- Home
- »
- Medical Devices
- »
-
Canadian Human Cadaveric Tissue Market Size Report, 2033GVR Report cover
![Canadian Human Cadaveric Tissue Market Size, Share & Trends Report]()
Canadian Human Cadaveric Tissue Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Soft Tissue Allografts, Musculoskeletal Allografts, Bone Allografts, Cardiovascular), By Indication, By End Use, By Source Of Supply, And Segment Forecasts
- Report ID: GVR-4-68040-855-6
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Canadian Human Cadaveric Tissue Market Summary
The Canadian human cadaveric tissue market size was valued at USD 129.50 million in 2025 and is anticipated to reach USD 212.74 million by 2033, expanding at a CAGR of 6.55% from 2026 to 2033. This growth is attributed to the rising number of orthopedic, spinal, and dental procedures requiring biological graft materials.
Key Market Trends & Insights
- By product, the bone allografts segment led the market with the largest revenue share in 2025.
- By indication, the orthopedic surgery segment led the market with the largest revenue share in 2025.
- By end use, the in-hospital procedures segment led the market with the largest revenue share in 2025.
- By source of supply, the imported cadaveric tissue segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 129.50 Million
- 2033 Projected Market Size: USD 212.74 Miillion
- CAGR (2026-2033): 6.55%
An aging population and the increasing prevalence of musculoskeletal disorders are further boosting demand for bone and soft-tissue reconstruction. Growing preference for biologic reconstruction over synthetic alternatives also supports market expansion. In addition, advancements in tissue processing and preservation technologies are enhancing graft safety and clinical outcomes, strengthening adoption across surgical applications.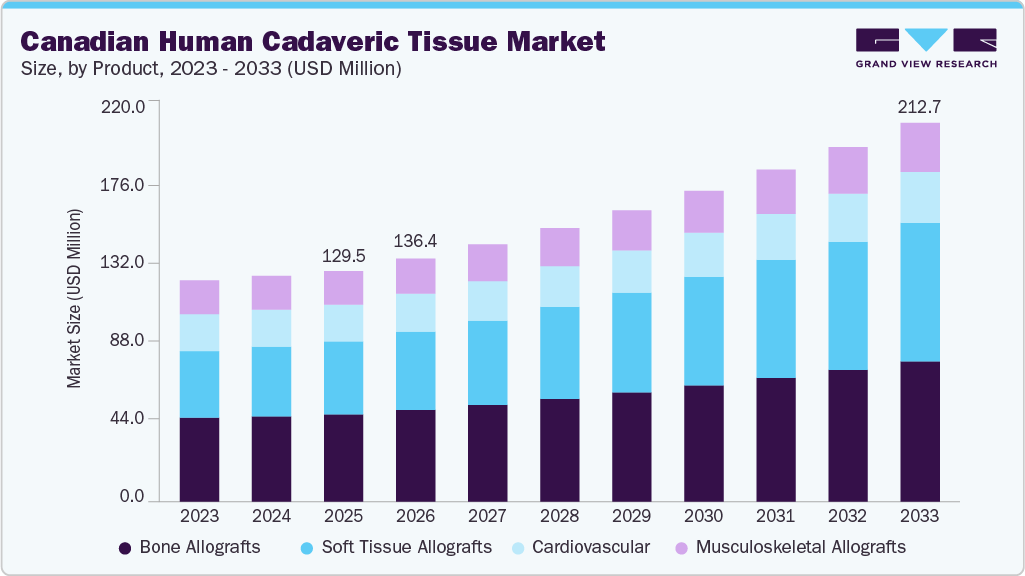
The increasing number of surgical procedures in Canada is a major driver of the human cadaveric tissue market. Rising volumes of orthopedic, spinal, dental, and reconstructive surgeries are driving sustained demand for bone and soft-tissue grafts. Allografts are widely used to restore structural support, promote healing, and reduce donor-site complications associated with autografts. As surgical capacity expands and waits times are addressed, utilization of cadaveric tissue products continues to grow.
Surgeries performed in Canada (April 1 - June 30, 2025)
Metric
Statistics
Surgeries Performed or Offered Within 3 Months
78%
Surgeries Performed or Offered Within 6 Months
88.50%
Surgeries Performed or Offered Within 12 Months
96.70%
Total Surgeries Performed
24,627
Source: Saskatchewan
Similarly, the growing number of heart valve procedures in Canada is driving demand for biologic graft materials, including cadaveric tissue used in valve repair and complex cardiac reconstruction. With an aging population and increasing prevalence of valvular heart disease, surgical volumes are significantly rising. This trend is strengthening the need for dependable, high-quality allografts across cardiac care settings in the Canadian market.
