- Home
- »
- Medical Devices
- »
-
Clinical Trial Equipment & Ancillary Solutions Market Report, 2033GVR Report cover
![Clinical Trial Equipment & Ancillary Solutions Market Size, Share & Trends Report]()
Clinical Trial Equipment & Ancillary Solutions Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Sourcing, Supply/Logistics, Service), By Phase (Phase I, II, III, IV), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-533-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Clinical Trial Equipment & Ancillary Solutions Market Summary
The global clinical trial equipment & ancillary solutions market size was valued at USD 3.21 billion in 2024 and is projected to reach USD 6.88 billion by 2033, growing at a CAGR of 8.92% from 2025 to 2033. The growth of the market is due to the increasing complexity and globalization of clinical trials, which demand highly specialized, protocol-specific equipment and logistics support.
Key Market Trends & Insights
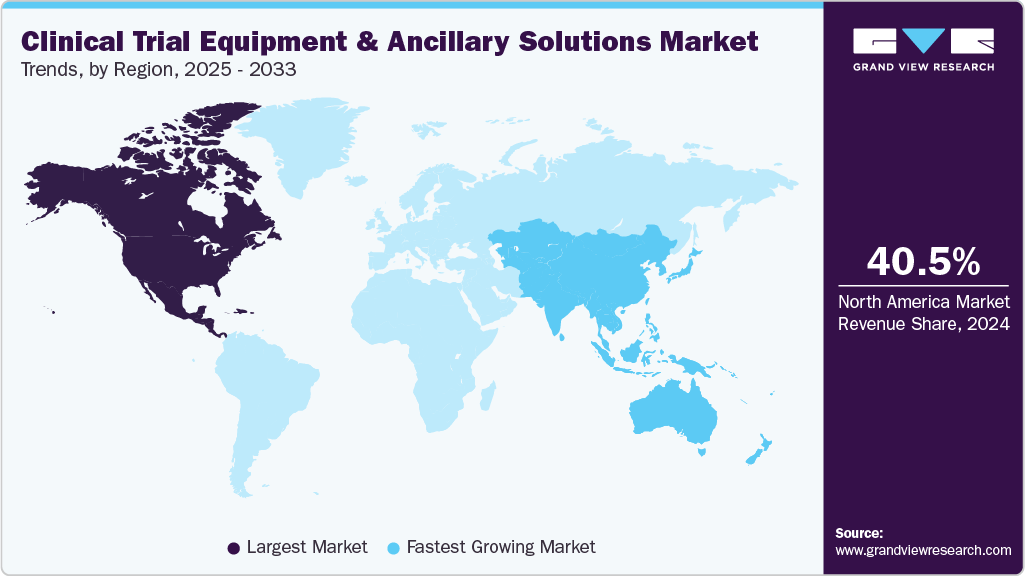
- North America clinical trial equipment & ancillary solutions market held the largest share of 40.46% of the global market in 2024.
- The clinical trial equipment & ancillary solutions industry in the U.S. is expected to grow significantly over the forecast period.
- By type, the supply and logistics segment led the market with the largest revenue share of 36.9% in 2024.
