- Home
- »
- Pharmaceuticals
- »
-
Cancer Vaccine Market Size, Share, Industry Report, 2033GVR Report cover
![Cancer Vaccine Market Size, Share & Trends Report]()
Cancer Vaccine Market (2026 - 2033) Size, Share & Trends Analysis Report By Vaccine Type (Preventive Cancer Vaccine, Therapeutic Cancer Vaccine), By Indication Type, By Technology Type, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-096-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
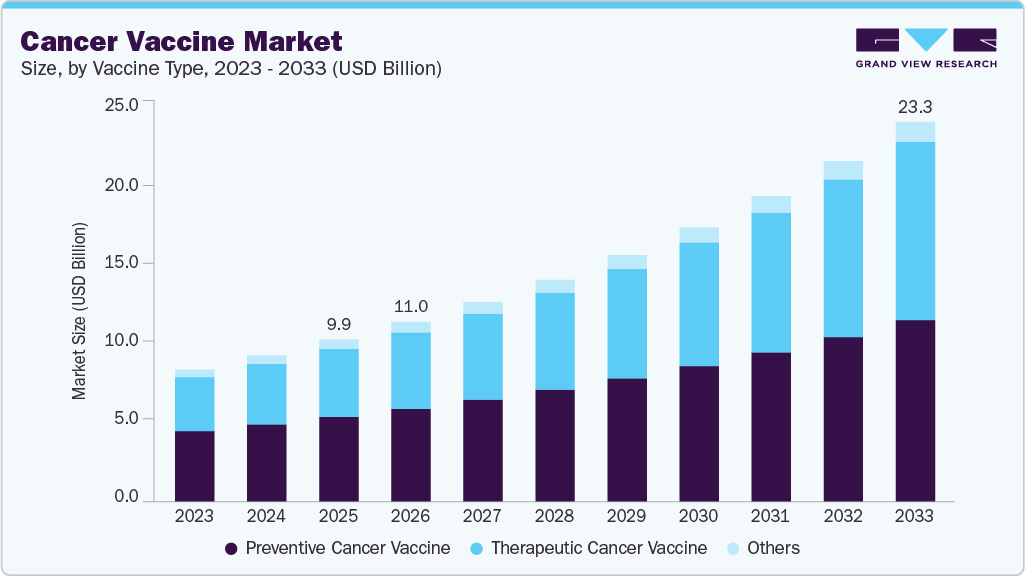
Cancer Vaccine Market Summary
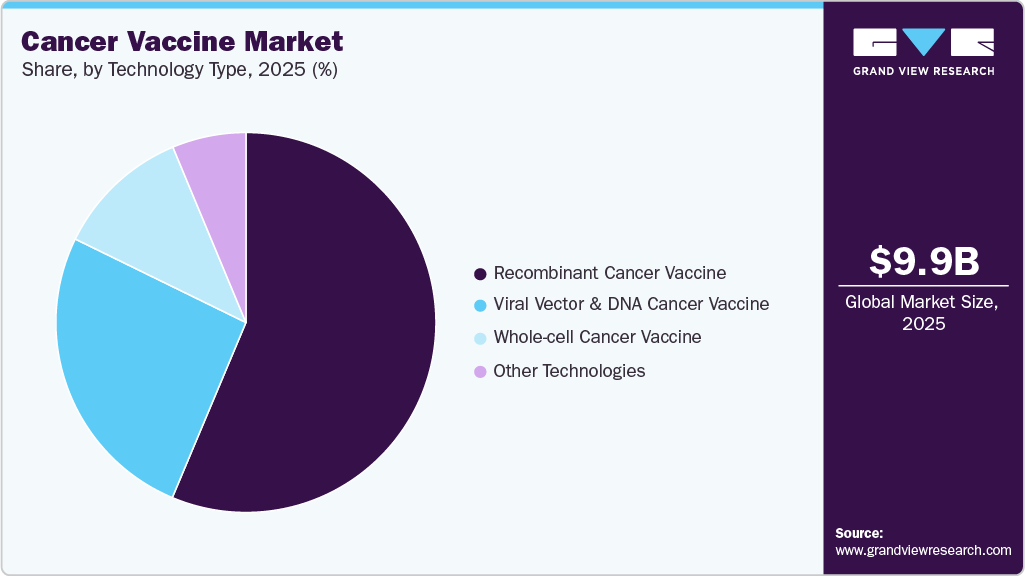
The global cancer vaccine market size was estimated at USD 9.93 billion in 2025 and is projected to reach USD 23.31 billion by 2033, growing at a CAGR of 11.30% from 2026 to 2033. This growth is primarily driven by the rising global burden of cancer, increasing adoption of immunotherapy-based treatments, and rapid advancements in vaccine technologies such as mRNA, dendritic cell platforms, and personalized neoantigen vaccines.