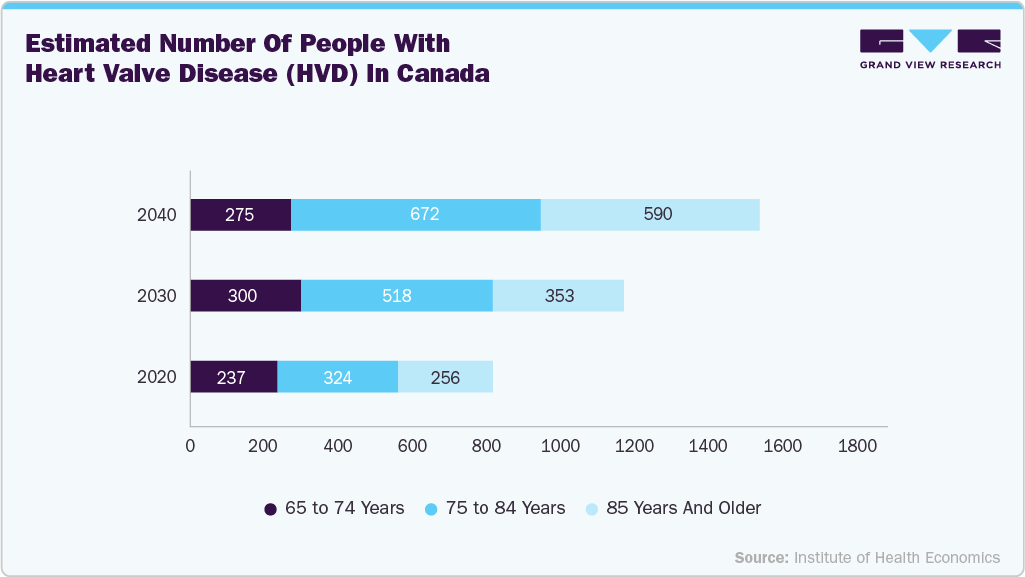
Prevalence of HVD in Canada
Age
% with HVD
65 to 74
6%
75 to 84
16%
85 +
30%
Source: Institute of Health Economics
Furthermore, the rising number of hip and knee replacement procedures in Canada is increasing demand for bone allografts, particularly for revision surgeries, bone defect management, and implant stabilization. As joint replacement volumes grow due to an aging population and higher prevalence of osteoarthritis, reliance on cadaveric tissue grafts continues to expand. This trend is contributing to sustained growth in the Canadian allograft market.
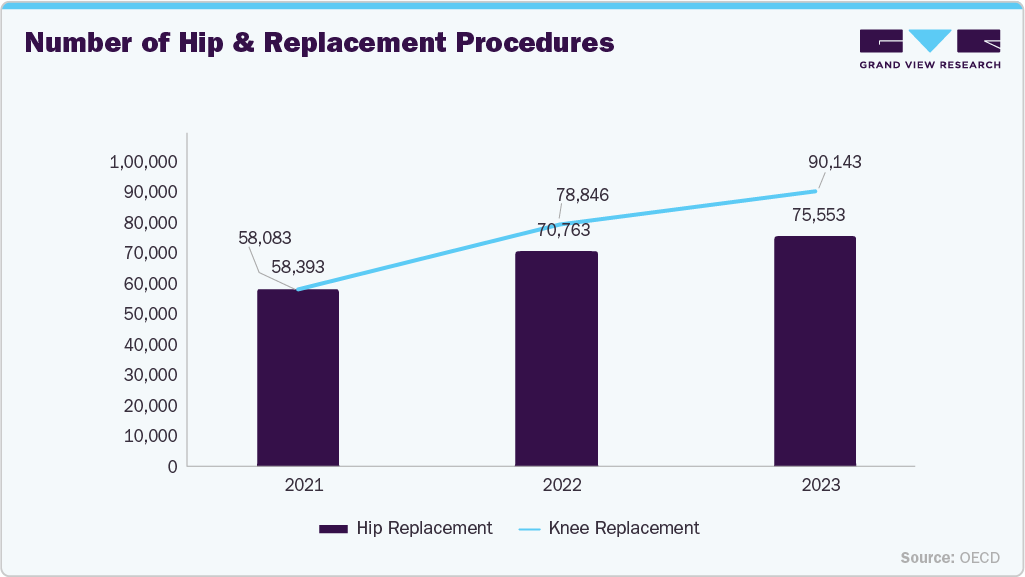
As a result, the increasing volume of surgical procedures including trauma repairs, heart valve surgeries, hip and knee replacements, spinal interventions, dental implant and bone grafting procedures, burn treatments, and plastic and reconstructive surgeries, continues to substantially drive demand in the Canadian human cadaveric tissue market.
