- Home
- »
- Clinical Diagnostics
- »
-
Diagnostic Reagents For Infectious Diseases Market, 2033GVR Report cover
![Diagnostic Reagents For Infectious Diseases Market Size, Share & Trends Report]()
Diagnostic Reagents For Infectious Diseases Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Molecular Reagents, Immunoassay Reagents), By Disease Area (Respiratory Infections, Enteric Infections), By Technology, By End Use, By Region, and Segment Forecasts
- Report ID: GVR-4-68040-817-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
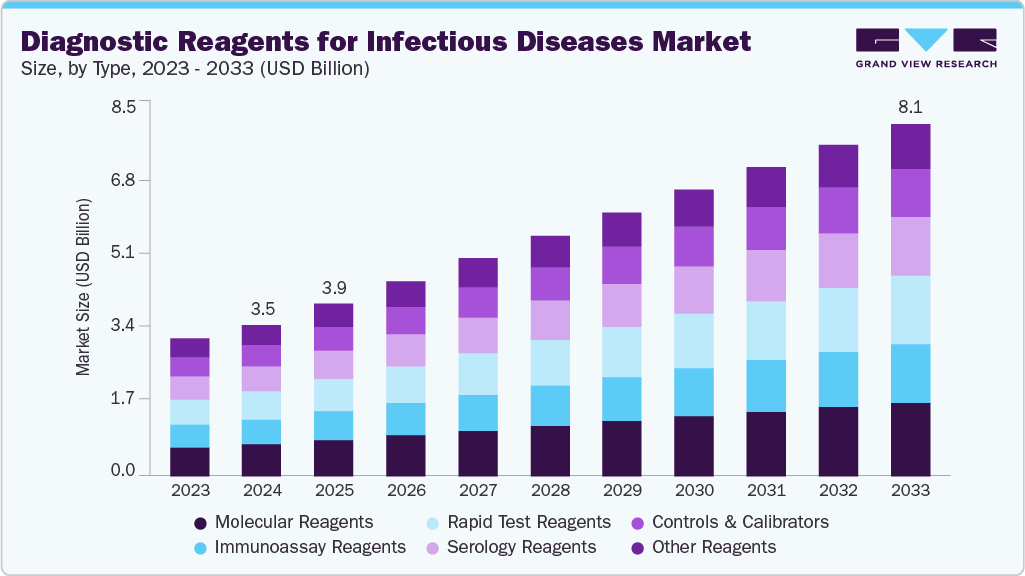
Diagnostic Reagents For Infectious Diseases Market Summary
The global diagnostic reagents for infectious diseases market size was estimated at USD 3,475.31 million in 2024 and is projected to reach USD 8,079.07 million by 2033, growing at a CAGR of 9.30% from 2025 to 2033. The market is primarily driven by the increasing prevalence of respiratory diseases and sexually transmitted infections (STIs).
Key Market Trends & Insights
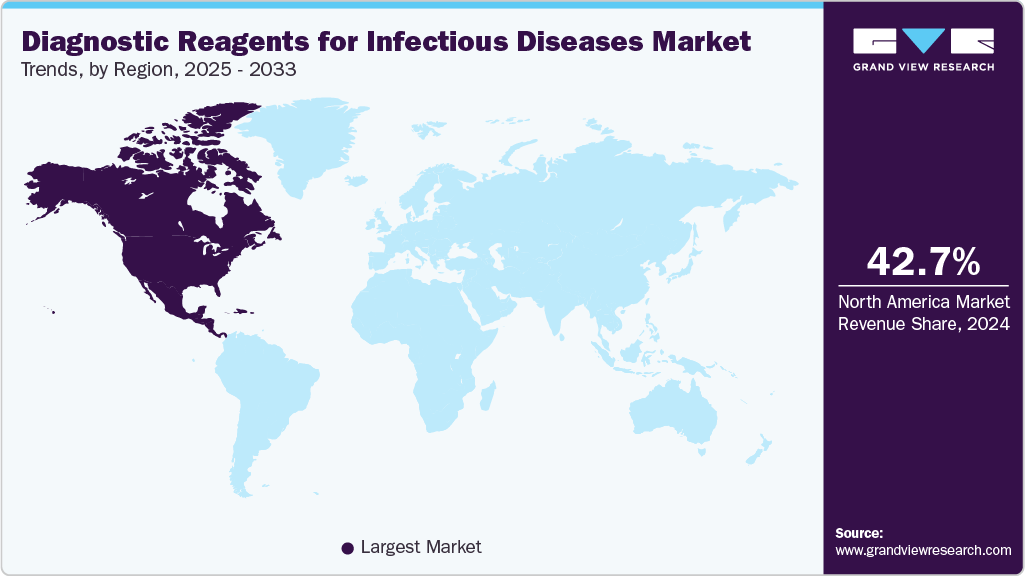
- North America diagnostic reagents for infectious diseases industry dominated the global market and accounted for the largest revenue share of 42.67% in 2024.
