioprocess international, currently 21 mAb candidates are under regulatory review, with approvals expected for at least two by next year. In addition, the approval rate of ten mAbs products is expected to be featured over 180 drugs by 2025. Furthermore, several COVID-19–targeted mAb therapies are anticipated to enter the market within the following year. Thus, growing innovations are expected to drive the market.
The nucleic acid therapeutics segment is expected to grow significantly during the forecast period. The segment growth is driven by its ability to provide targeted, personalized treatments for genetic and rare diseases. Besides, advances in delivery technologies, the success of mRNA vaccines, growing biopharma R&D investments & supportive regulatory frameworks are expected to drive the development of these therapeutics. Thus, these factors are expected to enhance adoption, expand pipelines, and position nucleic acid therapies over the estimated period.
Moreover, with the expanding pipeline of nucleic acid therapeutics, the biologics CDMO is expected to witness new growth opportunities over the estimated time period. For instance, in April 2025, AGC Biologics announced the launch of a new dedicated cell and gene therapy business division. The new division will focus on elevating the existing AGC biologics capabilities and supporting developers in need of capacity, scientific capabilities, and technically qualified cell and gene CDMO operators.
Service Insights
The contract manufacturing segment accounted for the largest share in 2025. The segment is driven by the rising complexity in biologics development, rising R&D costs, and the demand for faster time-to-market. In addition, contract manufacturing offers a range of services that allow companies to access advanced technologies, specialized expertise, and scalable manufacturing without heavy capital investment, further fueling the innovation in the market. Moreover, CDMOs provide end-to-end services, including process development, clinical and commercial manufacturing, and quality assurance for innovating biologics products. Furthermore, the contract manufacturing offers reduced capital expenditure, mitigating the operational risks and making it a strategic approach for emerging biotech companies & established pharmaceutical companies seeking to optimize production and maintain compliance with stringent regulatory standards.
The contract development segment is expected to grow significantly during the forecast period. The Contract development in the pharmaceutical and biotechnology industry involves outsourcing various drug or biologics production stages to specialized CDMOs encompassing cell line development, formulation, process development, analytical testing, and clinical batch production. By collaborating with CDMOs, companies gain advanced technical expertise, cutting-edge technologies, and extensive regulatory knowledge without the heavy financial burden. This approach enable companies speed up their timelines, lower costs, and minimize operational risks while adhering to high-quality standards. For instance, in October 2025, Rezon Bio mentioned the launch as a European CDMO for biologics. It enters the market as a new brand; however, it builds on a proven legacy of advancing biologics from gene to global commercialization through established partners.
Thus, contract development supports companies in efficiently moving their drug candidates from early discovery phases to clinical trials and commercial production. Such factors are expected to drive the market.
Source Insights
The microbial segment dominated the market with the largest revenue share in 2025. The segment is driven by the increasing requirement for cost-effective microbial production, offering higher yields at lower costs than mammalian systems. Besides, rising demand for vaccines, enzymes, and therapeutics is expected to drive the market over the estimated period. In addition, growing regulatory support with microbial platforms facilitates faster approvals, making them a preferred choice for developing safe, reliable, and commercially viable biologics. Moreover, advances in genetic engineering, fermentation technologies, and process optimization have enhanced yield, safety, and product consistency. Such factors are expected to drive segment growth.
The mammalian segment is projected to grow at a significant CAGR duringthe forecast period. The segment is driven by the rising need for complex, high-quality therapeutics requiring proper protein folding and post-translational modifications. Besides, growing advances in cell line engineering, bioreactor technologies, and process optimization that support enhanced yield, scalability, and consistency are expected to drive the segment growth. In addition, some other factors contributing to segment growth are the rising demand for monoclonal antibodies, recombinant proteins, and personalized therapies, which further contribute to market growth. In addition, the increasing prevalence of chronic, rare, and complex diseases drives investment in mammalian platforms, further supporting the segment growth.
Workflow Insights
The commercial segment accounted for the largest revenue share in 2025, driven by the growing demand for safe and effective therapeutics, including monoclonal antibodies, vaccines, and recombinant proteins. Besides, the increasing prevalence of chronic, rare, and complex diseases supports market growth. In addition, increasing advances in manufacturing technologies, process optimization, and quality control ensure consistent, high-quality output, which fuels the segment growth. In addition, regulatory support and streamlined approval pathways accelerate market entry. Moreover, increasing investments from pharma/biopharma companies and expanding healthcare infrastructure globally are boosting the market. Thus, these factors are expected to drive the market.