The growing preference for biologic reconstruction, supported by continuous advancements in allograft innovation, is a major driver of the Canadian human cadaveric tissue market. Surgeons favor biologic graft materials over synthetic alternatives due to their enhanced biocompatibility, natural tissue integration, and ability to promote bone regeneration and soft-tissue healing. Allografts also offer reliable structural support while avoiding donor-site complications associated with autografts. These advantages make them highly suitable for orthopedic, dental, spinal, and reconstructive applications.
For instance, in January 2026, MTF Biologics, a global nonprofit focused on advancing tissue and organ donation, transplantation, and research, announced the launch of its 2026 Allograft Research Grant Program. The initiative features the established Innovation in Allografts Translational Research Grant and introduces a new Dermal Allograft Innovation Grant designed to advance next-generation technologies in dermal and skin allograft applications.
“Our goal is to support research that bridges laboratory insight with clinical need. The continued Allograft Translational Research Grants-together with the introduction of the new Dermal Allograft Innovation Grant-will accelerate discoveries that meaningfully advance patient care,” said Marc Long, Ph.D., Chief Scientific Officer and Executive Vice President, Research & Development, MTF Biologics.
“Launching the Dermal Allograft Innovation Grant reflects our commitment to advancing the science that saves and heals lives. This new mechanism empowers researchers with the resources needed to shape the next generation of dermal and skin-based allograft technologies,” said Joe Yaccarino, President and Chief Executive Officer, MTF Biologics.
Furthermore, in 2025, MTF Biologics allocated USD 1,000,000 to seven researchers through its Innovation in Allograft Translational Research Grants Program, funding studies in orthopedics, plastic and reconstructive surgery, and regenerative medicine.
“We are proud to offer a suite of funding opportunities that support investigators at every career stage. From early career scientists to established leaders, our programs enable bold ideas to take shape and make an impact,” said Jeffrey Cartmell, Ph.D., Senior Director, Intellectual Property & Grants, MTF Biologics.
As a result, the increasing preference for biologic reconstruction, supported by ongoing advancements and initiatives such as translational research grants for allograft innovation, is driving greater adoption of cadaveric tissue allografts across various surgical specialties in Canada. This sustained emphasis on innovation and regenerative technologies is expected to support long-term growth in the Canadian human cadaveric tissue market.
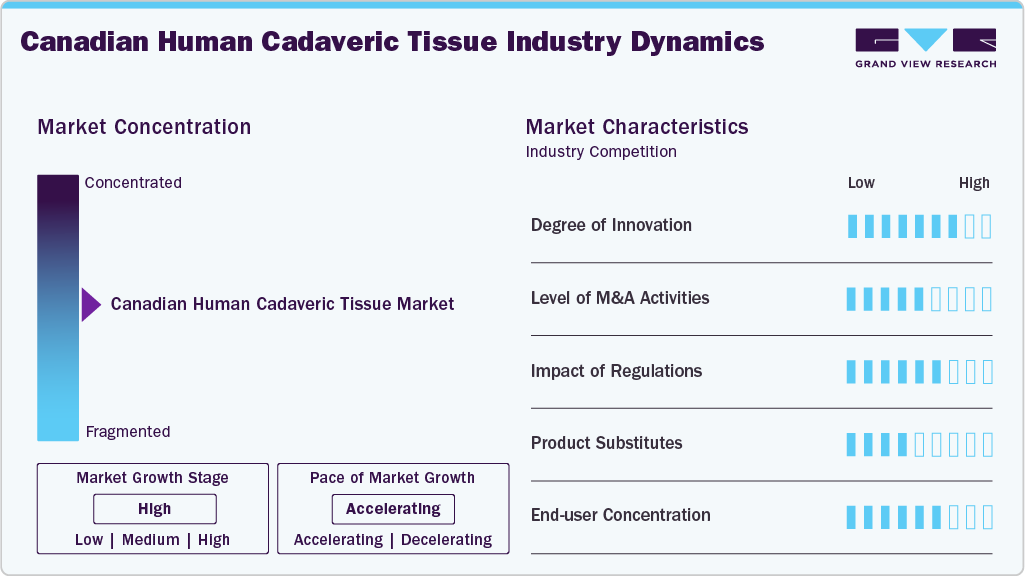
Market Concentration & Characteristics
The Canadian human cadaveric tissue market shows a moderate to high level of innovation, driven by advances in tissue processing, sterilization, and preservation technologies. Improved cryopreservation and graft preparation techniques are enhancing safety, viability, and clinical outcomes. Ongoing research and product development continue to expand applications in orthopedic, spinal, and reconstructive procedures. For instance, in February 2025, LifeNet Health announced the launch of OraGen, the first viable dental bone allograft composed of cryopreserved corticocancellous bone containing endogenous lineage-committed bone cells combined with demineralized bone matrix. This innovative composition provides three key properties for bone regeneration: osteogenicity through bone-forming cells, osteoconductivity by offering a supportive scaffold for cell growth, and osteoinduction by retaining natural growth factors that stimulate bone repair.
