- Home
- »
- Pharmaceuticals
- »
-
Cholera Vaccines Market Size, Share, Industry Report, 2030GVR Report cover
![Cholera Vaccines Market Size, Share & Report]()
Cholera Vaccines Market (2025 - 2030) Size, Share & Analysis Report By Type (Whole cell V. Cholerae O1 Recombinant With B-Subunit), By Product (Vaxchora, Dukoral), By Distribution Channel (Hospital Pharmacy, Retail Pharmacy), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-069-9
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Cholera Vaccines Market Summary
The global cholera vaccine market size was valued at USD 93.9 million in 2024 and is projected to reach USD 163.4 million by 2030, growing at a CAGR of 10.0% from 2025 to 2030. The market is expected to grow due to increased awareness of cholera outbreaks in endemic regions.
Key Market Trends & Insights
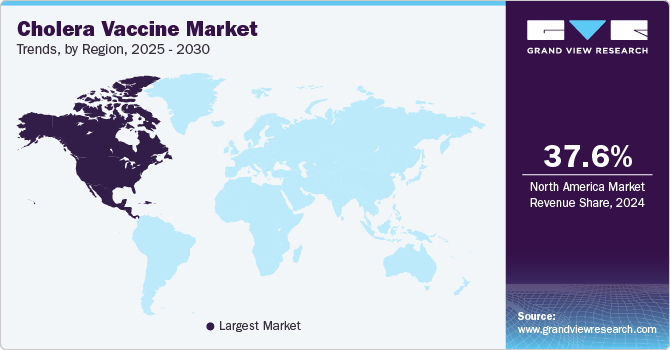
- North America cholera vaccines market dominated the global market and accounted for the largest revenue share of 37.6% in 2024.
- The cholera vaccine market in Canada is expected to grow at the fastest CAGR of 9.9% in 2024 over the forecast period.
- Based on type, the killed oral 01 and 0139 segment dominated the market and accounted for the largest revenue share of 60.0% in 2024 and is expected to grow at the fastest CAGR of 11.5% over the forecast period.
- Based on product, the dukoral segment dominated the market and accounted for the largest revenue share of 32.5% in 2024.
- Based on distribution channel, the hospital pharmacy segment dominated the market and accounted for the largest revenue share of 45.5% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 93.9 Million
- 2030 Projected Market Size: USD 163.4 Million
- CAGR (2025-2030): 10.0%
- North America: Largest market in 2024
Various vaccination options and oral and injectable formulations are the characteristics. The efforts of international health organizations and government initiatives have strengthened vaccination campaigns to reduce the outbreak. Furthermore, persistent research and technological advancements are expected to enhance the efficacy and accessibility of the vaccine program.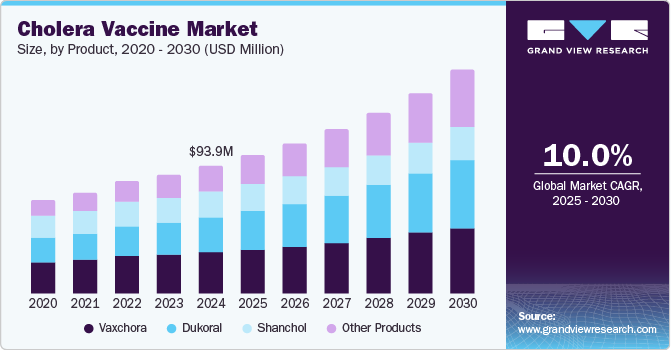
Cholera is caused by indigestion of food or water contaminated with the bacterium Vibrio cholera. According to the National Institutes of Health, as of August 2024, 60,834 new cases and 227 deaths were reported worldwide. The prevention of cholera has increased the demand for oral cholera vaccines, diagnostic tests, and oral rehydration solutions. As international travel increases and awareness of cholera risk grows, more individuals and organizations seek reliable vaccination options.
The expansion of international travel and migration has raised the need for vaccination in endemic regions, urging more individuals and organizations to seek reliable vaccination options. Improved innovation and availability of vaccines have led the government and NGOs to combat cholera outbreaks in endemic regions. As the global focus on infectious disease prevention intensifies, the market is poised to expand.
The GTFCC estimated that around a 90% reduction in cholera deaths is expected by 2030. This primarily includes eliminating cholera cases in 20 countries. Preventive measures such as providing safe water supplies, sanitation, and hygiene are maintained. Voluntary Intellectual Property (IP) licensing and technology transfer also play a vital role in increasing the production and distribution of oral cholera vaccines.
Type Insights
The killed oral o1 and o139 segment dominated the market and accounted for the largest revenue share of 60.0% in 2024 and is expected to grow at the fastest CAGR of 11.5% over the forecast period. The segment growth can be attributed to its effective vaccination strategies and increased awareness. Prevention measures, including the utilization of oral administrative offers and facilitating vaccination campaigns, are significantly improving. The rise in cholera cases has prompted public health agencies to prioritize vaccines as a frontline defense due to climate change and urbanization.
