- Home
- »
- Biotechnology
- »
-
Cell Therapy Monitoring Kits Market Size, Share Report, 2033GVR Report cover
![Cell Therapy Monitoring Kits Market Size, Share & Trends Report]()
Cell Therapy Monitoring Kits Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Cell Activation Kit, Stem Cell Kit), By Application (Oncology, Autoimmune Diseases), By Therapy Type, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-908-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
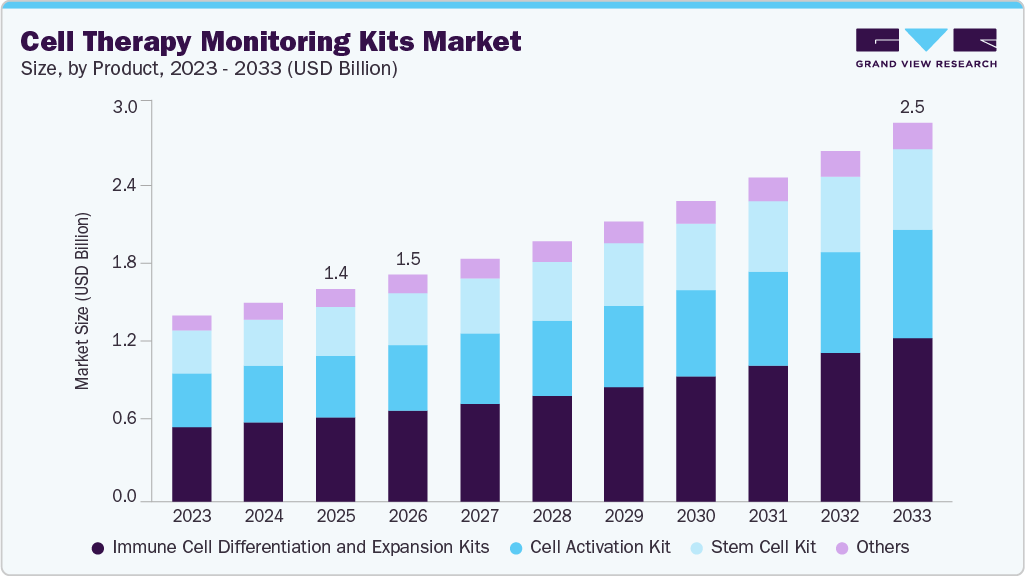
Cell Therapy Monitoring Kits Market Summary
The global cell therapy monitoring kits market size was estimated at USD 1.4 billion in 2025 and is projected to reach USD 2.5 billion by 2033, growing at a CAGR of 7.6% from 2026 to 2033. The market growth is driven by the rising adoption of cell therapies and the increasing need for quality monitoring.
Key Market Trends & Insights
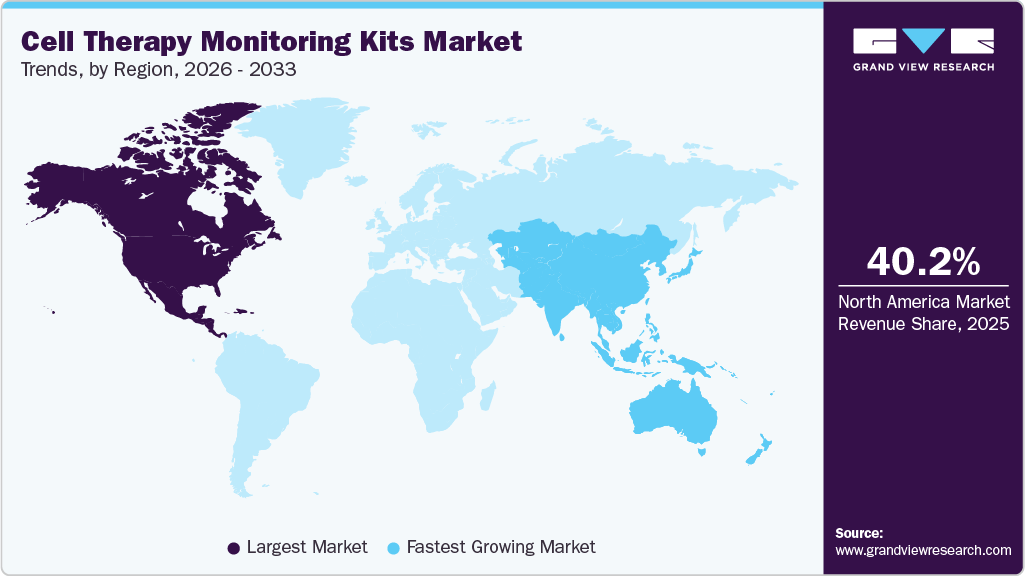
- The North America cell therapy monitoring kits market held the largest global share of 40.2% in 2025.
