Dacryocystitis Market Size & Trends
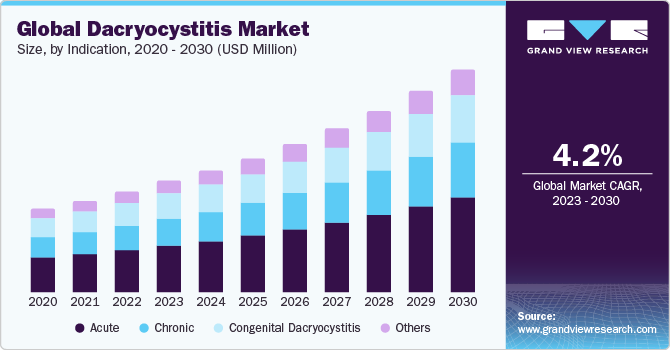
The global dacryocystitis market size expected to grow at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030. The increasing prevalence of eye infections and technological advancements, growing clinical research, and subsequent new product launches boost the market growth. Furthermore, according to research by the IOSR Journal of Dental and Medical Sciences (IOSR-JDMS), chronic dacryocystitis is more prevalent than acute dacryocystitis. Moreover, chronic dacryocystitis exhibits various stages of presentation, including symptoms such as mucoid discharge, chronic conjunctivitis, conjunctival hyperemia, and epiphora.
The increasing prevalence of eye disorders, particularly in the elderly population, is a significant driver for the dacryocystitis market. Age-related changes in the eye's anatomy and function can lead to lacrimal duct obstruction and an increased risk of dacryocystitis. As the global population ages, the demand for dacryocystitis treatments is expected to rise.
The rising incidence of eye diseases is anticipated to drive the utilization of dacryocystitis treatment during the projected timeframe. Dacryocystitis is an inflammation of the lacrimal or tear sacs, resulting in symptoms such as redness, pain, and discomfort in the lower corner of the eye. According to the Review of Ophthalmology findings, the progression from congenital dacryocele to acute dacryocystitis may manifest in approximately 20% to 72.5% of patients. Consequently, there has been a surge in the need for dacryocystitis management.
Furthermore, advances in medical technology have led to more effective and less invasive surgical procedures for treating dacryocystitis, such as endoscopic dacryocystorhinostomy (DCR) and balloon dacryoplasty. These innovative techniques offer quicker recovery times and fewer complications, making them more appealing to patients and healthcare providers.
Indication Insights
Based on the indication, the dacryocystitis market is segmented into acute, chronic, congenital dacryocystitis, and others. The acute segment held the largest market share in 2023. Acute dacryocystitis typically presents with abrupt onset symptoms, often accompanied by fever and eye discharge. Bacterial infections are the common cause behind acute dacryocystitis, and treatment with antibiotics typically results in the resolution of the infection within a few days. Individuals with acute dacryocystitis tend to experience more severe symptoms compared to those with chronic dacryocystitis. Nevertheless, in all age groups, acute dacryocystitis typically responds rapidly to oral antibiotic treatment.
Distribution Channel Insights
The market is segmented into institutional and retail sales based on distribution channels. Institutional sales held the largest market in 2023. Institutional sales channels ensure that the appropriate products and treatments reach patients, healthcare providers, and facilities. With the increasing demand for effective treatments and medical devices, healthcare institutions are presented with a significant opportunity within the dacryocystitis market.
Regional Insights
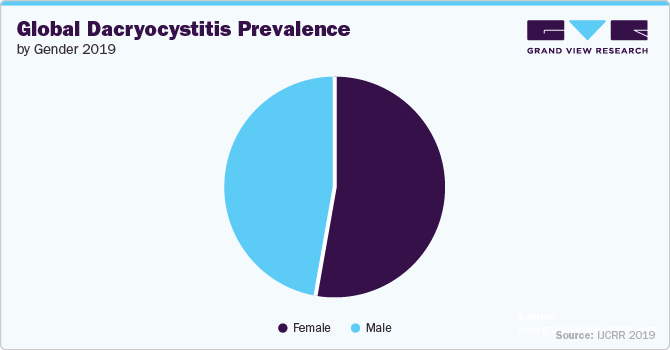
In 2023, North America held the largest market share. Within North America, the U.S. has experienced a significant increase in dacryocystitis patients. A 2019 study conducted by Grant D. Gilliland et al. revealed that in the adult population, females are more commonly affected by dacryocystitis. It is noted that 70-83% of dacryocystitis cases occur in females. In contrast, congenital dacryocystitis occurs with equal frequency in both sexes. Additionally, the relaxation of government regulations in the U.S. and the availability of funding from various organizations, such as the National Institute of Health, have created opportunities for researchers to invest more in drug discovery for this condition.
Competitive Insights
Key players operating in the market are Mylan NV, GlaxoSmithKline PLC, FCI Ophthalmics, Kaneka Corporation, Aurobindo Pharma, Orex Pharma Pvt Ltd, and Pfizer Inc. The market participants are constantly working towards new product development, M&A activities, and other strategic alliances to gain new market avenues. The following are some instances of such initiatives.
-
In April 2019, Alembic Pharmaceuticals obtained approval from the U.S. FDA for its Abbreviated New Drug Application (ANDA) for Tobramycin Ophthalmic Solution USP, 0.3%.
-
In January 2019, Ocular Therapeutix obtained approval from the U.S. FDA for its Supplemental New Drug Application (sNDA) related to DEXTENZA, specifically for addressing ocular inflammation after ophthalmic surgery.