"We are thrilled to collaborate with Salvin Dental Specialties to introduce OraGen to the market," said Daniel Osborne, Chief Growth Officer at LifeNet Health. "This partnership reflects our commitment to delivering cutting-edge solutions that not only drive better patient outcomes but also advance the field of regenerative medicine. Together, we’re not just facilitating healing; we’re improving lives.”
“Salvin Dental’s exclusive distribution agreement with LifeNet Health for OraGen® demonstrates our unwavering commitment to providing the most comprehensive and innovative regenerative solutions to our customers,” said Ken Serota, CEO of Young Innovations. “Salvin’s enduring partnership with LifeNet Health has consistently focused on enhancing patient outcomes through a dedication to quality and service. Together, we look forward to empowering dental surgeons to improve their patients' quality of life."
The Canadian human cadaveric tissue market is experiencing a moderate level of mergers and acquisitions, primarily driven by global tissue banks seeking to expand their geographic reach and product portfolios. Strategic acquisitions often focus on specialized processing technologies, distribution networks, or niche graft segments. Such consolidation strengthens supply chain capabilities and enhances access to Canadian hospital contracts. For instance, in December 2025, LifeNet Health, a global provider of regenerative medicine, announced the acquisition of Tissue Testing Technologies LLC (T3), a biotechnology company specializing in biopreservation. T3’s proprietary technologies and services support the preservation of living cells and tissues for clinical and research applications, helping maintain the quality and effectiveness of life-saving therapies. The acquisition strengthens LifeNet Health’s capabilities to enhance patient outcomes globally and expand access to critical organs and regenerative treatments.
“Saving lives through advancing medical innovation is at the core of everything we do,” said Rony Thomas, President and CEO of LifeNet Health. “Integrating T3’s technologies allow us to expand the global reach of our lifesaving and healing products, accelerate research pipelines for therapies that rely on preserved tissues, and create end-to-end solutions for clinicians and researchers.”
“These technologies have the potential to transform organ and tissue preservation, improving transplant success and accelerating research breakthroughs,” said Dr. Kelvin G.M. Brockbank, Chief Executive Officer of Tissue Testing Technologies LLC. “Joining LifeNet Health allows us to scale our impact, reaching more patients and researchers worldwide.”
The Canadian human cadaveric tissue market is regulated by Health Canada under the Safety of Human Cells, Tissues and Organs for Transplantation (CTO) Regulations within the Food and Drugs Act. These regulations set mandatory safety standards covering donor screening, processing, testing, storage, labeling, importation, distribution, and clinical use of human tissues. For instance, in March 2025, the Government of Canada issued updated guidance on the CTO Regulations, offering further clarification and implementation direction to strengthen federal transplant safety requirements.
Product substitutes in the Canadian human cadaveric tissue market include autografts, synthetic bone graft substitutes, xenografts, and bioengineered or regenerative biomaterials. Autografts, sourced from the patient’s own body, remain a common alternative due to their high compatibility and osteogenic potential. Synthetic materials, such as calcium phosphate and bioceramics, are also used to overcome donor-related limitations. In addition, advancements in tissue-engineered and growth factor-enhanced products are expanding non-allograft treatment options in orthopedic and reconstructive procedures.