- Based on phase, the phase III segment led the market with the largest revenue share in 2024
Market Size & Forecast
- 2024 Market Size: USD 3.21 Billion
- 2033 Projected Market Size: USD 6.88 Billion
- CAGR (2025-2033): 8.92%
- North America: Largest market in 2024
- Asia-Pacific: Fastest growing market
The rapid adoption of decentralized and hybrid trial models requires remote monitoring devices, wearable sensors, and home-use diagnostic kits, significantly expanding the scope of ancillary services. The transformation of clinical trial models, especially the increase of decentralized and hybrid trials, has significantly raised the demand for specialized equipment and support services that facilitate remote, site-less, and home-based research settings. The traditional model, which relied heavily on centralized trial sites, is being replaced by more patient-focused approaches, requiring remote monitoring devices, mobile health kits, wearables, and courier-enabled logistics for biological samples. For example, the U.S. FDA has promoted decentralized clinical trials (DCTs) through its 2023 draft guidance, urging sponsors to adopt digital health technologies and remote care tools to enhance patient access and data quality. These developments are leading CROs and sponsors to acquire reliable, temperature-sensitive packaging, validated medical devices for home use, and real-time tracking systems to oversee equipment shipments across different regions. This shift has transformed the role of clinical trial equipment suppliers from passive vendors into active, integrated service partners responsible for ensuring regulatory compliance, proper calibration, and delivery schedules aligned with study protocols.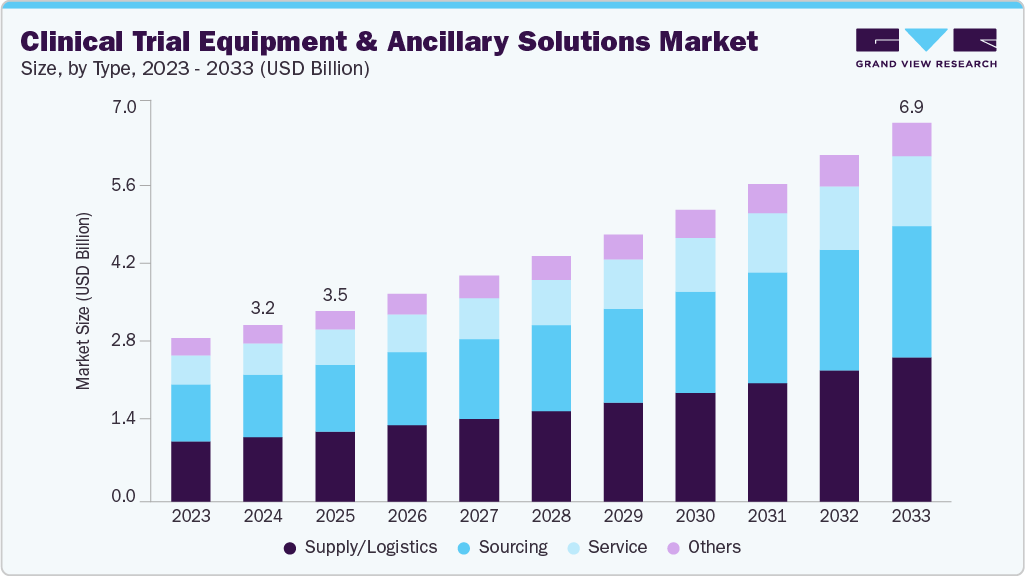
Additionally, government and non-profit funding organizations are increasingly supporting early-stage research, translational medicine, and investigator-initiated trials, all of which depend on timely access to ancillary trial supplies. Initiatives like the NIH’s All of Us Research Program and the European Union’s Horizon Europe framework are expanding the boundaries of patient cohort diversity and precision diagnostics, which require adaptive and flexible equipment supply strategies. For example, Horizon Europe allocates nearly USD 111.49 billion (€95 billion) toward science and innovation through 2027, with multiple projects focused on personalized medicine, wearable monitoring, and telehealth-integrated clinical trials. These initiatives generate demand for site-ready kits, cloud-connected monitoring devices, and digitally traceable shipping for reagents and biologics.