Key Market Trends & Insights
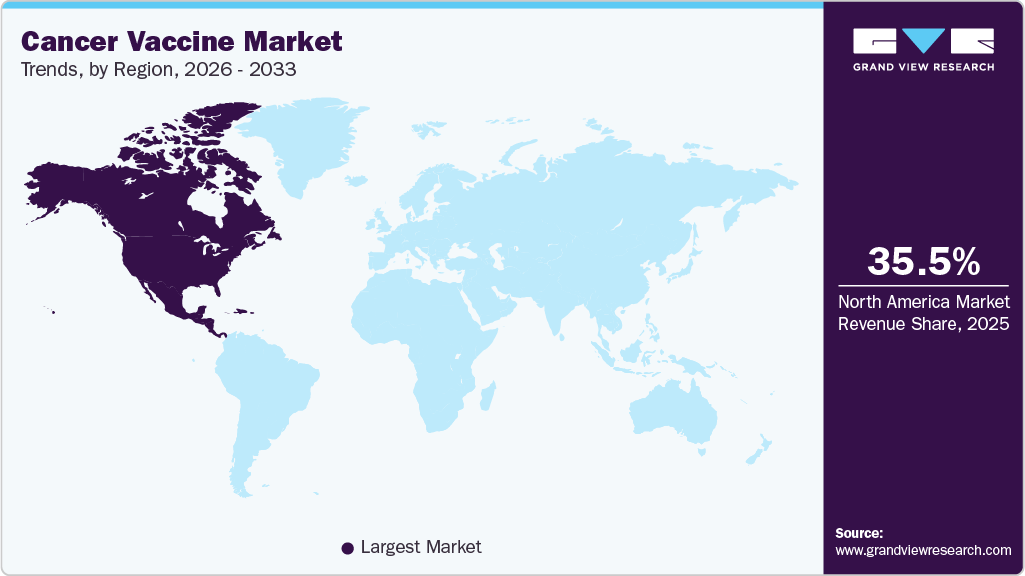
- North America dominated the global cancer vaccine market with the largest revenue share of 35.47% in 2025.
- The cancer vaccine industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By vaccine type, the preventive cancer vaccine segment led the market with the largest revenue share of 52.14% in 2025.
- By indication type, the cervical cancer segment accounted for the largest market revenue share in 2025.
- By technology type, the recombinant cancer vaccine segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 9.93 Billion
- 2033 Projected Market Size: USD 23.31 Billion
- CAGR (2026-2033): 11.30%
- North America: Largest market in 2025
The Cancer Vaccine industry is expanding rapidly because of the rising global burden of cancer and the increasing emphasis on immunotherapy as a viable long-term treatment option. Advances in vaccine technology, such as mRNA and DNA platforms, have significantly improved the ability to design vaccines that teach the immune system to recognize cancer-specific antigens and destroy tumor cells more effectively than traditional therapies.Interest in Cancer Vaccine has been bolstered by growing investment from pharmaceutical companies and academic research groups seeking more personalized treatments that can reduce side effects and improve patient outcomes, especially in solid tumors where options remain limited. For instance, in January 2026, Moderna and Merck reported that their personalized cancer vaccine used with Keytruda showed a sustained reduction in risk of recurrence or death in high-risk melanoma patients during long-term follow-up, reinforcing confidence in vaccine approaches and driving further clinical development and funding.