- The U.S. led the North American market and held the largest revenue share in 2024.
- By product, the molecular reagents segment dominated the global market with a 21.07% revenue share in 2024.
- By disease area, the respiratory infection segment held the largest revenue share of 25.93% in 2024.
- By technology, the molecular diagnostics segment held the largest revenue share of 29.87% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 3,475.31 Million
- 2033 Projected Market Size: USD 8,079.07 Million
- CAGR (2025-2033): 9.30%
- North America: Largest Market in 2024
Respiratory diseases, including influenza, COVID-19, pneumonia, and tuberculosis, have the highest prevalence worldwide. The UNICEF 2023 report stated that, every year, there are over 1,400 cases of pneumonia per 100,000 children worldwide, or one case per 71 children, with the highest incidences occurring in South Asia (2,500 cases per 100,000 children) and West and Central Africa (1,620 cases per 100,000) in 2023.Moreover, the WHO estimated 10.8 million people worldwide became ill with tuberculosis, including 6.0 million men, 3.6 million women, and 1.3 million. Therefore, the rising prevalence rate is driving strong demand for advanced diagnostic technologies such as molecular diagnostics and point-of-care testing, fueling market growth. In addition, the rapid expansion of telemedicine services and the growing adoption of home-based testing solutions are further contributing to the market’s expansion.
Furthermore, rising global awareness and government-supported screening programs are encouraging early detection and prevention initiatives. The Journal of Global Health reported in May 2025 that the distribution and health impact of infectious disease outbreaks from 1996 to 2023 were globally assessed. The World Health Organization has identified approximately 30 priority pathogens, including monkeypox virus, dengue virus, and influenza A viruses, that could cause global public health emergencies. This updated list emphasizes the significance of focusing efforts on these high-risk infections due to their high transmissibility, virulence, and the scarcity of vaccines and treatment options. As global concerns such as climate change, deforestation, and growing urbanization increase the risk of these illnesses spreading, boosting the demand for infectious disease reagents.

Furthermore, the expansion of advanced molecular technologies in developing regions is playing a crucial role in driving market growth. Growing investments in healthcare infrastructure and increasing access to innovative diagnostic tools are enabling faster, cost-effective, and more accurate detection of infectious diseases. For instance, in January 2024, QIAGEN (Netherlands) launched testing panels for its QIAstat-Dx instruments, along with reagents, in India for rapid and accurate diagnosis of infectious diseases. Such initiatives are accelerating the adoption of molecular diagnostics in developing regions and significantly contributing to market growth.
Moreover, the increasing funding by governments to target pathogen genomic surveillance is boosting the demand for infectious disease reagents associated with molecular diagnostics. In February 2024, the WHO announced that donors had contributed USD 4 million to establish a catalytic grant fund for organizations working in pathogen genomic surveillance. The fund will support pilot projects worldwide, particularly in low- and middle-income countries. As laboratories implement these pilot projects, there is an increased need for molecular diagnostics reagents, including PCR kits, sequencing reagents, and other consumables. This expansion of testing capabilities ensures more frequent and routine use of reagents, creating sustained demand while enhancing global capacity to detect, prevent, and respond effectively to infectious disease outbreaks.
Market Concentration & Characteristics
The market for diagnostic reagents for infectious diseases shows a medium level of innovation, mainly due to ongoing advancements in automation, assay sensitivity, and the combination of immunological and molecular techniques. Next-generation solutions that can detect multiple pathogens at once, like multiplex assays and rapid testing platforms, are being adopted rapidly. This improves workflow efficiency and diagnostic accuracy. Despite the importance of these developments, the majority of inventions focus on improving and refining already-existing technologies rather than bringing about groundbreaking discoveries. To support steady and sustainable growth and meet changing clinical and industry needs, the market places a high priority on enhancing operational efficiency, reliability, and scalability.