The clinical segment is expected to grow significantly during the forecast period. The growth is attributed to an expanding pipeline of innovative therapies, including monoclonal antibodies, cell and gene therapies, and recombinant proteins. Besides, rising R&D investments, advances in preclinical and clinical development technologies, and regulatory incentives accelerate progression from discovery to trials, which are expected to support the market. In addition, the Growing demand for treatments targeting rare, chronic, and complex diseases further fuels the clinical biologics development, driving the market. Thus, such factors are expected to drive new growth opportunities over the estimated period.
Therapeutic Area Insights
The oncology segment accounted for the largest share in 2025, driven by rising cancer prevalence, increasing patent expirations, growing demand for innovative treatments, and high demand for targeted therapies, including monoclonal antibodies, antibody-drug conjugates, and cell & gene therapies. For instance, the Cancer Atlas mentioned that the number of cancer cases is expected to reach 29 million globally by 2040. This is expected to drive the development of innovative treatments, creating opportunities for outsourced manufacturing and development services. Besides, CDMOs support oncology companies in achieving faster market entry from early-stage process development and clinical batch production to commercial-scale manufacturing, further contributing to segment growth. Moreover, advances in biologics platforms, high-potency manufacturing, and regulatory expertise enable CDMOs to deliver complex oncology therapeutics efficiently, ensuring quality, scalability, and faster time-to-market for addressing the market needs.
The autoimmune diseases segment is expected to grow at a significant CAGR during the forecast period. The rising prevalence of rheumatoid arthritis, psoriasis, and multiple sclerosis drives the segment. This has led pharmaceutical and biotech companies to shift towards CDMOs with increasing patient requirements for targeted and effective therapies. Moreover, most CDMO companies offer a range of advanced technologies in monoclonal antibodies, fusion proteins, and novel biologics that improve efficacy and safety profile, which further drives the market. Thus, innovation, adoption, and expansion of biologics for autoimmune disease are expected to drive the segment growth over the estimated period.
End Use Insights
The pharmaceutical companies segment accounted for the largest share in 2025. Pharmaceutical companies play a crucial role in driving the biologics CDMO market by outsourcing development and manufacturing to leverage specialized expertise, advanced technologies, and scalable production capabilities. Besides, rising R&D investments, demand for complex biologics, and the need to accelerate time-to-market encourage partnerships with CDMOs, which further contributes to market growth. Moreover, most of the pharma companies are benefited benefit from cost efficiencies, risk mitigation, and regulatory compliance support, enabling faster commercialization of therapies. Thus, such factors are expected to drive the market over the estimated time period.
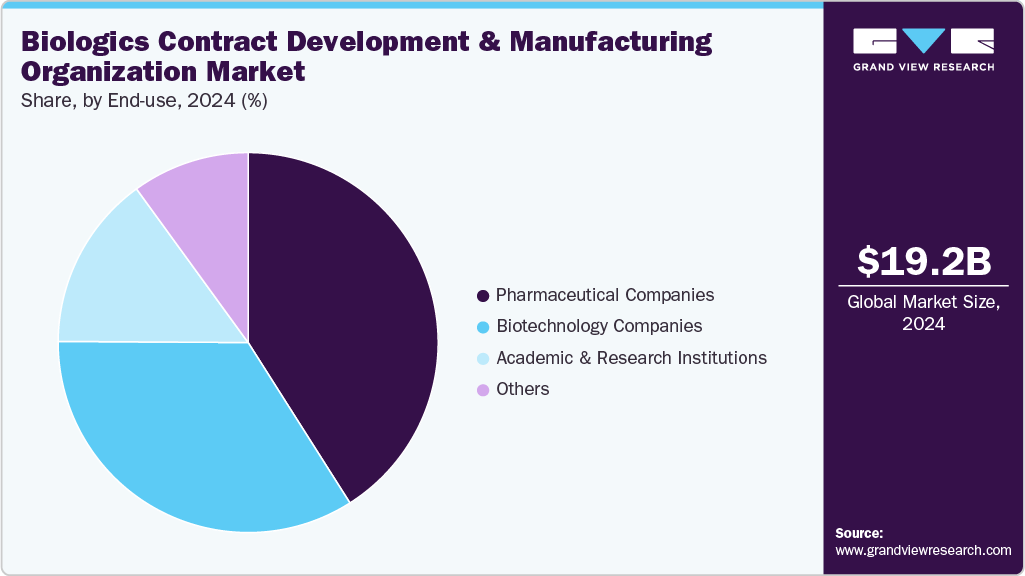
The biotechnology companies segment is expected to grow at the fastest CAGR during the forecast period. Biotechnology companies drive growth in the biologics CDMO market by developing innovative therapies such as monoclonal antibodies, nucleic acid therapeutics, and cell & gene therapies. Besides, most of the biotechnology companies lack the in-house manufacturing capabilities, often leading to partnership with CDMOs for process development, clinical, and commercial-scale production. These collaborations provide access to specialized expertise, advanced technologies, and regulatory support, enabling faster time-to-market. Moreover, rising investments, focus on personalized medicine, and the need for scalable, cost-effective production further contribute to segment growth.