Another major growth factor in the Cancer Vaccine industry is the rapid pace of scientific innovation improving the precision and effectiveness of vaccine candidates. New techniques in antigen identification, neoantigen targeting, and computational biology have enabled vaccines to be tailored to the unique genetic profile of individual tumors, which is critical for triggering a robust immune response. The expanding pipeline of clinical trials demonstrates increasing confidence among researchers and investors that Cancer Vaccine can deliver meaningful benefits across multiple cancer types, including those historically difficult to treat.
For instance, in July 2025, Scientists at the University of Florida developed an experimental mRNA vaccine that triggered a strong anticancer immune response against tumors in preclinical models, highlighting both the promise of next-generation vaccines and the technological momentum supporting market growth.
Market growth is also propelled by supportive policy environments, increasing public awareness of cancer prevention and early detection, and the integration of novel diagnostic tools that work in tandem with vaccine strategies. Governments and health agencies have intensified funding for immunotherapy research, and collaborations between biotech firms and research institutions are becoming more common, which accelerates product development and regulatory approval pathways.
Moreover, the success of vaccine platforms during the COVID-19 pandemic has reinforced confidence in mRNA technology for other diseases, including cancer, leading to greater investment and quicker translation of research into clinical applications. For instance, in September 2025, Russia announced that its new cancer vaccine Enteromix, developed using advanced mRNA technology, was ready for clinical use and would be provided free to patients following regulatory approval, illustrating both the technological progress and policy support driving market expansion.
Market Concentration & Characteristics
The Cancer Vaccine industry is marked by high levels of innovation, driven by breakthroughs in mRNA platforms, neoantigen targeting and personalized immunotherapy that tailor vaccines to individual tumor profiles. Advanced technologies like next‑generation sequencing and AI‑assisted antigen discovery are accelerating the design and optimization of novel vaccine candidates. Established players such as BioNTech, Moderna and emerging biotech firms are pushing the frontier with combination approaches that integrate vaccines with checkpoint inhibitors and other immune modulators. This continuous innovation creates a rich pipeline of clinical assets targeting diverse cancer types from melanoma to head and neck cancers. Partnerships between academic institutions and biopharma firms further enhance research throughput and cross‑disciplinary innovation. Overall, innovation is a central characteristic shaping competitive advantage and long‑term growth in the Cancer Vaccine sector.
Barriers to entering the Cancer Vaccine industry remain significant, particularly for smaller or new companies facing steep research and development costs that can exceed hundreds of millions of dollars per candidate. The complexity of vaccine design, manufacturing, and individualized production processes demands specialized infrastructure and expertise. Regulatory approval pathways for oncology vaccines are rigorous, often requiring extensive preclinical and multi‑phase clinical trials that extend timelines and heighten financial risk. Incumbent firms with deep pockets, established clinical track records and broad intellectual property portfolios hold advantages that discourage new entrants. In addition, sophisticated cold-chain logistics and skilled personnel are prerequisites for manufacturing and distribution, further limiting participation to well-funded, experienced organizations.
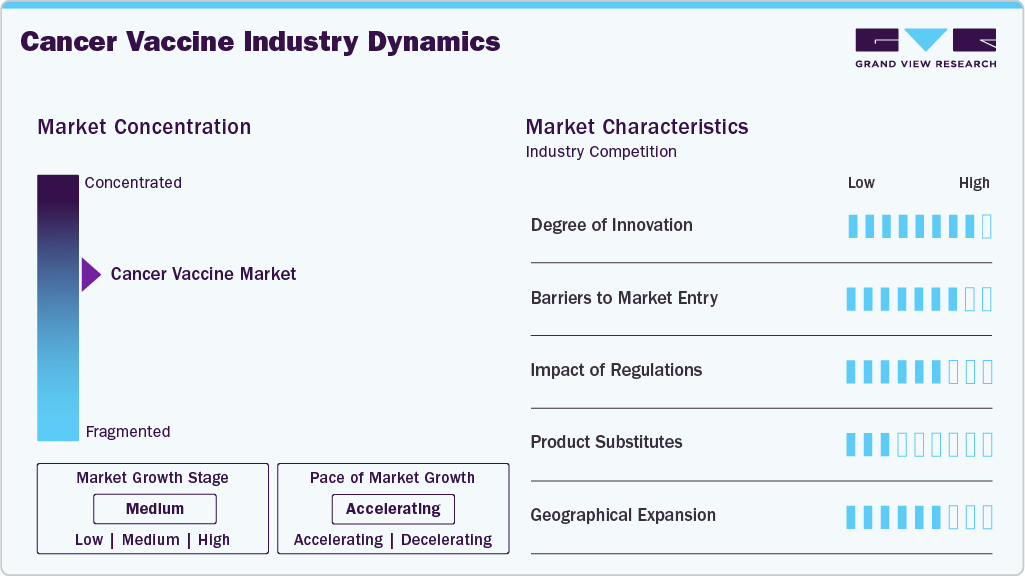