- The U.S. cell therapy monitoring kits industry is expected to grow significantly from 2026 to 2033.
- By product, the immune cell differentiation and expansion kits segment held the largest market share of 40.0% in 2025.
- By application, the oncology segment held the largest market share in 2025.
- By therapy type, the Chimeric Antigen Receptor (CAR) T-Cell therapies segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.4 Billion
- 2033 Projected Market Size: USD 2.5 Billion
- CAGR (2026-2033): 7.6%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing market
Rapid growth of cell & gene therapiesThe rapid expansion of the cell and gene therapy pipeline is a key driver of the cell therapy monitoring kits market. Advanced modalities such as chimeric antigen receptor T-cell (CAR-T) therapy, stem cell therapies, and gene-edited treatments are advancing across clinical and commercial stages. As approvals and clinical trials increase, the demand for reliable monitoring solutions rises, with multiple checkpoints across development and manufacturing driving consistent kit usage.
Approved Cellular And Gene Therapy Products
Product & Trade Name
Manufacturer
SKYSONA (elivaldogene autotemcel)
bluebird bio, Inc.
STRATAGRAFT (allogeneic cultured keratinocytes and dermal fibroblasts in murine collagen-dsat)
Stratatech Corporation
TECARTUS (brexucabtagene autoleucel)
Kite Pharma, Inc.
TECELRA (afamitresgene autoleucel)
Adaptimmune LLC
VYJUVEK (beremagene geperpavec-svdt)
Krystal Biotech, Inc.
WASKYRA (etuvetidigene autotemcel)
Fondazione Telethon ETS
YESCARTA (axicabtagene ciloleucel)
Kite Pharma, Incorporated
ZEVASKYN (prademagene zamikeracel)
Abeona Therapeutics, Inc.
ZYNTEGLO (betibeglogene autotemcel)
bluebird bio, Inc.
ZOLGENSMA (onasemnogene abeparvovec-xioi)
Novartis Gene Therapies, Inc.
Source: FDA, Secondary Research, Grand View Research
In addition, cell therapies require strict evaluation throughout processes such as cell isolation, modification, expansion, and infusion. Parameters, including viability, purity, identity, and potency, must be continuously monitored to meet regulatory standards and ensure safety. Due to their sensitivity to process variations, these therapies require real-time, batch-specific monitoring, leading to higher testing frequency and greater adoption of specialized monitoring kits.
Strong Regulatory Requirements (GMP, FDA, EMA)
Stringent regulatory requirements are a major driver of the cell therapy monitoring kits market. As cell therapies are living, highly variable products, regulatory bodies such as the U.S. Food and Drug Administration and the European Medicines Agency enforce strict guidelines for quality control, safety, and efficacy. Manufacturers must comply with frameworks like Good Manufacturing Practice and Good Laboratory Practice, which require comprehensive monitoring at every stage of development and production.
To meet these standards, continuous assessment of critical parameters such as cell identity, purity, potency, and sterility is essential. Monitoring kits play a crucial role in ensuring batch-to-batch consistency, traceability, and regulatory compliance, reducing the risk of product failure or rejection. As regulatory scrutiny intensifies alongside increasing therapy approvals, the reliance on advanced monitoring solutions continues to grow, driving sustained market demand.
Market Concentration & Characteristics
The cell therapy monitoring kits industry demonstrates a moderate degree of innovation, driven by the complexity of advanced therapies such as CAR-T cell therapy and gene-edited treatments. Advancements in technologies such as next-generation sequencing, flow cytometry, and digital PCR have improved the accuracy and speed of cell analysis. At the same time, the integration of automation and AI enables real-time, high-throughput monitoring. These innovations enhance process control, reduce errors, and support scalable, standardized testing, thereby accelerating adoption across research and commercial settings.
The market exhibits a moderate to high level of M&A activity, as companies seek to enhance technological capabilities and expand their portfolios. Major players are acquiring specialized firms in cell analysis, assay development, and advanced diagnostics to strengthen their position in the cell and gene therapy value chain. These deals also focus on integrating automation and digital solutions to enable end-to-end monitoring and improve efficiency. As competition intensifies, M&A remains a key strategy for market consolidation and innovation.