Regional Insights
North America Biologics Contract Development And Manufacturing Organization Market Trends
North America dominatedthe global biologics contract development and manufacturing organization (CDMO) market in 2025, holding a revenue share of 43.78%. The market growth is attributed to presence of strong biopharmaceutical infrastructure, high R&D investment and increasing presence of biotech and pharmaceutical companies. Besides, presence of advanced manufacturing facilities, skilled workforce, and increasing adoption of innovative technologies drive the region’s innovations of biologics products. In addition, favorable regulatory frameworks, and high healthcare expenditure support the market. Moreover, increasing demand for monoclonal antibodies, cell & gene therapies, and personalized medicines are anticipated to drive the biologics outsourcing, innovation, and commercialization in the region. For instance, in January 2026, AGC Biologics partnered with Asimov in Milan, deploying the LV Edge Packaging system to streamline lentiviral vector production, reducing plasmid complexity, costs, and accelerating global cell and gene therapy development.
U.S. Biologics Contract Development And Manufacturing Organization Market Trends
The biologics contract development and manufacturing organization (CDMO) market in the U.S. accounted for the largest market share in the North America market, owing to strong presence of established biotech and pharmaceutical companies, increased R&D investments, and advanced manufacturing infrastructure. Besides, early adoption of cutting-edge technologies, including single-use bioreactors, continuous bioprocessing, and gene therapy platforms, supports innovative, robust biologics production. Moreover, favorable regulatory frameworks and streamlined approval pathways accelerate the commercialization of monoclonal antibodies and personalized medicines, further creating the U.S. as a critical hub for biologics innovation. Such factors are expected to drive the U.S. market.
The Canada biologics contract development and manufacturing organization market is expected to grow at a significant CAGR during the forecast period.The market is driven by increasing biotech activities, supportive government policies, and growing advancements in manufacturing facilities. Besides, most companies are opting for CDMO partnerships for both clinical and commercial-scale biologics production, benefiting from regulatory guidance and cost-effective solutions. In addition, growing demand for monoclonal antibodies, vaccines, and therapies for rare diseases is expected to establish the country as an emerging hub for biologics innovation and outsourcing in North America.
Europe Biologics Contract Development And Manufacturing Organization Market Trends
Europe's biologics CDMO market benefits from a strong biopharma industry, government incentives, and investment in advanced manufacturing technologies. Countries such as Germany, France, and Switzerland are major biotech hubs with skilled workforces and robust regulatory expertise. The presence of established companies in the region is expected to meet the end-use demand for monoclonal antibodies, gene and cell therapies, and vaccines, further fueling the collaborations with CDMOs. Moreover, favorable conditions for clinical trials and GMP compliance further drive the region as Europe's Central market for biologics outsourcing and production.
The biologics contract development and manufacturing organization market in Germany held the highest share in 2025. Germany is one of the leading biologics CDMO hub, supported by advanced infrastructure, a skilled workforce, and strong biopharma R&D activities. Besides, increased demand for monoclonal antibodies, vaccines, and innovative therapies drives outsourcing to CDMOs, further supporting the market growth. Moreover, regulatory expertise and GMP-compliant facilities attract global clients, reinforcing Germany's critical position in European clinical and commercial biologics manufacturing.
The UK biologics contract development and manufacturing organization market is expected to grow significantly over the forecast period.The country's growth is fueled by the presence of established biotech clusters, regulatory support, and advanced manufacturing capabilities. Besides, significant demand for monoclonal antibodies, cell and gene therapies, and vaccines propels outsourcing to CDMOs. Moreover, with strong R&D investments, a skilled workforce, and favorable government initiatives, the UK remains an influential hub for biologics development and production in Europe.