Regulatory frameworks exert a strong influence on the structure and pace of the Cancer Vaccine industry, as agencies such as the U.S. Food and Drug Administration and European regulators demand stringent evidence of safety and clinical efficacy before granting approval. These rigorous standards often translate into long development timelines and elevated compliance costs, particularly for first‑in‑class or personalized vaccine approaches that deviate from traditional drug modalities. Regulatory designations like breakthrough therapy or fast track can expedite review processes, yet variability in regional regulatory requirements adds complexity for global market strategies. Post‑approval monitoring and pharmacovigilance obligations also shape lifecycle management and labeling updates. Overall, regulation is a defining factor in shaping investment decisions, clinical strategy and market timing for cancer vaccine developers.
Product substitutes for Cancer Vaccine include established oncology treatments such as chemotherapy, targeted small molecule therapies, monoclonal antibodies and adoptive cell therapies like CAR‑T. While these alternatives do not work through vaccine‑induced immunization, they fulfill overlapping therapeutic needs, particularly in indications where vaccines remain investigational. Targeted therapies and checkpoint inhibitors may serve as partial substitutes in some treatment regimens, influencing physician choice based on disease stage and patient profile. The high differentiation of cancer vaccine mechanisms reduces direct substitution risk, yet conventional modalities retain dominance due to broader clinical adoption and longer commercial histories. This competitive landscape encourages combination strategies where vaccines are paired with other therapies to enhance outcomes and reduce therapeutic substitution.
Geographical expansion is a key growth driver for the Cancer Vaccine industry as companies seek to access new patient populations and diversify revenue streams beyond traditional strongholds in North America and Europe. North America currently holds a significant market share supported by robust healthcare infrastructure and R&D investment, while Asia‑Pacific is projected to grow rapidly as local biotechs, rising healthcare expenditure and national cancer programs expand access to advanced therapies. Expansion strategies include establishing regional manufacturing hubs, forging distribution partnerships and navigating local regulatory landscapes to secure approvals. Emerging markets such as China, India and Brazil present large unmet needs and growing prevalence of cancer, offering long‑term potential for market penetration. Localization of clinical research and manufacturing helps firms mitigate supply chain challenges and tailor offerings to regional healthcare systems.
Vaccine Type Insights
The preventive Cancer Vaccine segment led the market with the largest revenue share of 52.14% in 2025, driven by strong public health policies, increasing adoption of HPV immunization into national schedules, and growing evidence that broad vaccination reduces future cancer burdens, strengthening buy-in from governments and healthcare payers. Routine inclusion of HPV vaccines in national immunization programmes ensures predictable demand and stable revenue streams for manufacturers while supporting long-term healthcare cost savings as infection-driven cancers decline. For instance, September 2025, Associated Press news reported that Pakistan vaccinated around 9 million girls aged 9 to 14 against HPV as part of a nationwide campaign, overcoming hesitancy and demonstrating how large-scale preventive programmes can rapidly scale immunization to reduce cervical cancer risk in high-burden countries. These nationwide campaigns also generate public awareness that reinforces demand across regions, encourage multilateral funding and support for vaccine procurement, and help manufacturers secure long-term supply agreements.