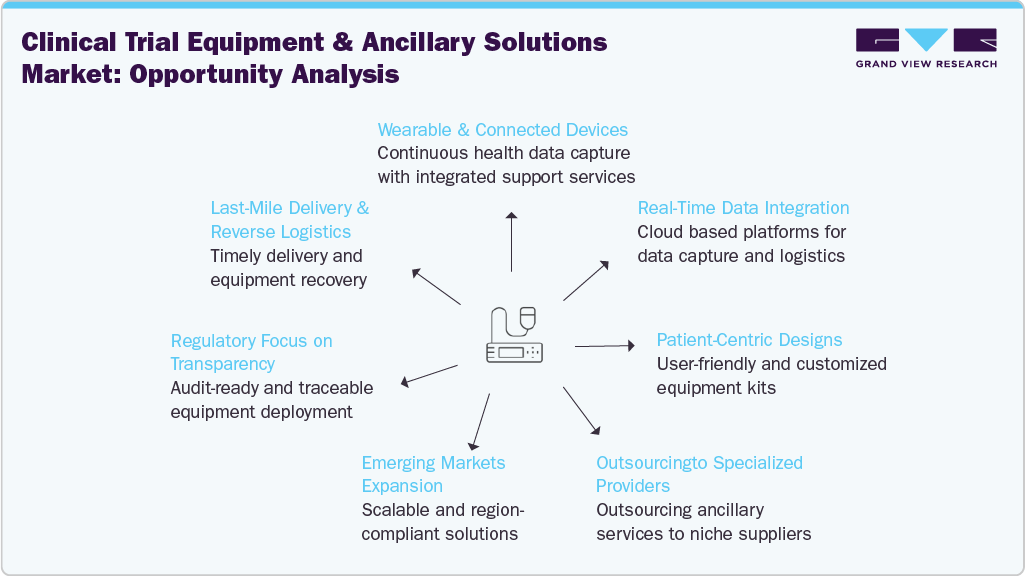
Opportunity Analysis
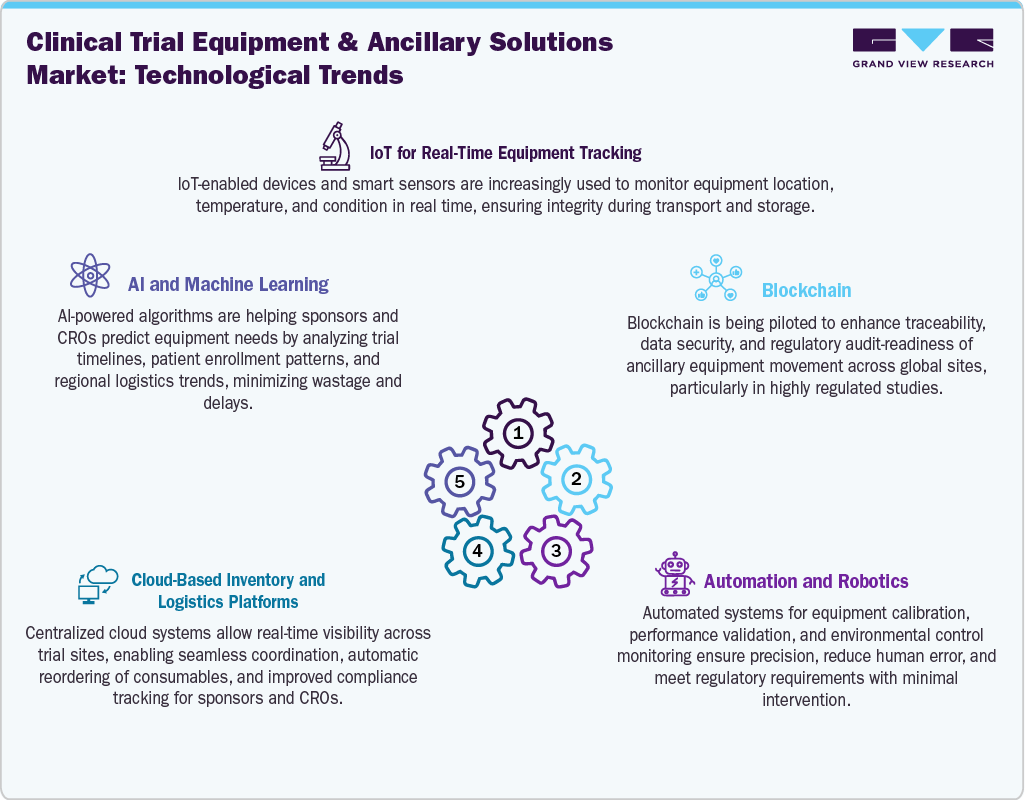
Technological Advancements
The global clinical trial equipment and ancillary solutions market is rapidly changing, driven by innovations that improve efficiency, compliance, and patient engagement. Digital integration leads the way, with cloud-based inventory systems, IoT-enabled tracking devices, and AI-powered demand forecasting tools becoming more common. Wearable health technology, remote monitoring devices, and smart kits enable seamless data collection and decrease the need for on-site visits in decentralized trials. Automation in equipment calibration, temperature-controlled logistics, and packaging validation boosts accuracy and reduces manual work. Additionally, blockchain and RFID technologies are being explored to ensure secure, transparent, and traceable handling of critical clinical supplies across regions. This move toward connected, smart systems not only streamlines trial operations but also creates new opportunities for vendors to deliver bundled, end-to-end ancillary solutions tailored to modern trial needs.