Mergers and acquisitions (M&A) are playing an increasingly influential role in shaping the competitive landscape of the diagnostic reagents market. Major players are strategically acquiring specialized companies and complementary technologies to expand their product portfolios, enhance research capabilities, and increase global market reach. For instance, in June 2025, bioMérieux (France) acquired Day Zero Diagnostics (U.S.), a company specializing in infectious disease diagnostics. The acquisition included NGS workflows and reagents that contributed to the company's revenue growth. These strategic alliances enable companies to expand their market share, diversify their product offerings, and speed up the adoption of new technologies, contributing to market growth.
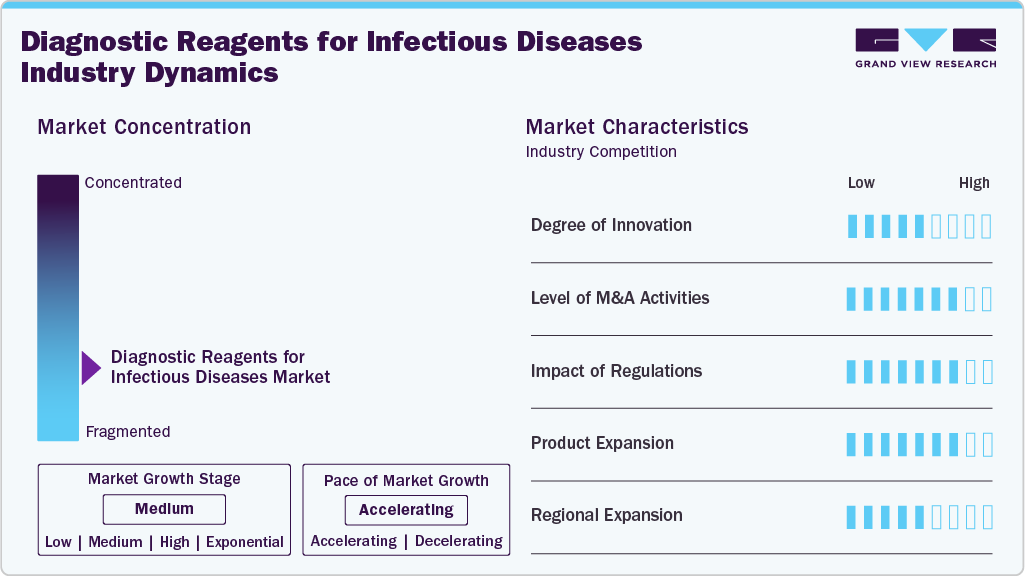
Regulatory frameworks have a substantial influence on the diagnostic reagents market, due to the strict approval and quality standards imposed by global authorities. Agencies such as the U.S. Food and Drug Administration, the European Medicines Agency, and Japan’s Pharmaceuticals and Medical Devices Agency require rigorous validation, testing, and compliance procedures to ensure the safety, efficacy, and reliability of diagnostic products. Although these requirements may extend development timelines and increase costs, they are crucial for ensuring product quality, maintaining market credibility, and safeguarding patient safety.
Product expansion in the diagnostic reagents market for infectious diseases is moderately driven by rising technological adoption and growing investment in R&D. The increasing prevalence of infectious disease outbreaks worldwide underscores the urgent need for rapid, accurate, and scalable testing solutions. In addition, the rising emphasis on early diagnosis, point-of-care testing, and personalized medicine is creating new opportunities for industry players to broaden their reagent portfolios and introduce innovative diagnostic solutions. Companies are leveraging technological advancements to develop reagents capable of multiplex detection, faster turnaround times, and enhanced sensitivity, aligning with evolving clinical requirements.
Regional expansion in the diagnostic reagents market is moderate, with established companies increasingly targeting developing regions including Asia-Pacific and Latin America. These regions have significant growth potential due to rising healthcare costs, improved laboratory infrastructure, and increased awareness of infectious diseases. Likewise, strategic collaborations and partnerships fuel the market growth. For instance, in October 2025, Sysmex partnered with QIAGEN for the distribution and supply of diagnostic products, including reagents for infectious diseases in Japan. Overall, regional expansion offers companies an opportunity in a developing region, fosters long-term growth, and diversifies revenue streams.