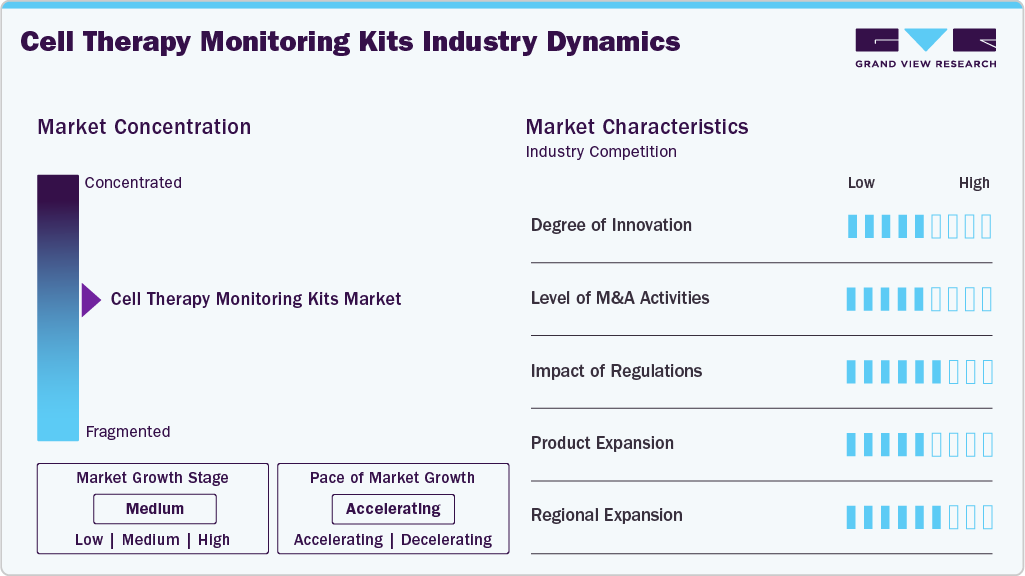
Regulations strongly influence the cell therapy monitoring kits market, as agencies such as the U.S. Food and Drug Administration and the European Medicines Agency mandate strict quality, safety, and efficacy standards. Compliance with Good Manufacturing Practice and Good Laboratory Practice requires continuous monitoring of parameters such as cell identity, purity, and potency, driving demand for monitoring kits. While this increases costs and complexity, it also promotes the adoption of advanced, standardized solutions, thereby supporting market growth.
Product expansion is a key trend in the cell therapy monitoring kits industry, with companies broadening their portfolios to address multiple analytical needs, including cell viability, identity, purity, and potency. Vendors are introducing advanced and integrated solutions, including flow cytometry, PCR-based assays, and sequencing, to enable end-to-end monitoring. In addition, application-specific kits for therapies like CAR-T cell therapy are gaining traction, with a focus on high-throughput, user-friendly formats that enhance efficiency and scalability.
Regional expansion is a key growth strategy in the market, with companies expanding across North America, Europe, and Asia-Pacific to tap into rising demand. Growth is driven by increasing investments in cell therapy research, expanding clinical trials, and improving healthcare infrastructure. Companies are strengthening local manufacturing and partnerships to enhance market access and capture emerging opportunities, particularly in Asia Pacific. For instance, in April 2025, Thermo Fisher Scientific Inc. opened an Advanced Therapies Collaboration Center in the U.S., strengthening end-to-end cell therapy workflows and enhancing the adoption of integrated analytical and monitoring solutions.
Product Insights
The immune cell differentiation and expansion kits segment accounted for the largest market share of 40.0% in 2025 and is expected to grow at the fastest CAGR throughout the forecast period. The growth is driven by increasing adoption of CAR-T and other immunotherapies, which require precise monitoring of cell growth, phenotype, and functionality. The growing demand for standardized, GMP-compliant, and automated manufacturing processes further supports the segment's dominance.
The cell activation kit segment is expected to grow at a significant CAGR during the forecast period, driven by rising demand for efficient T-cell and NK cell activation in advanced cell therapies, expanding CAR-T pipelines, and the growing need for standardized, high-efficiency activation protocols for scalable, automated manufacturing processes.
Application Insights
The oncology segment dominated the cell therapy monitoring kits market in 2025 with a 36.1% share and is expected to grow at the fastest CAGR during the forecast period. The growth is driven by the rising prevalence of cancer, increasing adoption of cell-based immunotherapies such as CAR-T, and a strong pipeline of oncology-focused clinical trials. In addition, high demand for precise monitoring of cell quality, efficacy, and safety in complex cancer therapies further supports segment growth.