The expected increase of the segment uses advanced recombinant techniques to boost immune response, increasing their effectiveness in preventing cholera, especially in vulnerable populations. In addition, the versatility of these vaccines allows for broader coverage against various strains, which is crucial in regions experiencing diverse serotype prevalence. Therefore, the demand for killed oral o1 and o139 vaccines are anticipated to rise, solidifying the global cholera vaccine market.
Product Insights
Dukoral dominated the market and accounted for the largest revenue share of 32.5% in 2024 on account of its increased awareness and effective preventive measures. In addition, it provides immunity against both vibrio cholerae O1 and enterotoxigenic Escherichia coli. The ease of oral administration formulation eliminates the need for injections, enhancing patient compliance and accessibility. As cholera remains a concern in several regions, such as Africa and Asia Pacific, the vaccine plays an effective role in public health campaigns and vaccination programs. Its oral administration makes it easier to distribute and administer.
Vaxchora is expected to grow at the fastest rate CAGR of 14.1% over the forecast period, contributing to its effectiveness and ease of administration. It offers a simplified administrative route that enhances adherence, especially in regions with endemic cholera. It is effective against cholera serogroup O1, which boosts its protective capacity, thus making it accessible for travelers and vulnerable populations. In addition, the demand for awareness of cholera and its preventive measures has impacted worldwide.
Distribution Channel Insights
Hospital pharmacies in the distribution channel segment dominated the market and accounted for the largest revenue share of 45.5% in 2024, attributed to its proper storage, administration of vaccines, and integrated patient care systems. They are equipped with the necessary infrastructure to maintain the cold chain required for the efficacy of vaccines, which is essential for sensitive products such as cholera vaccines.
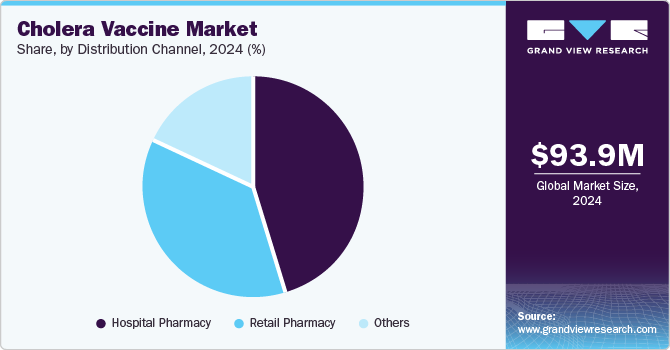
In addition, hospitals play a major role in managing outbreaks and providing adherence. Therefore, there is a significance in promoting readily- available on-site vaccinations to affected patients and preventive measures to manage the cholera outbreak.
Regional Insights
North America cholera vaccines market dominated the global market and accounted for the largest revenue share 37.6% in 2024, on account of its increased awareness and preventive measures. While cholera is not endemic in North America, the region is significant in funding and supporting global health initiatives, such as providing vaccines to countries with high cholera incidence. In addition, the rising concerns over international travel and migration have highlighted the need for vaccination to protect travelers and humanitarian workers going to cholera-prone areas. Centers for Disease Control and Prevention (CDC) are also active in promoting cholera vaccinations.
U.S. Cholera Vaccine Market Trends
The cholera vaccine market in the U.S. dominated North America, with the largest revenue share in 2024, attributed to its role in international vaccine distribution. While cholera is not a significant concern in the U.S., the country has been proactive in addressing the disease in regions where it remains endemic, such as parts of Africa and the Caribbean. In addition, awareness of the importance of global health security, especially climate change and increasing migration, has driven U.S. investments in developing and distributing vaccines.
The cholera vaccine market in Canada is expected to grow at the fastest CAGR of 9.9% in 2024 over the forecast period, contributing to increased travel and migration patterns. For instance, Bavarian Nordic announced the availability of Vaxchora, the only single-dose oral vaccine approved in Canada, in September 2024. To prevent the illness while traveling internationally, Canada is expanding its vaccination programs for safety protocols. Therefore, the development of guidelines for citizens enhances the potential growth of the cholera vaccine market in Canada.
Asia Pacific Cholera Vaccine Market Trends
The cholera vaccine market in the Asia Pacific accounted for a significant revenue share in 2024, attributed to its increased awareness and effective campaigns. The region is quite vulnerable to cholera outbreaks due to frequent natural disasters, dense population, and inadequate sanitation. This results in public health authorities prioritizing vaccinations to prevent the spread of the disease. In addition, international health organizations and local health authorities are facilitating vaccines by enhancing distribution channels and educating several guidelines to avoid disease outbreaks. Furthermore, the demand for facilitating immunization programs has increased among the communities due to the support of government authorities.