Product Insights
The molecular reagents segment held the largest market share of 21.07% in 2024. This strong position reflects the growing reliance on molecular techniques like polymerase chain reaction and next-generation sequencing, both of which require specialized reagents such as primers, probes, enzymes, and buffers to produce accurate and timely results. The preference for molecular reagents stems from their high sensitivity, precision, and ability to detect multiple pathogens at once, making them indispensable in clinical laboratories and research institutions. Furthermore, the recurring need for these reagents in routine diagnostics ensures consistent demand and revenue for manufacturers.
Moreover, the rapid test reagents segment is projected to be the fastest-growing segment during the forecast period. This is due to the higher demand for faster and more accurate diagnostics in the decentralized settings, including home-use tests and point-of-care tests. Further, research studies on the efficacy of the reagents are boosting market growth. For instance, in February 2024, Johns Hopkins Medicine conducted research and showed that rapid COVID-19 tests done at home are valid. Further, rapid antigen tests use a prepared kit containing antibodies and specific reagents for SARS-CoV-2. The test can be taken by anybody, anywhere, and yields results in about 15 minutes.
Disease Area Insights
The respiratory infections segment represents the largest market share of 25.93% in 2024, owing to increasing awareness of the importance of early and accurate diagnosis, as well as the rising prevalence of respiratory diseases such as sinusitis, pharyngitis, bronchitis, pneumonia, and other related infections. The growing burden of respiratory illnesses becomes worse by environmental factors, pollution, and seasonal outbreaks, emphasizing the importance of early detection and effective management. UNICEF 2023 report stated that every year, there are about 1,400 cases of pneumonia per 100,000 children worldwide, or one case per 71 children, with the highest incidences happening in South Asia (2,500 cases per 100,000 children) and West and Central Africa (1,620 cases per 100,000).
In addition, the sexually transmitted infections (STIs) segment is expected to register the fastest CAGR during the forecast period, driven by increasing public awareness of STIs, rising prevalence rates, and intensified government initiatives aimed at screening, prevention, and early diagnosis. The growing burden of respiratory illnesses becomes worse by environmental factors, pollution, and seasonal outbreaks, emphasizing the importance of early detection and effective management. For instance, WHO's new guideline introduced in July 2023, includes target product profiles (TPPs) for point-of-care diagnostic technologies for syphilis, Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis, with the mission of facilitating the development of high-quality STI diagnostics.
Technology Insights
Based on technology, the molecular diagnostics segment accounted for the largest market share of 29.87% in 2024, driven by the increasing adoption of advanced techniques such as real-time PCR, next-generation sequencing (NGS), and conventional PCR for the rapid and accurate identification of pathogens. Molecular diagnostics offer high sensitivity and specificity, enabling early detection of infectious agents and improving patient outcomes. A published study on Frontiers in January 2024 reported that NGS is an important and strong approach in infectious disease epidemiology because it provides great resolution of bacterial genotypes. The growing reliance on molecular technologies in clinical laboratories, coupled with advancements in automation and workflow integration, is expected to further reinforce the dominance of this segment in the global diagnostics market.
Further, the point-of-care reagents segment is expected to be the fastest-growing segment over the forecast period, fueled by the rising demand for on-site and rapid diagnostic testing across diverse healthcare settings. Furthermore, the growth of this segment is supported by technological advancements in portable, user-friendly testing devices that enable quick and accurate results without the need for centralized laboratories. In addition, the increasing prevalence of infectious diseases in developing regions often exacerbated by limited awareness of early diagnosis, inadequate hygiene practices, and constrained healthcare infrastructure fuels the adoption of POC diagnostics. For instance, as per the One Health Trust, South Asia has the world's third largest Human immunodeficiency viruses (HIV) epidemic and the highest Tuberculosis (TB) burden, accounting for more than 25% of the worldwide burden in 2021. The urgent need for accessible and rapid testing solutions in such high-burden regions underscores the critical role of POC reagents in improving early detection, timely intervention, and disease management. With continued innovation and expanding deployment of portable diagnostic technologies, the point-of-care reagents segment is poised for significant growth during the forecast period.