The autoimmune diseases segment is expected to grow at a significant CAGR during the forecast period, driven by increasing research on cell-based therapies for immune modulation, rising prevalence of autoimmune disorders, and expanding clinical pipelines targeting conditions such as rheumatoid arthritis and multiple sclerosis. In addition, growing demand for precise monitoring of immune response and treatment efficacy further supports segment growth.
Therapy Type Insights
The Chimeric Antigen Receptor (CAR) T-Cell therapies segment dominated the cell therapy monitoring kits industry in 2025 with a 34.2% share and is expected to grow at the fastest CAGR throughout the forecast period, driven by increasing approvals, strong clinical outcomes, and high demand for advanced monitoring in complex manufacturing processes. In addition, expanding clinical pipelines and growing investments from biopharma companies are accelerating adoption. The need for stringent quality control and real-time monitoring further strengthens segment growth.
The Natural Killer (NK) Cell Therapies segment is expected to grow at a significant CAGR during the forecast period, driven by increasing focus on allogeneic, off-the-shelf therapies, lower risk of graft-versus-host disease, and advancements in NK cell engineering. Moreover, expanding clinical pipelines and rising investment in next-generation immunotherapies are accelerating segment growth.
End Use Insights
The hospitals and clinics segment dominated the cell therapy monitoring kits market in 2025 with a share of 41.5%. This can be attributed to the high volume of cell therapy procedures performed in clinical settings, the availability of advanced infrastructure, and the presence of skilled healthcare professionals. Moreover, increasing adoption of approved cell therapies and integration of point-of-care monitoring solutions further drive segment growth.
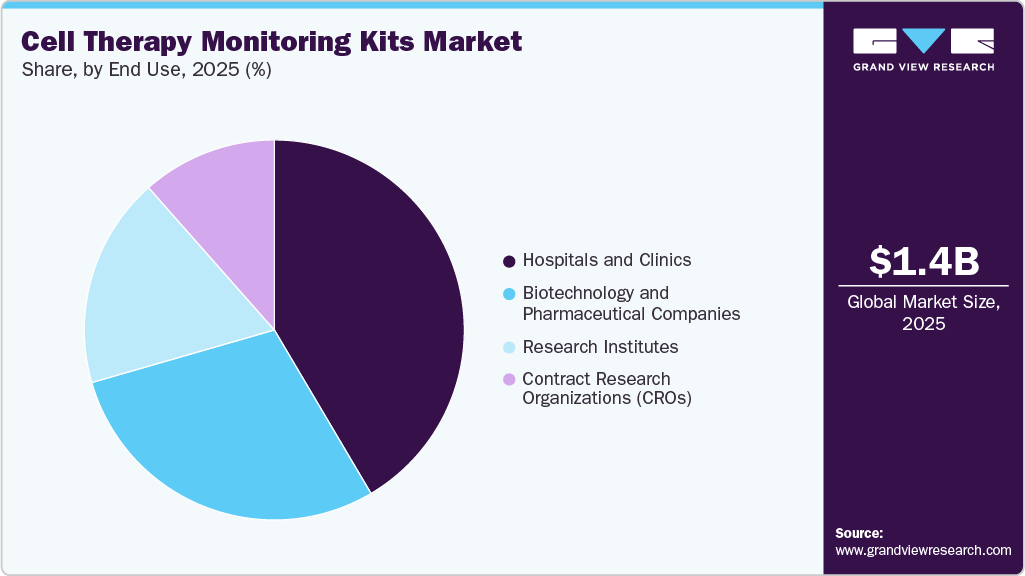
The biotechnology and pharmaceutical companies segment is expected to grow at the fastest CAGR during the forecast period. The growth of the segment is driven by increasing investment in epigenetics-based drug discovery, rising focus on precision medicine, and growing development of targeted therapies for cancer and other chronic diseases.
Regional Insights
North America dominated the cell therapy monitoring kits market with a share of 40.2% in 2025, driven by the strong presence of leading biopharmaceutical companies, advanced healthcare infrastructure, and high clinical trial activity. Supportive regulatory frameworks from agencies like the U.S. Food and Drug Administration, along with significant R&D investments, continue to accelerate the development and commercialization of advanced therapies such as CAR-T cell therapy.