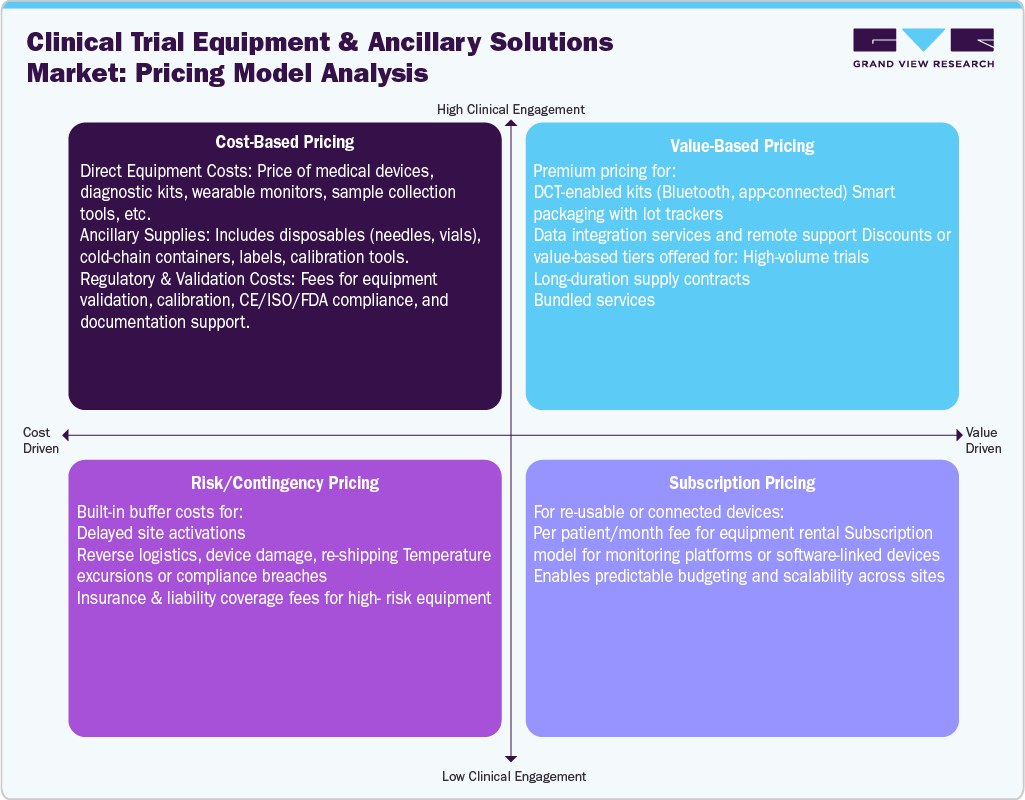
Pricing Analysis
Type Insights
On the basis of type segment, the market is classified into sourcing, supply/logistics, service, and others. The supply/logistics segment accounted for the largest revenue share in the clinical trials equipment & ancillary solutions industry of 36.9% in 2024. The growth of the segment is due to increasing complexity and globalization of clinical trials. As trials expand to multiple regions, including emerging markets in Asia-Pacific, Latin America, and Eastern Europe-demand has surged for reliable cold chain logistics, time-sensitive deliveries, and compliant transport of temperature-sensitive and perishable supplies.
The sourcing segment is anticipated to grow at a considerable CAGR during the forecast period. The segment growth is driven due to the rising complexity of trial protocols and the increasing diversity of required ancillary materials. Clinical trials today require a wide array of equipment, including wearable devices, point-of-care diagnostics, customized sample kits, and region-specific medical supplies-prompting sponsors and CROs to outsource sourcing functions to specialized vendors with global procurement networks.
Phase Insights
Based on phase segmentation, the market is divided into Phase I, Phase II, Phase III, and Phase IV. The Phase III segment held the largest market share in 2024 due to its extensive scale, longer duration, and complex logistical needs. Phase III trials typically involve large patient populations across multiple geographic regions, requiring high volumes of medical equipment, diagnostic kits, and ancillary supplies. This phase also requires strong supply chain coordination to ensure consistent availability of calibrated devices, biosample kits, and temperature-sensitive materials at each trial site.