End Use Insights
Hospitals and clinics dominated the market in 2024, accounting for a 24.20% share, supported by the widespread adoption of advanced diagnostic techniques, including molecular diagnostics, immunoassays, point-of-care reagents, and multiplex platforms. These healthcare settings are increasingly investing in comprehensive diagnostic solutions to enable rapid, accurate, and high-throughput testing, which supports timely patient management and improves overall healthcare outcomes. For instance, in July 2022, in PLOS Digital Health, researchers emphasize how early use of digital molecular diagnostics could improve healthcare supply in sub-Saharan Africa in the hospitals.
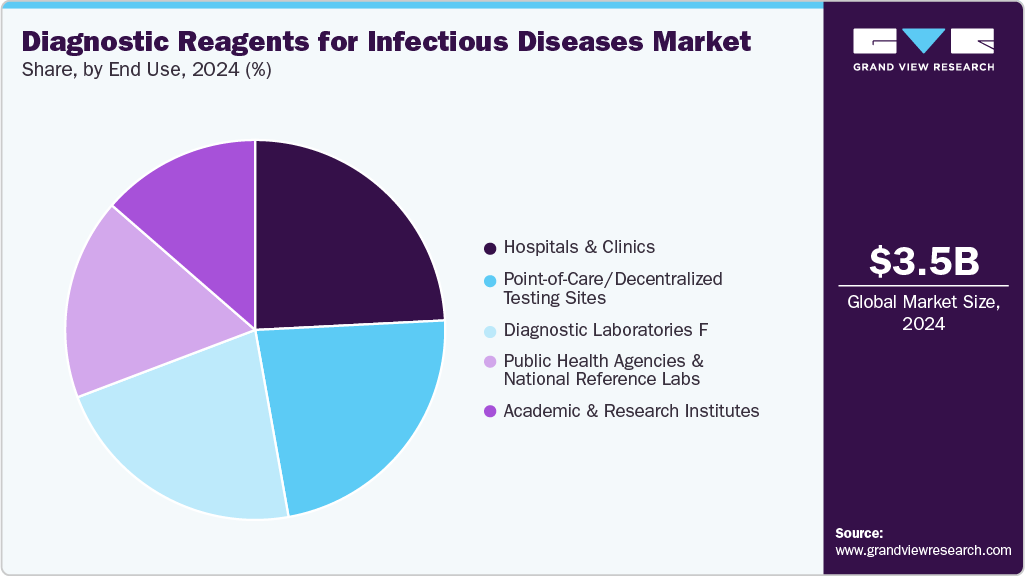
In addition, the diagnostic laboratories segment is expected to witness the fastest CAGR during the forecast period, supported by increased investments in laboratory infrastructure and rising demand for accurate, high-throughput tests. The expansion of modern laboratories equipped with advanced diagnostic instruments allows for faster and more reliable detection of infectious and chronic diseases, meeting the increasing demands of healthcare providers and patients. Furthermore, the growing of laboratory networks and partnerships, combined with the adoption of automation and digital solutions, facilitates the efficient processing of large volumes of samples, improving workflow and reducing turnaround times.
Regional Insights
The diagnostic reagents for infectious diseases industry in North America held the largest share in 2024, driven by several key factors including a well-established healthcare infrastructure and the strong presence of leading industry players such as Abbott (U.S.), Thermo Fisher Scientific (U.S.), and Becton Dickinson (U.S.). The region’s growth is fueled from favorable reimbursement policies, ongoing regulatory and public health initiatives aimed at early disease detection and outbreak management. Furthermore, increasing collaboration between private companies, government agencies, and research institutions is accelerating innovation and market expansion. For instance, in April 2023, Abbott (U.S.) partnered with the Climate Amplified Disease and Epidemics (CLIMADE) consortium, which includes over 100 scientists from academia, public health agencies, and industry interested in using data science technology and diagnostic testing to assess and potentially mitigate the impact of climate change on disease outbreaks. Moreover, the Asia-Pacific region is the fastest-growing market during the forecast period.