The cholera vaccine market in China has grown significantly due to its awareness of the potential health risks and preventive measures. The government has invested in expanding the healthcare infrastructure and enhancing the disease surveillance system, facilitating the effective distribution of vaccines. For instance, Zambia has signed a memorandum of understanding with China to establish the first cholera manufacturing plant in South Africa in October 2024. The project costs USD 37.0 million, alongside three million doses, are to be produced through a joint venture between Zambia Industrial Development Corporation and Jijia International Medical Technology Corporation. Therefore, these elements contribute to the growth of the cholera vaccine market in China.
Key Cholera Vaccines Company Insights
Some key players in the market are Valneva SE, Sanofi S.A, Emergent BioSolutions Inc., and others. These companies employ various strategies to gain a competitive edge, such as launching new products to cater to diverse patient needs, focusing on personalized medicine to enhance treatment efficacy, and leveraging emerging markets to tap into increasing awareness about cholera. Furthermore, these businesses engage in collaborative research initiatives with healthcare organizations to drive innovation while integrating digital health solutions for enhanced patient access and adherence.
-
Valneva SE specializes in developing, manufacturing, and commercializing prophylactic vaccines to prevent infectious diseases and provide adherence. Dukoral, an active immunization vaccine, is consumed, especially by travelers heading toward the endemic regions. In addition, the company is advancing its cholera vaccine program by enhancing its formulation and accessibility.
-
Sanofi S.A. develops a wide range of pharmaceutical products. Vaxchora is a vaccine used by adults and children aged two to prevent cholera disease. The company has continued advancing its cholera vaccine efforts, improving vaccine accessibility and affordability, especially in endemic areas. This includes efforts to increase the supply of oral cholera vaccines to countries experiencing outbreaks and working closely with organizations such as WHO to ensure broader distribution.
Key Cholera Vaccines Companies:
The following are the leading companies in the cholera vaccines market. These companies collectively hold the largest market share and dictate industry trends.
- Valneva SE
- Sanofi S.A.
- Emergent BioSolutions Inc.
- PaxVax Inc.
- EuBiologics Co., Ltd.
- Shantha Biotechnics Limited
- Hilleman Laboratories
- Astellas Pharma Inc.
- Incepta Vaccine Ltd.
- Vabiotech
Recent Developments
-
In April 2024, EuBiologics Co., Ltd. announced that a new cholera vaccine, Euvichol-S, had received WHO prequalification and is now made available to countries globally. According to UNICEF, the overall supply of oral cholera vaccines reached approximately 50 million doses in 2024 compared to 38 million doses in 2023. Therefore, this new product is anticipated to help EuBiologics Co., Ltd. produce volumes of vaccines at a lower cost.
-
In February 2023, Emergent BioSolutions Inc. entered a partnership with Bavarian Nordic. The company acquired the rights to the Vaxchora vaccine. In addition, it acquired Emergent’s manufacturing site in Bern, Switzerland, and other facilities in San Diego, California. Furthermore, it is declared that around 280 Emergent employees are expected to join the company.
Cholera Vaccine Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 101.3 million
Revenue forecast in 2030
USD 163.4 million
Growth Rate
CAGR of 10.0% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Report updated
November 2024
Quantitative units
Revenue in USD Million and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, Product, Distribution Channel, Region
Regional scope
North America, Europe, Asia Pacific, Latin America, MEA
Country scope
U.S., Canada, Mexico, UK, Germany, France, Italy, Spain, Denmark, Sweden, Norway, Japan, China, India, Australia, South Korea, Thailand, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait
Key companies profiled
Valneva SE; Sanofi S.A.; Astellas Pharma, Inc.; Emergent BioSolutions Inc.; PavVax Inc.; EuBiologics Co., Ltd.; Shantha Biotechnics Limited; Hilleman Laboratories; Astellas Pharma Inc.; Incepta Vaccine Ltd.; Vabiotech
Customization scope
Free report customization (equivalent to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
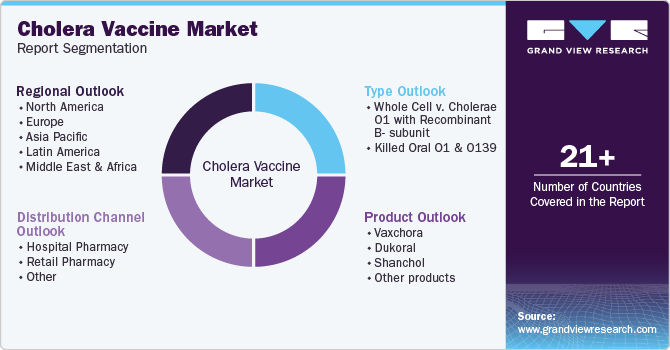
Global Cholera Vaccines Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and analyzes the latest industry trends in each sub-segment from 2018 to 2030. For this study, Grand View Research has segmented the global cholera vaccine market report based on type, product, distribution channel, and region.
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Whole Cell v. Cholerae O1 with Recombinant B- subunit
-
Killed Oral O1 and O139
-
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Vaxchora
-
Dukoral
-
Shanchol
-
Other products
-
-
Distribution Channel Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospital Pharmacy
-
Retail Pharmacy
-
Other
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