Product Insights
Bone allografts dominated the Canadian human cadaveric tissue market due to their extensive use in orthopedic, spinal, dental, and trauma-related procedures. High volumes of joint replacements, fracture repairs, and spinal fusion surgeries significantly contribute to demand for structural and cancellous bone grafts. Their ability to provide mechanical support while promoting natural bone regeneration strengthens clinical preference. As surgical volumes continue to rise, bone allografts maintain a leading share within the market.
Soft tissue allografts are expected to grow at the fastest rate during the forecast period in the Canadian Human Cadaveric Tissue Market. Rising sports injuries, ligament reconstructions, and tendon repair procedures are increasing the demand for grafts such as ACL, meniscus, and dermal matrices. Growing preference for minimally invasive orthopedic surgeries further supports adoption. Furthermore, advancements in processing technologies are enhancing graft strength and clinical outcomes, driving segment expansion.
Indication Insights
Orthopedic surgeries dominated the Canadian human cadaveric tissue market in 2025 due to the high volume of joint replacements, fracture repairs, and spinal fusion procedures performed nationwide. Bone and soft tissue allografts are widely used to restore structural integrity and support natural healing in these interventions. The growing aging population and rising incidence of musculoskeletal disorders further strengthen demand. As a result, orthopedic applications account for the largest share of allograft utilization in Canada.
The dental segment is expected to grow at the fastest rate during the forecast period in the Canadian human cadaveric tissue market. The increasing demand for dental implants, bone grafting procedures, and periodontal regeneration is driving greater adoption of bone allografts in oral surgeries. Growing awareness of aesthetic dentistry and improved access to advanced dental care further support segment expansion. In addition, the rising aging population requiring restorative dental procedures is contributing to sustained growth.
End Use Insights
In-hospital procedures dominated the Canadian human cadaveric tissue market in 2025, driven by the high concentration of complex orthopedic, spinal, cardiovascular, and reconstructive surgeries performed in hospital settings. Hospitals serve as primary centers for tissue transplantation, supported by specialized surgical teams and advanced infrastructure. Strict regulatory compliance and centralized procurement systems further reinforce hospital-based utilization. As a result, most allograft procedures were conducted within inpatient facilities.
Out-of-hospital settings are expected to grow at the fastest rate during the forecast period in the Canadian human cadaveric tissue market. The increasing shift toward ambulatory surgical centers, specialty clinics, and private facilities is expanding the use of allografts outside traditional hospital environments. Minimally invasive procedures and shorter recovery times are supporting this transition. Furthermore, efforts to reduce healthcare costs and improve patient convenience are accelerating growth in these settings.
Source of Supply Insights
The imported cadaveric tissue segment dominated in 2025 and is expected to grow at the fastest rate during the forecast period. The dominance of this segment is primarily due to Canada’s continued reliance on established international tissue banks, particularly those in the U.S., which offer a wide portfolio of high-quality, safety-screened, and readily available allograft products. Limited domestic donor availability, stringent regulatory requirements, and increasing demand for orthopedic, dental, spinal, and reconstructive procedures further support import dependence. In addition, strong distribution networks, advanced processing technologies, and consistent supply from global suppliers are expected to accelerate segment growth over the coming years.