U.S. Diagnostic Reagents for Infectious Diseases Market Trends
The U.S. diagnostic reagents for infectious diseases industry is expanding steadily, which is attributed to the rising prevalence of sexually transmitted infections (STIs) resulting from underfunded prevention programs, asymptomatic infections, social and behavioral factors, healthcare disparities, and decreased condom use. For instance, according to the Centers for Disease Control and Prevention, in 2024, there were over 2.2 million reported STIs. Compared to ten years ago, total cases have increased by 13 percent, and congenital syphilis is almost 700 % higher. This growing disease burden is intensifying the need for efficient diagnostic solutions, creating strong demand for reagents used in molecular diagnostics, multianalyte panels, and rapid tests.
Europe Diagnostic Reagents for Infectious Diseases Market Trends
The diagnostic reagents for infectious diseases industry in Europe is expected to grow steadily over the forecast period, due to the high adoption of the technological advanced diagnostics. The region has a strong healthcare and laboratory infrastructure, which enables widespread use of advanced diagnostic testing. Further, ongoing public health initiatives and infectious disease surveillance programs contribute to consistent reagent demand. Moreover, the consumable and repeat-purchase nature of diagnostic reagents provide manufacturers with predictable revenue streams and long-term market stability.
The UK diagnostic reagents for infectious diseases industry is experiencing rapid growth. The expansion of mandatory infectious disease reporting. This regulatory development has led to higher test volumes and compliance driven market expansion. For instance, in March 2025, the UK Health Security Agency (UKHSA) reported that it had expanded the list of infectious diseases that diagnostic laboratories and medical professionals in England are required to report. These government initiatives significantly strengthen the diagnostic ecosystem by encouraging increased reagent utilization in clinical laboratories and public health, fostering the development of new assays, increasing testing frequency as well demand.
The Germany diagnostic reagents market for infectious disease testing is primarily fueled by a convergence of strategic and operational factors. High-frequency testing, supported by a well-established and robust healthcare infrastructure, boosts the demand, while Germany’s extensive network of clinical and reference laboratories enables consistently high-test volumes and rapid reagent turnover. Further, ongoing technological advancements such as molecular diagnostics, multiplex assays, and point-of-care testing platforms are reshaping the diagnostic landscape, shifting the reagent portfolio toward higher-value, specialized tests. This evolution not only improves diagnostic precision but also increases reagent consumption per test, allowing suppliers to focus on higher-margin segments. These dynamics highlight Germany as a mature but growing market for innovative infectious disease diagnostic solutions, with long-term commercial opportunities for reagent manufacturers.
Asia Pacific Diagnostic Reagents for Infectious Diseases Market Trends
The diagnostic reagents for infectious diseases industry in Asia-pacific is experiencing significant growth, driven by multiple demographic, epidemiological, and structural factors. A rapidly aging population, coupled with ongoing expansion of healthcare infrastructure and proactive government initiatives, is supporting increased access to diagnostic services across the region. In addition, the persistently high burden of infectious diseases is creating substantial demand for diagnostic testing, further fueling market expansion. Moreover, the high burden of infectious diseases in the region is increasing the demand for diagnostic tests, fueling market growth. According to UNAIDS, in 2022, about 6.5 million people were living with HIV in the Asia Pacific region. This is the second-highest number of people with HIV compared to other UNAIDS regions.
The diagnostic reagents industry for infectious diseases in Japan is predominantly due to the country’s advanced healthcare infrastructure, a robust focus on early disease detection, and increasing prevalence of both emerging and re-emerging infectious pathogens. With government initiatives promoting rapid diagnostic testing and stringent regulatory frameworks ensuring the safety and efficacy of reagents, healthcare providers are increasingly adopting cutting-edge diagnostic solutions. The market growth is further accelerated by rising public awareness regarding timely disease diagnosis, technological advancements in molecular and immunoassay-based testing, and strong investments from leading life sciences and biotechnology companies. Strategic collaborations between domestic and international players to enhance product portfolios and distribution networks also contribute significantly to market expansion, positioning Japan as a key hub for innovative infectious disease diagnostics.