U.S. Cell Therapy Monitoring Kits Market Trends
The U.S. remains the global hub for cell therapy innovation, characterized by a dense network of biotech firms, CDMOs, and academic centers. High commercialization rates, strong venture funding, and accelerated approval pathways from the U.S. Food and Drug Administration position the country at the forefront of advanced therapy deployment.
Europe Cell Therapy Monitoring Kits Market Trends
Europe represents a mature, steadily growing market, supported by strong government funding, collaborative research initiatives, and a well-established regulatory framework under the European Medicines Agency. Increasing adoption of advanced therapies, coupled with rising investments in regenerative medicine and biomanufacturing capabilities, is driving regional market growth.
The UK cell therapy monitoring kits market is anticipated to grow during the forecast period. The UK is distinguished by its integrated innovation framework, combining academic excellence with government-backed commercialization programs. Dedicated initiatives in advanced therapy manufacturing and a favorable regulatory environment are strengthening its global competitiveness.
The cell therapy monitoring kits market in Germany is anticipated to grow during the forecast period. Germany stands out for its precision-driven manufacturing capabilities and strong translational research ecosystem. The country plays a central role in scaling cell therapy production within Europe, supported by industrial expertise and alignment with European Medicines Agency standards.
Asia Pacific Cell Therapy Monitoring Kits Market Trends
Asia Pacific is expected to grow at the fastest CAGR of 12.9% during the forecast period, fueled by expanding healthcare infrastructure, rising clinical trial activity, and increasing government support in countries such as China, Japan, and India. Cost advantages, a large patient population, and growing investments by global and regional players are accelerating the adoption and production of cell therapies. For instance, in March 2026, PHC Corporation launched the LiCellGrow system, enabling real-time in-line metabolic monitoring and automated culture control to enhance process analytics, quality, and scalability in CGT manufacturing.
The China cell therapy monitoring kits market is expected to grow over the forecast period. China is rapidly emerging as a high-volume development and manufacturing base, driven by aggressive biotech investments and streamlined clinical pathways. The presence of numerous domestic players and large-scale trial activity supports fast market expansion.
The cell therapy monitoring kits market in Japan is notable for its early adoption of regenerative medicine frameworks, enabling conditional and time-limited approvals. This regulatory agility, along with strong domestic innovation, accelerates patient access to novel cell therapies.
MEA Cell Therapy Monitoring Kits Market Trends
The Middle East and Africa cell therapy monitoring kits industry is in a developing stage, with growth driven by improving healthcare infrastructure, increasing awareness of advanced therapies, and gradual investments in biotechnology. Countries such as the United Arab Emirates and Saudi Arabia are leading regional adoption through healthcare modernization initiatives and strategic partnerships with global biotech firms.
The Kuwait cell therapy monitoring kits market is at a nascent stage, with growth primarily driven by healthcare modernization and increasing adoption of advanced treatment modalities. Strategic collaborations with international providers are gradually building local capabilities in specialized therapies.
Key Cell Therapy Monitoring Kits Company Insights
The cell therapy monitoring kits industry is led by major players such as Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group, and BD Biosciences, which benefit from strong global reach, broad portfolios, and advanced analytical technologies. Their focus on GMP-compliant reagents, integrated platforms (flow cytometry, PCR, and cell analysis systems), and scalable solutions supports the increasing demand from pharmaceutical and biotechnology companies for standardized, regulatory-aligned monitoring.
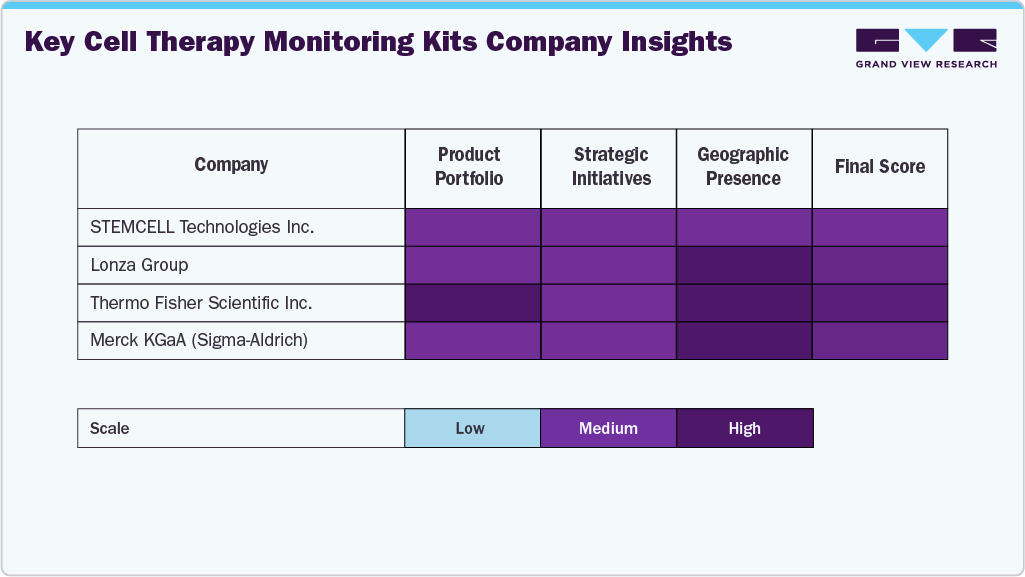
Specialized companies such as STEMCELL Technologies Inc., Miltenyi Biotec, Sartorius AG, PerkinElmer Inc., and Cell Biolabs Inc. drive innovation through advanced cell separation, assay development, and real-time monitoring technologies, catering to evolving research, translational, and clinical manufacturing needs.