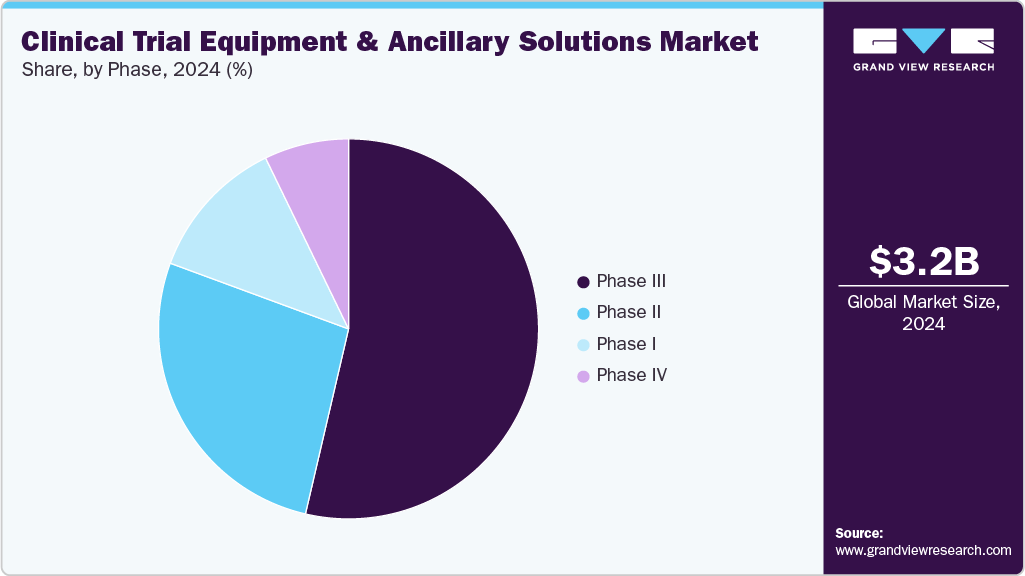
The Phase I segment is expected to grow at the fastest CAGR during the forecast period. Its growth is driven by an increase in early-stage clinical trials, especially in oncology, rare diseases, and cell and gene therapies. These trials often need specialized and customized equipment, such as first-in-human (FIH) dosing kits, rapid-response biosample collection tools, and highly controlled infusion devices, all requiring precise sourcing and handling.
Regional Insights
North America dominated the market with the largest market share of 40.46% in 2024, owing to the increased R&D investments, the presence of global players, and rising efforts to come up with newer patents. In addition, the increasing number of clinical trials is also fueling market growth.
U.S. Clinical Trial Equipment & Ancillary Solutions Market Trends
The clinical trial equipment & ancillary solutions market in the U.S. held the largest share in 2024. This is attributed to the increasing number of clinical trials being performed in the U.S. For instance, according to the National Clinical Trials Registry (NCT), around 12,326 clinical trials studies were undergoing across various phases for the treatment of cancer in 2022.
Europe Clinical Trial Equipment & Ancillary Solutions Market Trends
The clinical trial equipment & ancillary solutions market in Europe is expected to grow significantly due to rising focus on clinical trial studies, favorable government initiatives and policies, compliance with the Good Clinical Practice (GCP) standards, larger patient pools and rising number of clinical trials are some of the key factors fueling the market growth.
The clinical trial equipment & ancillary solutions market in Germany is driven by the factors such as advanced medical expertise and easy access to patients, low rate of saturation of competing trials, and high number of participants contributing to the clinical research studies.
The clinical trial equipment & ancillary solutions market in the UK is driven by factors such as rising government spending on clinical research and rising number of clinical trial studies in the region. According to the National Institute for Health Research (NIHR), UK, clinical research is worth USD 3.74 billion each year and includes USD 2.07 billion from commercial sources, which supports around 47,000 jobs.
Asia Pacific Clinical Trial Equipment & Ancillary Solutions Market Trends
Asia Pacific is expected to be the fastest growing market due to growing investment by developed countries in the regions and various amendments made by the regulatory agencies to facilitate local manufacturing & contract services.