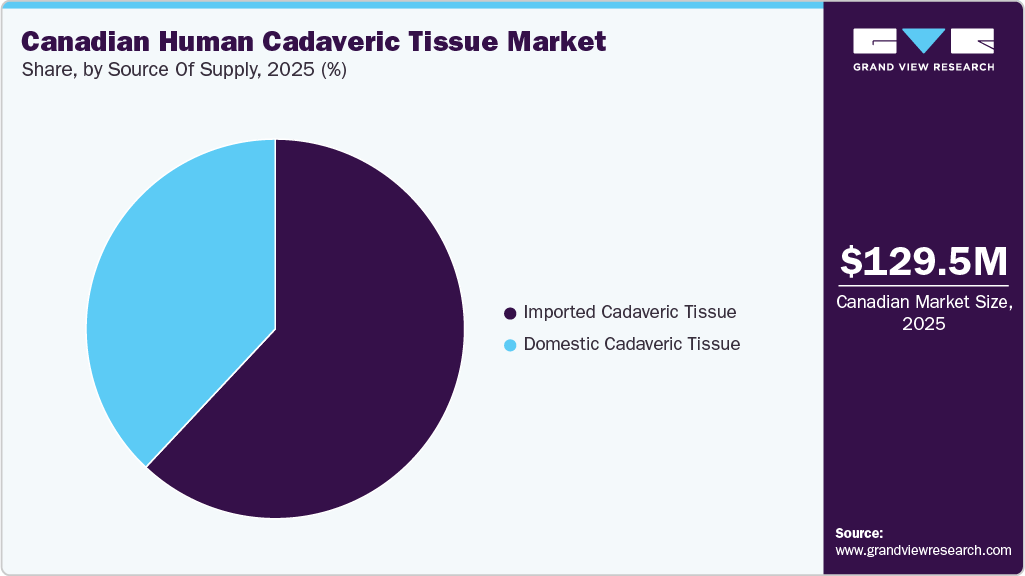
The domestic cadaveric tissue segment is witnessing significant growth in the Canadian human cadaveric tissue market. This growth is driven by increasing government initiatives to strengthen local tissue donation programs and expand tissue bank infrastructure. Rising awareness about organ and tissue donation, along with efforts to reduce dependency on imports, is further supporting domestic supply. In addition, improvements in processing standards and regulatory compliance are enhancing the reliability and acceptance of locally sourced allografts across healthcare institutions.
Key Canadian Human Cadaveric Tissue Company Insights
The Canadian human cadaveric tissue market is led by major players such as MTF Biologics, LifeNet Health, and AlloSource, as well as national organizations such as Canadian Blood Services. Market share is concentrated among these large tissue banks due to established hospital networks and supply agreements, while smaller regional providers maintain niche participation. Overall, the market remains moderately consolidated.
Key Canadian Human Cadaveric Tissue Companies:
- MTF Biologics
- LifeNet Health
- AlloSource
- Artivion (formerly CryoLife)
- LeMaitre
- AlloWide Health Inc.
- BioHorizons (Henry Schein, Inc.)
- Zimmer Biomet
- Medtronic
- Stryker
Recent Developments
-
In January 2026, MTF Biologics, a global nonprofit organization dedicated to saving and healing lives through tissue and organ donation, transplantation, and research, announced the opening of proposals for its 2026 Allograft Research Grant Program. The program includes the long-standing Innovation in Allografts Translational Research Grant and introduces a new Dermal Allograft Innovation Grant to accelerate next-generation technologies in allograft dermis and skin.
“Our goal is to support research that bridges laboratory insight with clinical need,” said Marc Long, Ph.D., Chief Scientific Officer and Executive Vice President, Research & Development, MTF Biologics. “The continued Allograft Translational Research Grants-together with the introduction of the new Dermal Allograft Innovation Grant-will accelerate discoveries that meaningfully advance patient care.”
“Launching the Dermal Allograft Innovation Grant reflects our commitment to advancing the science that saves and heals lives,” said Joe Yaccarino, President and Chief Executive Officer, MTF Biologics. “This new mechanism empowers researchers with the resources needed to shape the next generation of dermal and skin-based allograft technologies.”
-
In December 2025, LifeNet Health, a global provider of regenerative medicine, announced the acquisition of Tissue Testing Technologies LLC (T3), a biotechnology company specializing in biopreservation. T3’s proprietary technologies and services are designed to preserve living cells and tissues for clinical and research applications, supporting the quality and effectiveness of life-saving therapies.
“Saving lives through advancing medical innovation is at the core of everything we do,” said Rony Thomas, President and CEO of LifeNet Health. “Integrating T3’s technologies allow us to expand the global reach of our lifesaving and healing products, accelerate research pipelines for therapies that rely on preserved tissues, and create end-to-end solutions for clinicians and researchers.”
“These technologies have the potential to transform organ and tissue preservation, improving transplant success and accelerating research breakthroughs,” said Dr. Kelvin G.M. Brockbank, Chief Executive Officer of Tissue Testing Technologies LLC. “Joining LifeNet Health allows us to scale our impact, reaching more patients and researchers worldwide.”
-
In October 2025, AlloSource, a life sciences organization dedicated to restoring patient functionality through advanced medical products derived from human tissue donation, announced that research highlighting its proprietary ViaTrue Cryopreservation Process was presented at the 18th International Cartilage Regeneration & Joint Preservation Society (ICRS) World Congress. The findings demonstrated positive clinical and scientific outcomes for two of the company’s cartilage allografts.