The China diagnostic reagents industry for infectious diseases is experiencing robust growth due to the approval of a broader range of reagents by regulatory authorities that is expanding the product launches and market access, thereby accelerating adoption across clinical laboratories and point‑of‑care settings. For instance, in May 2024, Cytek Biosciences, Inc. received an approval for 1-laser and 2-laser 6-color TBNK reagent cocktails from the China National Medical Products Administration (NMPA) for clinical diagnostic use on the Cytek Northern Lights Clinical (NL-CLC) cell analysis systems across the China hospitals, laboratories, and clinics. Furthermore, the rising prevalence of infectious diseases coupled with increased government investment in earlydetection infrastructure has increased the demand for reagent‑based diagnostic solutions. Moreover, manufacturers are responding by expanding reagent portfolios specifically for molecular and immuno‑diagnostic assays, which are gaining traction as preferred testing approaches.
Latin America Diagnostic Reagents for Infectious Diseases Market Trends
The diagnostic reagents market for infectious diseases in Latin America is experiencing steady growth, driven by increasing awareness of early disease detection, improvements in healthcare infrastructure, and a rising incidence of infectious diseases across the region. The WHO stated in April 2021 that over 7 million people, primarily in Latin America, are infected with Trypanosoma cruzi, the parasite responsible for Chagas disease. Due to the prevalence of such diseases, the adoption of advanced molecular diagnostic techniques, immunoassays, and rapid point-of-care testing is further increasing the demand for high-quality reagents. Moreover, government initiatives aimed at strengthening disease surveillance, and growing healthcare expenditure from both public and private sectors are supporting the expansion of diagnostic capabilities, collectively presenting lucrative growth opportunities for the market.
The Brazil diagnostic reagents market for infectious diseases is experiencing steady growth owing to rising awareness of the importance of early diagnosis, the expansion of diagnostic infrastructure across public and private healthcare institutions, and increased collaboration to conduct research projects. In addition, in December 2023, the Charles Mérieux Center for Infectious Disease, in Rio Branco (Brazil), became the 22nd member of the GABRIEL network. It contributes to international research projects on tuberculosis, acute respiratory infections, and antimicrobial resistance. Following joining the GABRIEL international scientific network, the Charles Mérieux Center for Infectious Disease in Brazil formed partnerships with network members, increasing its impact on research initiatives and healthcare outcomes in Brazil. Such initiatives are increasing the need for high-quality diagnostic reagents in the country by propelling the use of innovative molecular testing and serological assays.
Middle East and Africa Diagnostic Reagents for Infectious Diseases Market Trends
The diagnostic reagents for infectious diseases industry in the Middle East and Africa is experiencing comparatively slower growth due to challenges such as inadequate laboratory infrastructure, a limited skilled workforce, high costs, and affordability constraints that restrict access to advanced diagnostic solutions. Despite these hurdles, the market is witnessing gradual expansion, driven by increasing awareness of early disease detection, government and donor-led programs, and the significant burden and rising incidence of infectious diseases across the region. In addition, several factors contribute to the emergence and spread of infectious diseases in the region, including population displacement, socioeconomic disparities, climate change, changes in land and water use, and the high prevalence of metabolic and other underlying health conditions. Population mobility across disease hotspot regions, mass gatherings, and altered human-animal interactions further increases the need for effective diagnostic reagents, creating opportunities for market growth and investment.
The Saudi Arabia diagnostic reagents market for infectious disease testing is primarily fueled by the rapid expansion of molecular diagnostic platforms, especially PCR‑based assays, which continue to dominate reagent spending. High demand is driven by a rising prevalence of infectious diseases in the country, coupled with the legacy effects of the COVID‑19 pandemic and renewed focus on pathogen surveillance. In addition, major government initiatives under Vision 2030, along with substantial investment in healthcare infrastructure and the expansion of diagnostic laboratories, enabling high reagent consumption across both the private and public sectors.
Key Diagnostic Reagents For Infectious Diseases Company Insights
The diagnostic reagents for infectious diseases industry features several key players driving innovation and adoption. Leading companies include F. Hoffmann-La Roche Ltd, Abbott, QIAGEN, and among others. These firms are heavily investing, rapidly evolving with continuous product innovation, geographic expansion, and strategic collaboration.
Key Diagnostic Reagents For Infectious Diseases Companies:
The following are the leading companies in the diagnostic reagents for infectious diseases market. These companies collectively hold the largest market share and dictate industry trends.
- F. Hoffmann-La Roche Ltd
- Abbott
- QIAGEN
- bioMérieux
- Bio-Rad Laboratories, Inc.
- Seegene Inc.
- Thermo Fisher Scientific, Inc.