Asia Pacific Biologics Contract Development And Manufacturing Organization Market Trends
Asia Pacific is expected to grow at a significant CAGR over the forecast period. The market is driven by increasing biopharma investment, growing healthcare demands, and cost-effective manufacturing solutions. Countries such as China, India, and Japan lead the regional market, offering skilled talent and advanced facilities with regulatory alignment. Rising demand for monoclonal antibodies, vaccines, and biosimilars drives outsourcing, allowing CDMOs to cater efficiently to global clients while boosting local market growth.
The biologics contract development and manufacturing organization market in China iswitnessing new growth opportunities due to leveraging government support and expanding biotech R&D alongside cost-effective manufacturing. Besides, the increasing demand for monoclonal antibodies, vaccines, and gene therapies further supports the market. In addition, the presence of advanced facilities and a skilled workforce for both domestic and international companies is expected to drive the clinical and commercial biologics production, enhancing China's position in the global biopharmaceutical market. For instance, in January 2026, WuXi Biologics secured UK MHRA GMP certification in China for commercial manufacturing of an ophthalmic biologic, reinforcing global-quality compliance and end-to-end drug substance and product capabilities.
The biologics contract development and manufacturing organization market in Japan is driven by characterized by a strong pharmaceutical infrastructure, a skilled workforce, and regulatory support. The country's high demand for monoclonal antibodies, vaccines, and cell therapies, as well as outsourcing, supports market growth. Moreover, Japan's Advanced manufacturing capabilities and GMP-compliant facilities, combined with a focus on innovative biologics, further support the market growth.
The India biologics contract development and manufacturing organization marketis experiencingrapid expansion, attributed to low labor cost, cost-effective hub for biologics CDMO offering skilled talent, advanced manufacturing capabilities, and regulatory alignment. The growing demand for monoclonal antibodies, biosimilars, and vaccines propels outsourcing activities. In addition, local and global biopharma companies are leveraging India's scalable facilities to accelerate development and reduce costs, strengthening the country's footprint in the Asia Pacific biologics market.
Latin America Biologics Contract Development And Manufacturing Organization Market Trends
The biologics contract development and manufacturing organization (CDMO) market in the Latin America region isexpected to significantly grow over the estimated time period. The market is driven by increasing healthcare demands, government support, and investments in manufacturing infrastructure. By outsourcing CDMOs, companies can access cost-effective production, regulatory expertise, and scalable facilities, bolstering regional and international biologics supply chains.
Brazil biologics contract development and manufacturing organization (CDMO) market is driven by supportive government initiatives and increasing biopharma R&D investments. The rising demand for monoclonal antibodies, vaccines, and biosimilars is fueling the market. Besides, CDMOs in the country provide scalable, cost-effective, and regulatory-compliant manufacturing solutions, further supporting companies to drive the development and expand regional access, cementing Brazil's status as a key biologics production hub.
Middle East & Africa Biologics Contract Development And Manufacturing Organization Market Trends
The biologics contract development and manufacturing organization (CDMO) market in the MEA region is expected to experience steady growth over the forecast period. The market is driven by increasing healthcare investments, government initiatives, and a growing demand for vaccines and biologics. Besides, outsourcing to CDMOs offers access to expertise, scalable manufacturing, and regulatory guidance. Moreover, the growing expansion of the market is enhancing the supply chain resilience, enabling companies to serve local populations while strengthening their biologics manufacturing capabilities effectively.
UAE biologics contract development and manufacturing organization (CDMO) market is experiencing growth driven by government support, investments in advanced manufacturing, and its strategic location. There is significant demand for vaccines, monoclonal antibodies, and innovative therapies, which fuels outsourcing. CDMOs in the UAE provide scalable, compliant, and cost-effective production, allowing both global and regional biopharma companies to enhance their biologics development and market presence within the Middle East.
Key Biologics Contract Development And Manufacturing Organization Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in December 2025,Samsung Biologics expanded its CDMO footprint in the U.S. by acquiring a Maryland-based drug manufacturing facility from GSK to strengthen long-term biologics production capacity.
Key Biologics Contract Development And Manufacturing Organization Companies:
The following key companies have been profiled for this study on the biologics contract development and manufacturing organization market.
- Lonza Group
- Catalent Pharma Solutions
- Samsung Biologics
- WuXi Biologics / WuXi AppTec
- Thermo Fisher Scientific
- Fujifilm Diosynth Biotechnologies
- Boehringer Ingelheim (Biopharma CDMO)
- Rentschler Biopharma SE
- AGC Biologics
- Charles River Laboratories
- Siegfried Holding
- Sandoz
- GenScript Biologics
- Vetter Pharma
- IDT Biologika
Recent Developments
-
In January 2026, Charles River Laboratories acquired PathoQuest in France and K.F. in Cambodia, enhancing NGS-based biologics testing and securing nonhuman primate supply to strengthen drug development and QC capabilities.