Key Cell Therapy Monitoring Kits Companies:
The following key companies have been profiled for this study on the cell therapy monitoring kits market.
- STEMCELL Technologies Inc.
- Lonza Group
- Thermo Fisher Scientific Inc.
- Merck KGaA (Sigma-Aldrich)
- BD Biosciences
- Miltenyi Biotec
- GE Healthcare
- Sartorius AG
- PerkinElmer Inc.
- Cell Biolabs Inc.
Recent Developments
-
In March 2026, Lineage Cell Therapeutics launched a preclinical COR1 cell therapy program, increasing demand for advanced monitoring kits to ensure cell quality, consistency, and scalability in allogeneic manufacturing processes.
-
In January 2025, CellFE launched the T-Rest resting T cell kit in the U.S., advancing CAR-T manufacturing workflows and driving demand for specialized monitoring kits to ensure cell quality, viability, and gene-editing efficiency.
“We're thrilled to launch T-Rest - the first-in-class resting T cell commercial product - to offer a new paradigm for cell therapy manufacturing.”
- Alla Zamarayeva, CEO of CellFE.
Cell Therapy Monitoring Kits Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.5 billion
Revenue forecast in 2033
USD 2.5 billion
Growth rate
CAGR of 7.6% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, therapy type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; UAE; South Africa; Kuwait
Key companies profiled
STEMCELL Technologies Inc.; Lonza Group; Thermo Fisher Scientific Inc.; Merck KGaA (Sigma-Aldrich); BD Biosciences; Miltenyi Biotec; GE Healthcare; Sartorius AG; PerkinElmer Inc.; Cell Biolabs Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Cell Therapy Monitoring Kits Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the cell therapy monitoring kits market report based on product, application, therapy type, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Cell Activation Kit
-
Immune Cell Differentiation and Expansion Kits
-
Stem Cell Kit
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Autoimmune Diseases
-
Cardiovascular Diseases
-
Neurological Disorders
-
Musculoskeletal Disorders
-
Others
-
-
Therapy Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Stem Cell Therapies
-
Chimeric Antigen Receptor (CAR) T-Cell Therapies
-
Natural Killer (NK) Cell Therapies
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Clinics
-
Research Institutes
-
Biotechnology and Pharmaceutical Companies
-
Contract Research Organizations (CROs)
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global cell therapy monitoring kits market size was estimated at USD 1.4 billion in 2025 and is expected to reach USD 1.5 billion in 2026
b. The global cell therapy monitoring kits market is expected to grow at a compound annual growth rate of 7.6% from 2026 to 2033 to reach USD 2.5 billion by 2033.
b. North America dominated the cell therapy monitoring kits industry with a share of 40.2% in 2025, driven by a strong presence of leading biopharmaceutical companies, advanced healthcare infrastructure, and high clinical trial activity.
b. STEMCELL Technologies Inc.; Lonza Group; Thermo Fisher Scientific Inc.; Merck KGaA (Sigma-Aldrich); BD Biosciences; Miltenyi Biotec; GE Healthcare; Sartorius AG; PerkinElmer Inc.; Cell Biolabs Inc.
b. The rapid expansion of the cell therapy pipeline is a key driver of the cell therapy monitoring kits market. Advanced modalities such as chimeric antigen receptor T-cell (CAR-T cell therapy), stem cell therapies, and gene-edited treatments are growing across clinical and commercial stages.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