- SD Biosensor
- Mindray
- Boditech Med Inc.
Recent Developments
-
In July 2025, Bio-Rad Laboratories, Inc. (U.S.) announced the availability of four new Droplet Digital PCR (ddPCR) platforms following the acquisition of digital PCR developer Stilla Technologies (France). With this launch, it has expanded its array of instruments and reagents to provide customers with unrivaled workflow simplicity and throughput capabilities across the whole spectrum of digital PCR applications, including infectious disease.
-
In April 2023, A. Menarini Diagnostics S.r.l. entered into a distribution agreement for the AFIAS-10 System and IGRA-TB Assay, including reagents of Boditech Med Inc., in Italy. This agreement gives Menarini exclusive distribution rights in 34 European countries for the integrated sample-to-answer system AFIAS-10, a fully automated diagnostic platform capable of performing ten different tests independently in parallel.
-
In February 2023, ELITechGroup MDx LLC (US) introduced its Candida product line, which includes reagents for detecting C. auris, identifying fluconazole resistance, and multiplex detection of Candida pathogens. This suite of real-time PCR products features analyte-specific reagents (ASR) and research uses only (RUO) materials for clinical and research laboratories. The latest additions to the EGMDx portfolio are: DSQ Alert Candida auris primer and probe ASRs for specific detection of C. auris, MGB Alert Candida auris with fluconazole resistance RUO Detection Reagent, and MGB Alert Candida species with C. auris RUO Detection Reagent for multiplex detection and discrimination of seven Candida species.
-
In September 2021, F. Hoffmann-La Roche Ltd acquired 100% of the outstanding shares of the TIB Molbiol Group, which has specialized in supplying reagents for research and medical diagnostics, including infectious diseases, for over 30 years globally.
Diagnostic Reagents For Infectious Diseases Market Report Scope
Report Attribute
Details
Market size in 2025
USD 3,967.42 million
Revenue forecast in 2033
USD 8,079.07 million
Growth rate
CAGR of 9.30% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, disease area, technology, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd; Abbott; QIAGEN; bioMérieux; Bio-Rad Laboratories, Inc.; Seegene Inc.; Thermo Fisher Scientific, Inc.; SD Biosensor; Mindray; Boditech Med Inc.
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Diagnostic Reagents For Infectious Diseases Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global diagnostic reagents for infectious diseases market report based on product, disease area, technology, end-use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Molecular Reagents
-
Immunoassay Reagents
-
Rapid Test Reagents
-
Serology Reagents
-
Controls and Calibrators
-
Other Reagents
-
-
Disease Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Respiratory Infections
-
Vector-Borne & Tropical Diseases
-
Enteric Infections
-
Sexually Transmitted Infections (STIs)
-
Other Emerging / High-Burden Infectious Diseases
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Molecular Diagnostics
-
Immunoassays
-
Point-of-Care Reagents
-
Multiplex Platforms
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinics
-
Diagnostic Laboratories
-
Point-of-Care / Decentralized Testing Sites
-
Public Health Agencies & National Reference Labs
-
Academic & Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global diagnostic reagents for infectious diseases market size was estimated at USD 3,475.31 million in 2024 and is expected to reach USD 3,967.42 million in 2025.
b. The global diagnostic reagents for infectious diseases market is expected to grow at a compound annual growth rate of 9.30% from 2025 to 2033 to reach USD 8,079.07 million by 2033.
b. The diagnostic reagents for infectious diseases market in North America held the largest share in 2024, driven by several key factors including a well-established healthcare infrastructure and the strong presence of leading industry players such as Abbott (U.S.), Thermo Fisher Scientific (U.S.), and Becton Dickinson (U.S.).
b. Some key players operating in the diagnostic reagents for infectious diseases market include F. Hoffmann-La Roche Ltd, Abbott, QIAGEN, bioMérieux, Bio-Rad Laboratories, Inc., Seegene Inc., Thermo Fisher Scientific, Inc., SD Biosensor, Mindray, Boditech Med Inc.
b. Key factors that are driving the diagnostic reagents for infectious diseases market growth include the increasing prevalence of respiratory diseases and sexually transmitted infections (STIs). Respiratory diseases, including influenza, COVID-19, pneumonia, and tuberculosis, have the highest prevalence worldwide.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