“Cryopreserving allografts with ViaTrue helps to alleviate inventory management challenges and provides surgeons with more flexibility for their patients,” said Carolyn Rorick, AlloSource Senior Director Product Development, Innovation and Clinical Research. “Showing the positive patient outcomes from this ongoing evaluation is an important step in proving the impact cryopreserved cartilage can have on healing patients.”
“There is still research to do in the body to show that cryopreserved osteochondral allografts are as good as fresh allografts, but this work is the first step towards proving that cryopreserved grafts can be a viable option for the millions of patients that need them,” said Dr. Laurie Goodrich, Director of the Orthopaedic Research Center at Colorado State University’s C. Wayne McIlwraith Translational Medicine Institute.
-
In March 2025, AlloSource, a life sciences organization focused on restoring patient functionality through enhanced medical products derived from human tissue donation, announced the 200th implant of its AceConnex Pre-Sutured Fascia. The device is the only pre-sutured fascia allograft cleared under the 510(k) pathway for use in hip labral reconstruction and augmentation.
“Having used AceConnex Pre-Sutured Fascia, I believe it answers an unmet need to better support surgeons performing hip labral reconstruction and augmentation,” said Dr. Andrew Jimenez, a sports and hip preservation surgeon at Yale University. “I appreciate the quality and consistency of AceConnex, which helps me take better care of my patients.”
“We created AceConnex Pre-Sutured Fascia to help streamline hip arthroscopy,” said Dean Elliott, AlloSource President and CEO. “This 200-implant milestone is one step in our goal to provide innovative solutions that help surgeons more easily perform complex hip arthroscopy procedures.”
Canadian Human Cadaveric Tissue Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 136.44 million
Revenue forecast in 2033
USD 212.74 million
Growth rate
CAGR of 6.55% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD Million and CAGR from 2026 to 2033
Segments covered
Product, indication, end use, supply of source
Regional scope
Canada
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
MTF Biologics; LifeNet Health; AlloSource; Artivion (formerly CryoLife); LeMaitre; AlloWide Health Inc.; BioHorizons (Henry Schein, Inc.); Zimmer Biomet; Medtronic; Stryker
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Canadian Human Cadaveric Tissue Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the Canadian human cadaveric tissue market report on the basis of product, indication, end use and source of supply:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Soft Tissue Allografts
-
Cadaveric skin
-
Acellular Dermal Matrix (ADM)
-
-
Musculoskeletal Allografts
-
Bone Allografts
-
Demineralized Bone Matrix (DBM)
-
Others
-
-
Cardiovascular
-
Heart Valves
-
Vascular Grafts
-
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Burn Care & Wound Reconstruction
-
Plastic & Reconstructive Surgery
-
Orthopedic Surgery (Spine, Trauma, Sports Medicine)
-
Dental
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
In-Hospital Procedures
-
Out-of-Hospital Settings
-
-
Source of Supply Outlook (Revenue, USD Million, 2021 - 2033)
-
Domestic Cadaveric Tissue
-
Imported Cadaveric Tissue
-
Frequently Asked Questions About This Report
b. The Canadian human cadaveric tissue market size was estimated at USD 129.50 million in 2025 and is expected to reach USD 136.44 million in 2026.
b. The Canadian human cadaveric tissue market is expected to grow at a compound annual growth rate of 6.55% from 2026 to 2033 to reach USD 212.74 million by 2033.
b. Bone allografts dominated the Canadian human cadaveric tissue market with a share of 37.84% in 2025. This is attributable to their extensive use in orthopedic, spinal, dental, and trauma-related procedures. High volumes of joint replacements, fracture repairs, and spinal fusion surgeries significantly contribute to demand for structural and cancellous bone grafts.
b. Some key players operating in the Canadian human cadaveric tissue market include MTF Biologics, LifeNet Health, AlloSource, Artivion (formerly CryoLife), LeMaitre, AlloWide Health Inc., BioHorizons (Henry Schein, Inc.), Zimmer Biomet, Medtronic, Stryker
b. Key factors that are driving the market growth include the rising number of orthopedic, spinal, and dental procedures requiring biological graft materials. An aging population and the increasing prevalence of musculoskeletal disorders are further boosting demand for bone and soft-tissue reconstruction.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