The therapeutic Cancer Vaccine segment is projected to grow at the fastest CAGR of 12.86% over the forecast period, due to strong clinical progress and increasing adoption of vaccines that can stimulate targeted immune responses in patients with existing cancers, making them strategic assets in combination with other immunotherapies and standard treatments. These vaccines are designed to augment the body’s natural defenses to recognize and destroy tumor cells, which can improve survival outcomes and reduce recurrence when paired with checkpoint inhibitors or other modalities, encouraging pharmaceutical investment and expanded development pipelines. For instance, April 2024, Reuters news reported that Moderna’s personalized mRNA cancer vaccine candidate showed promising early results in a head and neck cancer trial when combined with Merck’s Keytruda, with immune responses linked to tumor regression, highlighting real-world progress in therapeutic vaccine applications. Such positive clinical signals support confidence among oncology developers, drive strategic collaborations, and help attract regulatory interest in advancing therapeutic vaccine candidates toward later-stage trials and commercialization
Indication Type Insights
The cervical cancer segment led the market with the largest revenue share of 30.74% in 2025, driven by expanded access initiatives, stronger policy support for HPV vaccination, and growing evidence that broad immunization significantly reduces disease burden and downstream treatment costs. National immunization programmes and global partnerships have increasingly prioritized cervical cancer prevention, resulting in higher vaccine uptake and more robust procurement agreements between governments and manufacturers. For instance, October 2024, World Health Organization news reported that the WHO had added a new HPV vaccine to its list of prequalified vaccines, increasing global supply and enabling more countries to introduce or scale up routine HPV immunization, which directly supports efforts to reduce cervical cancer incidence worldwide. Such enhancements in vaccine availability and international support have encouraged more health ministries to adopt HPV vaccination strategies, strengthening long-term demand and contributing to the cervical cancer segment’s leading market position.
The bladder cancer segment is projected to grow at the fastest CAGR of 11.76% over the forecast period. supported by advances in immunotherapeutic and vaccine-related strategies that address high recurrence rates and unmet clinical needs, especially in non-muscle invasive disease. Bladder cancer has historically relied on Bacillus Calmette-Guérin (BCG) vaccination as a standard immune-based therapy to reduce recurrence after surgery, and recent improvements in vaccine approaches and combination regimens are strengthening clinical confidence and investment. For instance, March 2025, eCancer.org news reported that a second tuberculosis (TB) vaccination given after surgery boosted immunity in bladder cancer patients and reduced cancer recurrence in a pilot study, demonstrating how augmenting existing vaccine-based immunotherapy can improve outcomes and fuel interest in evolving vaccine platforms for bladder cancer management. These developments underscore growing research momentum, regulatory interest, and commercial potential in therapies that enhance immune responses, driving the bladder cancer segment’s anticipated rapid market growth.
Technology Type Insights
The recombinant Cancer Vaccine segment led the market with the largest revenue share of 56.33% in 2025, driven by widespread clinical success, established manufacturing platforms, and strong integration of recombinant technologies into preventive and therapeutic vaccine design, appealing to public and private buyers alike. Recombinant vaccines can deliver precise antigens to the immune system while avoiding pathogen replication, thereby improving safety profiles and enabling large-scale production with consistent quality, making them attractive for inclusion in national immunization schedules and private healthcare formularies. For instance, in January 2026, Reuters news reported that a recombinant HPV vaccine exhibited strong immunogenicity in long-term follow-up studies while being incorporated into expanded immunization programmes in multiple countries, reinforcing confidence in recombinant platforms and driving broader adoption. This combination of proven efficacy, scalable manufacturing, and policy support continues to strengthen the commercial dominance of recombinant Cancer Vaccine in the market.
The viral vector and DNA Cancer Vaccine segment is projected to grow at the fastest CAGR of 12.42% over the forecast period. This growth is primarily driven by sustained technological progress that improved antigen delivery and immune activation while broadening the spectrum of targetable tumors. Viral vector platforms enabled efficient intracellular antigen expression and robust T-cell responses, whereas DNA vaccines offered molecular stability, scalable manufacturing, and the capacity to encode multiple antigens, making them attractive for complex and heterogeneous cancers. These platform advantages supported increased capital allocation, partnership formation, and pipeline expansion as developers advanced candidates beyond early clinical phases.