-
In October 2025, Lonza launched TheraPEAK AmpliCell Cytokines and 293-GT Medium to support GMP-compliant immune cell expansion and AAV production, enhancing its TheraPEAK portfolio used in FDA-approved therapies and 130+ clinical trials.
-
In September 2025,Rezon Bio launched as a European CDMO specializing in biologics. While as a new brand, it leverages an established track record of supporting biologics development from gene-level research to global commercialization through partnerships with experienced industry collaborators.
-
In July 2025, Samsung Biologics partnered with 35Pharma, enabling development and clinical approvals of two recombinant proteins through coordinated CDMO support, operational excellence, and trusted collaboration, accelerating therapeutic innovation from concept to clinic.
-
In May 2025, Radyus Research and Eurofins CDMO Alphora launched a strategic partnership, integrating development, regulatory, and GMP manufacturing capabilities to streamline global biotech drug programs from preclinical through clinical proof-of-concept with end-to-end solutions.
Biologics Contract Development And Manufacturing Organization Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 51.50 billion
Revenue forecast in 2033
USD 84.91 billion
Growth rate
CAGR of 7.4% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD Million/Billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, service, source, workflow, therapeutic area, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Lonza Group; Catalent Pharma Solutions; Samsung Biologics; WuXi Biologics / WuXi AppTec; Thermo Fisher Scientific; Fujifilm Diosynth Biotechnologies; Boehringer Ingelheim (Biopharma CDMO); Rentschler; Biopharma SE; AGC Biologics; Charles River Laboratories; Siegfried Holding; Sandoz; GenScript Biologics; Vetter Pharma; IDT Biologika
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Biologics Contract Development And Manufacturing Organization Market Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global biologics contract development and manufacturing organization market report based on product, service, source, workflow, therapeutic area, end use, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Monoclonal antibodies (mAbs)
-
Recombinant proteins & enzymes
-
Vaccines
-
Cell & Gene Therapies
-
Nucleic acid Therapeutics
-
Others
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Cell Line Development
-
Process Development
-
Upstream
-
Downstream
-
-
Analytical Testing & Method Validation
-
Scale-Up & Tech Transfer
-
-
Contract Manufacturing
-
API Manufacturing
-
Finished Drug Products Manufacturing
-
-
Packaging and Labelling
-
Regulatory Affairs
-
Logistics & Storage
-
Others
-
-
Source Outlook (Revenue, USD Million, 2021 - 2033)
-
Mammalian
-
Microbial
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Commercial
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Autoimmune Diseases
-
Infectious Diseases
-
Cardiovascular Diseases
-
Metabolic Diseases
-
Neurology
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotechnology Companies
-
Academic and Research Institutions
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global biologics contract development and manufacturing organization market is expected to grow at a compound annual growth rate of 7.40% from 2026 to 2033 to reach USD 84.91 billion in 2033.
b. The monoclonal antibodies (mAbs)segment dominated the biologics CDMO market with a share of 41.79% in 2025. The market growth is attributed to superior physicochemical properties, including high expression yields in mammalian cell cultures, excellent solubility, and robust stability during processing, which facilitate efficient scale-up and cost-effective production. These factors are expected to make mAbs advantageous for outsourced development and manufacturing models, driving growth for the biologics CDMO market.
b. The global biologics contract development and manufacturing organization market size was estimated at USD 47.84 billion in 2025 and is expected to reach USD 51.50 billion in 2026.
b. Some key players operating in the biologics CDMO market include Lonza Group, Catalent Pharma Solutions, Samsung Biologics, WuXi Biologics / WuXi AppTec, Thermo Fisher Scientific, Fujifilm Diosynth Biotechnologies, Boehringer Ingelheim (Biopharma CDMO), Rentschler Biopharma SE, AGC Biologics, Charles River Laboratories, Siegfried Holding, Sandoz, GenScript Biologics, Vetter Pharma, and IDT Biologika, among others.
b. The market growth is driven by rising prevalence of chronic and rare diseases, an aging global population, and the growing demand for highly targeted therapies. Besides, increasing biosimilar adoption provides cost-effective alternatives and expands patient access, further supporting market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