The clinical trial equipment & ancillary solutions market in China held the largest share in 2024. The growth is attributed to the government initiatives to regulate the clinical trials as per the global standards and better infrastructure, which includes 4,000 hospital beds. Initially in China, Centre of Drug Evaluation (CDE) was responsible was regulating the clinical trials
The clinical trial equipment & ancillary solutions market in Japan is expected to grow over the forecast period. The regulatory process is supportive of faster clinical trial approval. For instance, clinical trials can be started once the probable benefits are demonstrated by carrying out a relatively smaller number of clinical trials.
The clinical trial equipment & ancillary solutions market in India is anticipated to grow at the lucrative CAGR over the forecast period. This can be attributed to the increasing government funds. The government of India focuses on expanding affordable healthcare facilities in the country. This propelled the growth of the clinical trials sector in India. In addition, local Indian players such as Vitas Pharma (Hyderabad) and Bugworks (Bengaluru) ventured into drug development activities.
Key Clinical Trials Equipment & Ancillary Solutions Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures merger, partnership & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key Clinical Trials Equipment & Ancillary Solutions Companies:
The following are the leading companies in the clinical trials equipment & ancillary solutions market. These companies collectively hold the largest market share and dictate industry trends.
- Ancillare, LP
- Imperial CRS, Inc.
- Woodley Equipment Company Ltd.
- Thermo Fisher Scientific, Inc.
- Parexel International (MA) Corporation
- Emsere (formerly MediCapital Rent)
- Quipment SAS
- IRM
- Marken (UPS-United Parcel Service)
- Myonex
- Yourway
Recent Developments
-
In January 2025, Ancillare announced the launch of its state-of-the-art cold chain management service capabilities for clinical trial supplies. The enhancements focus on maintaining temperature integrity, expanding global logistics capabilities, and incorporating real-time tracking and visibility tools.
-
In May 2024, Emsere entered into a partnership agreement with the Clario to enhance ophthalmic imaging capacity in clinical trials by integrating Clario’s clinical trial software with Emsere’s barcoded imaging devices. The expansion addresses needs in retina-focused ophthalmology studies, improving equipment provision and imaging workflow efficiency.
Clinical Trial Equipment & Ancillary Solutions Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 3.47 billion
Revenue forecast in 2033
USD 6.88 billion
Growth rate
CAGR of 8.92% from 2025 to 2033
Actual data
2018 - 2024
Forecast period
2025 - 2033
Report updated
July 2025
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, phase, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE
Key companies profiled
Ancillare, LP; Imperial CRS, Inc.; Woodley Equipment Company Ltd.; Thermo Fisher Scientific, Inc.; Parexel International (MA) Corporation; Emsere (MediCapital Rent); Quipment SAS; IRM; Marken (UPS-United Parcel Service); Myonex; Yourway
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Clinical Trials Equipment & Ancillary Solutions Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global clinical trial equipment & ancillary solutions market report based on type, phase and region.
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Sourcing
-
Procurement
-
Equipment
-
Ancillaries
-
-
Rental
-
Equipment
-
Ancillaries
-
-
-
Supply/Logistics
-
Transportation
-
Packaging
-
Others
-
-
Service
-
Calibrations
-
Equipment servicing
-
Others
-
-
Others
-
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global clinical trial equipment & ancillary solutions market size was estimated at USD 3.21 billion in 2024 and is expected to reach USD 3.47 billion in 2025.
b. The global clinical trial equipment & ancillary solutions market is expected to grow at a compound annual growth rate of 8.92% from 2025 to 2033 to reach USD 6.88 billion by 2033.
Which segment accounted for the largest clinical trial equipment & ancillary solutions market share?b. North America dominated the clinical trial equipment & ancillary solutions market with a share of 40.5% in 2024. This is attributable to the high R&D investments, the strong presence of global players, and their efforts to come up with newer patents.
b. Some key players operating in the clinical trial equipment & ancillary solutions market include Ancillare LP, Imperial Clinical Research Services, Woodley Equipment Company LTD, Thermo Fisher Scientific Inc., PAREXEL International Corp., etc.
b. Key factors that are driving the clinical trial equipment & ancillary solutions market growth include rising R&D investments, the evolving need for clinical trials, and the increase in the growth of pharmaceutical and medical industries.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