For instance, in October 2025, the Journal of Personalized Medicine news, researchers described personalized DNA Cancer Vaccine as a rapidly evolving therapeutic approach, reporting that more than 100 clinical trials involving neoantigen-targeted vaccines had been registered, underscoring rising scientific momentum. The review explained that vaccines were engineered through tumor sequencing, computational analysis, and customized production, and highlighted how next-generation sequencing and delivery technologies improved tumor-specific antigen identification and recurrence-free survival outcomes in selected indications, despite persistent challenges across tumor types, including head and neck cancers.
Regional Insights
North America dominated the global Cancer Vaccine market with the largest revenue share of 35.47% in 2025, due to an advanced healthcare system and strong research and development focus from major biopharmaceutical companies. High clinical trial activity and broad adoption of immunotherapy contribute to market leadership, while substantial healthcare infrastructure supports both preventive and therapeutic Cancer Vaccine. The region’s patient awareness and screening practices help accelerate vaccine uptake and clinical acceptance. Collaboration between industry and academic research centres drives innovation and rapid product development cycles. Rapid integration of cutting-edge vaccine technologies into clinical practice further reinforces regional dominance.
U.S. Cancer Vaccine Market Trends
The Cancer Vaccine market in the U.S. is the leading contributor within North America, driven by extensive oncology research and early adoption of novel vaccine platforms, including mRNA and personalized approaches. Robust private sector investment fuels clinical trial expansion and regulatory filings for both preventive and therapeutic Cancer Vaccine. Widespread healthcare access and high prevalence of target cancers create consistent demand for advanced immunotherapies. Large biotech and pharmaceutical companies headquartered in the U.S. also enhance market scale and competitiveness. Patient preference for innovative treatments supports faster uptake and commercial success. Strategic industry partnerships further strengthen the U.S. market position.
Europe Cancer Vaccine Market Trends
The Cancer Vaccine market in the Europe maintains a significant position in the cancer vaccine ecosystem supported by strong biomedical research networks and established pharmaceutical sectors. Countries across Western and Central Europe contribute substantial clinical expertise and manufacturing capability for cancer immunotherapies. High levels of clinical collaboration and cross-border trials help expand the regional pipeline of vaccine candidates. Public and private healthcare systems emphasize preventive care, aiding the adoption of vaccine solutions. The presence of notable biotech clusters in several European cities enhances innovation and cross-industry partnerships. Steady market growth reflects balanced demand across preventive and therapeutic segments.
The UK Cancer Vaccine market stands out in Europe due to concentrated oncology research and leading academic institutions that support clinical development of Cancer Vaccine. A sophisticated healthcare delivery system and high patient access to novel therapies drive demand and adoption trends. Strategic alliances between local biotech firms and multinational companies broaden the commercial landscape. Clinical expertise in immuno-oncology accelerates trial recruitment and data generation. The pharmaceutical ecosystem in the UK emphasizes rapid integration of emerging vaccine technologies. Continued investment in clinical infrastructure enhances regional competitiveness.
The Cancer Vaccine market in Germany contributes to the European region with strong industrial research capacity and participation in international clinical studies. The nation’s biotech and life sciences sectors provide robust support for both vaccine innovation and commercialization. High healthcare expenditure per capita enables wider access to new immunotherapy options for cancer patients. German research institutions often engage in early-stage discovery and translational studies that feed global pipelines. Established manufacturing capabilities ensure efficient production scalability for emerging vaccine products. This combination of scientific expertise and industrial strength sustains market growth.
The France Cancer Vaccine market plays a key role in Europe’s cancer vaccine segment through active involvement in oncology research and expanding clinical trial activity. Established pharmaceutical companies and research institutes work on advanced vaccine platforms aimed at improving patient outcomes. Strong healthcare infrastructure supports clinical adoption and distribution of Cancer Vaccine across patient populations. French biotech firms increasingly collaborate with global partners to bring innovative candidates to market. Rising public awareness of cancer prevention and treatment options further fuels demand. The country’s scientific base contributes to sustained regional significance.
Asia-Pacific Cancer Vaccine Market Trends
The Cancer Vaccine market in Asia Pacific is expected to register at a significant CAGR of 12.42% over the forecast period, backed by rising healthcare investment and growing awareness of preventive oncology. Large and aging populations in major countries expand the patient base for both therapeutic and prophylactic vaccines. Increasing numbers of clinical trials and strategic entry by global vaccine developers amplify regional activity. Expanding manufacturing capacity in China, India, and Japan supports broader access to advanced vaccine solutions. Healthcare modernization and improving diagnostic infrastructure drive uptake of Cancer Vaccine. Rapid economic growth and rising middle-class demand further accelerate market expansion.
The Japan Cancer Vaccine market advances with strong biotechnology research and high healthcare expenditure that enable rapid adoption of novel immunotherapies. The country’s clinical research output in cancer treatment contributes valuable data for global vaccine development. An aging demographic significantly increases demand for improved cancer care and prevention strategies. Local biotech firms partner with international companies to bring cutting-edge vaccines into clinical stages. Efficient healthcare systems support early integration of innovative vaccine options for patients. Continued focus on precision medicine enhances Japan’s regional role.
The Cancer Vaccine market in China emerges as a key regional driver in Asia Pacific with expanding R&D activities and increasing clinical trial volume for Cancer Vaccine. Large patient populations create significant demand for both preventive and therapeutic vaccines across multiple cancer types. Growing domestic capabilities in biotech innovation reduce reliance on imported products and support market diversification. Investments in technology platforms, including mRNA and recombinant vaccines, enhance competitive positioning. Healthcare access improvements facilitate wider reach of advanced oncology treatments. Strategic industry collaborations accelerate commercial entry into global markets.
Latin America Cancer Vaccine Market Trends
The Cancer Vaccine market in Latin America remains a developing segment with gradual uptake and expanding clinical focus. Rising incidence of cancer and increasing public awareness of preventive measures support incremental demand for vaccine interventions. Brazil stands as the largest contributor in the region, with growing adoption trends and participation in multinational research programs. Healthcare infrastructure improvements and broader vaccine distribution networks help extend access to immunotherapy options. Collaborations with global pharmaceutical companies introduce innovative cancer vaccine solutions locally. Market potential continues to strengthen as regional hospitals integrate new treatment modalities.
The Brazil Cancer Vaccine market accounted for the largest market revenue share in Latin America in 2025, with growing research interests and expanding healthcare services that support oncology vaccine adoption. Rising cancer prevalence and improving patient awareness encourage uptake of preventive vaccines and clinical trial participation. Partnerships between local biotech firms and global developers enhance portfolio diversification and commercial reach. Investment in cold chain and distribution networks improves accessibility across urban centres. Established healthcare providers incorporate novel vaccine options into treatment pathways for eligible patients. This cumulative momentum drives Brazil’s regional influence.
Middle East & Africa Cancer Vaccine Market Trends
The Cancer Vaccine market in Middle East and Africa show emerging growth, as healthcare systems expand and clinical trial capacity increases. Improving infrastructure and rising incidence of cancer drive interest in both preventive and therapeutic vaccine solutions. Increased engagement with international research initiatives strengthens regional participation in global oncology advances. Countries such as Saudi Arabia and the UAE invest in medical innovation and attract collaborations that support market entry. Awareness campaigns and educational initiatives promote adoption of new Cancer Vaccine. This progressive trend enhances MEA’s role as a developing regional market.
The Saudi Arabia Cancer Vaccine market contributes to MEA’s cancer vaccine advancement with growing clinical research activity and investments in healthcare capabilities that support vaccine development. Increasing cancer prevalence and demand for advanced treatment options encourage interest in innovative immunotherapies. Strategic partnerships with international pharmaceutical companies help introduce new vaccine technologies. Expansion of oncology care services strengthens local adoption and patient access. Awareness of preventive cancer measures rises among the population. This expanding participation positions Saudi Arabia as a noteworthy market within MEA.
Key Cancer Vaccine Company Insights
Merck & Co., Inc. maintains a strong position in the Cancer Vaccine indusry through its broad oncology portfolio, robust immuno-oncology pipeline, and global commercialization capabilities. GSK plc holds a substantial market presence supported by its long standing expertise in vaccines, adjuvant technologies, and continued investment in therapeutic cancer vaccine research. Dendreon Pharmaceuticals LLC. is a prominent specialized player, known for its work in autologous cellular immunotherapy for prostate cancer.
Dynavax Technologies strengthens the market through proprietary immune-stimulating platforms that enhance vaccine efficacy. Ferring B.V. focuses on niche oncology and urology driven vaccine approaches. Amgen, Inc. and Bristol-Myers Squibb Company leverage deep immunology and oncology expertise to advance therapeutic vaccine strategies. Moderna, Inc. and Sanofi drive innovation through mRNA platforms and strategic collaborations, while AstraZeneca supports market growth by integrating Cancer Vaccine within its broader immuno-oncology portfolio.
Key Cancer Vaccine Companies:
The following key companies have been profiled for this study on the cancer vaccine market.
- Merck & Co., Inc.
- GSK plc
- Dendreon Pharmaceuticals LLC.
- Dynavax Technologies.
- Ferring B.V.
- Amgen, Inc.
- Moderna, Inc.
- Sanofi
- AstraZeneca
- Bristol-Myers Squibb Company
Recent Developments
-
In January 2026, Merck and Moderna’s personalized cancer vaccine in combination with Keytruda demonstrated a sustained reduction in recurrence or death risk by 49 percent in high-risk melanoma patients over a five-year follow-up period, reinforcing confidence in innovative vaccine approaches and prompting expansion of clinical studies into additional cancer types including lung and bladder cancer.
-
In April 2024, BioNTech SE published three-year follow-up data from a Phase 1 trial of the individualized mRNA cancer vaccine candidate autogene cevumeran in 16 patients with resected pancreatic ductal adenocarcinoma, showing that in 8 of 16 patients the treatment elicited high-magnitude T cells specific to encoded neoantigens, that 98 percent of those neoantigen-targeting T cells were de novo, over 80 percent of vaccine-induced neoantigen-specific T cells persisted up to three years, that 6 of 8 responders remained disease free while 7 of 8 non-responders developed tumor recurrence, and that these immune responses correlated with prolonged median recurrence-free survival.
-
In January 2024, the University of Oxford and GSK Press announced a collaboration valued at approximately USD 63 million between GSK and the University of Oxford, which was focused on cancer vaccine research. The partnership aimed to explore immuno-prevention strategies and neoantigen identification, with the objective of informing future cancer vaccine development designed to stop cancer progression before full disease onset.
Cancer Vaccine Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 11.02 billion
Revenue forecast in 2033
USD 23.31 billion
Growth rate
CAGR of 11.30% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Vaccine type, indication type, technology type, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Merck & Co., Inc.; GSK plc; Dendreon Pharmaceuticals LLC.; Dynavax Technologies.; Ferring B.V.; Amgen, Inc.; Moderna, Inc.; Sanofi; AstraZeneca; Bristol-Myers Squibb Company
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Cancer Vaccine Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global Cancer Vaccine market report based on vaccine type, indication type, technology type, and region:

-
Vaccine Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Preventive Cancer Vaccine
-
Therapeutic Cancer Vaccine
-
Others
-
-
Indication Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Prostate Cancer
-
Bladder Cancer
-
Melanoma
-
Cervical Cancer
-
Other
-
-
Technology Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Recombinant Cancer Vaccine
-
Whole-cell Cancer Vaccine
-
Viral Vector and DNA Cancer Vaccine
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the cancer vaccine market include Merck & Co., Inc., GSK plc, Dendreon Pharmaceuticals LLC., Dynavax Technologies., Ferring B.V., Amgen, Inc., Moderna, Inc., Sanofi, AstraZeneca, and Bristol-Myers Squibb Company.
b. Key factors that are driving the cancer vaccine market growth include factors such as the rising prevalence of cancer, increasing awareness for vaccination, and growing focus on personalized vaccines among key players in the market.
b. The global cancer vaccine market size was estimated at USD 9.93 billion in 2025 and is expected to reach USD 11.02 billion in 2026.
b. The global cancer vaccine market is expected to grow at a compound annual growth rate of 11.30% from 2026 to 2033 to reach USD 23.31 billion by 2033.
b. North America dominated the cancer vaccine market with a share of 35.47% in 2025. This is attributable to the increasing burden of cancer, growing research and development in field of vaccine development, and a growing healthcare infrastructure in the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










