- Home
- »
- Medical Devices
- »
-
Diagnostics Device CDMO Market, Industry Report, 2033GVR Report cover
![Diagnostics Device CDMO Market Size, Share & Trends Report]()
Diagnostics Device CDMO Market (2025 - 2033) Size, Share & Trends Analysis Report By Service (Contract Development, Contract Manufacturing), By Class (Class I, Class II), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-833-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Diagnostics Device CDMO Market Summary
The global diagnostics device CDMO market size was estimated at USD 40.33 billion in 2024 and is projected to reach USD 113.12 billion by 2033, growing at a CAGR of 12.43% from 2025 to 2033. The market is driven by the rising burden of chronic & infectious diseases, shift toward point-of-care and rapid diagnostics, cost efficiency and faster time-to-market, and technological advancements in manufacturing.
Key Market Trends & Insights
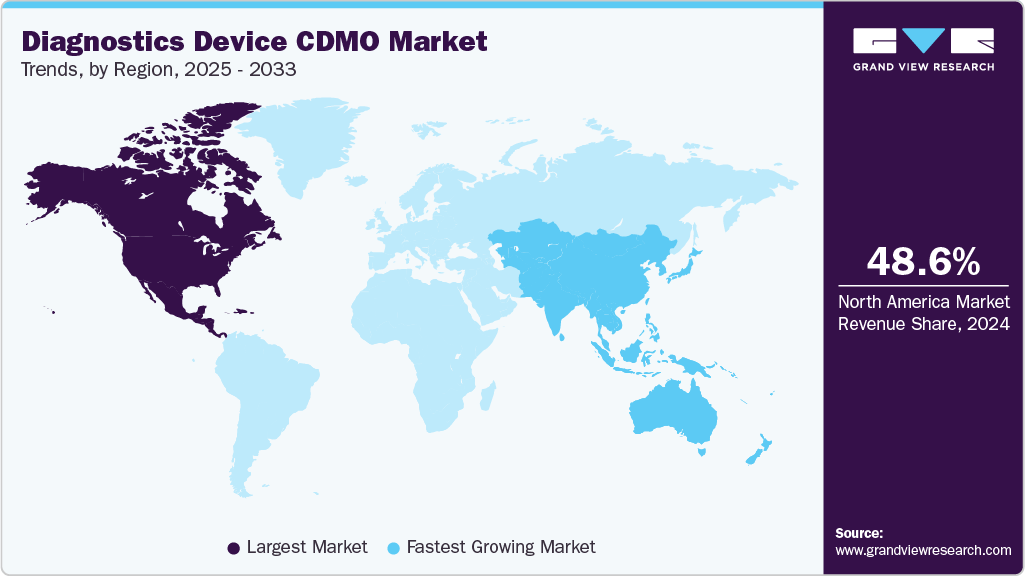
- North America diagnostics device CDMO industry held the largest share of 48.59% in the global market in 2024.
- The diagnostics device CDMO industry in the U.S. is expected to grow significantly over the forecast period.
- By service, the contract manufacturing segment led the market with the largest revenue share of 49.65% in 2024.
- Based on class, the Class II segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 40.33 Billion
- 2033 Projected Market Size: USD 113.12 Billion
- CAGR (2025-2033): 12.43%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
In addition, the market is driven by the healthcare systems shifting toward faster, more accessible, and technology-driven diagnostic solutions. The rising prevalence of chronic and infectious diseases has compelled test developers to accelerate innovation, which has strengthened the need for specialized outsourcing partners. As diagnostic companies aim to shorten development timelines and navigate an increasingly complex regulatory landscape for device design, validation, and commercial-scale manufacturing, they are increasingly relying on CDMOs to manage production, quality assurance, and compliance activities that would otherwise slow in-house operations. This shift is further supported by the rapid move toward decentralized and point-of-care testing, where manufacturers depend on CDMOs capable of delivering compact, high-precision components and fully integrated platforms that meet stringent global quality standards.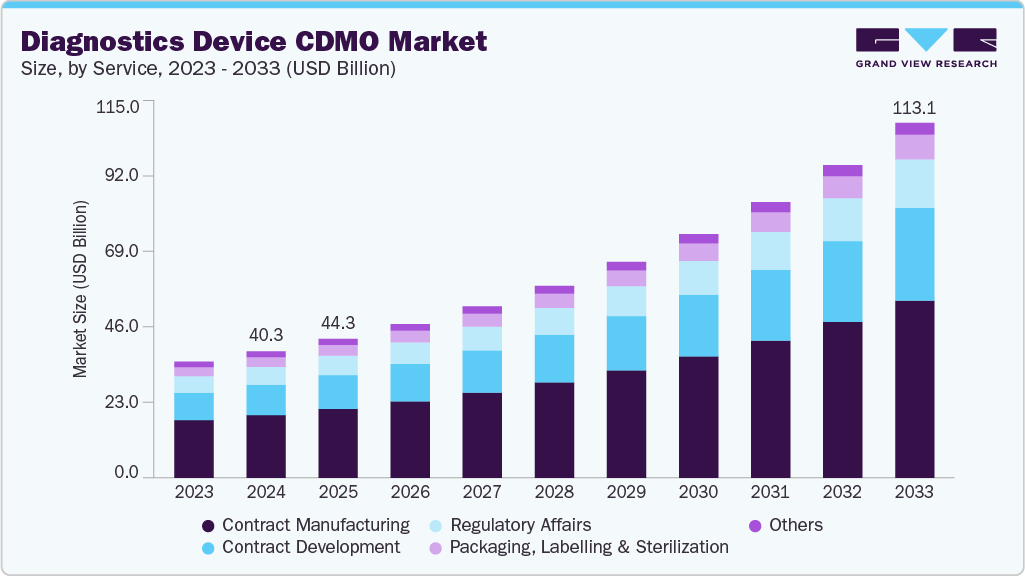
Furthermore, evolving clinical needs have driven technological advancements within the manufacturing ecosystem, further strengthening the role of CDMOs in the diagnostics industry. Advancements in microfluidics, sensor integration, automation, and high-precision plastics have made outsourcing an efficient way for OEMs to access specialized infrastructure without significant capital investment. As companies focus more heavily on R&D, companion diagnostics, and molecular testing, they increasingly depend on CDMOs to manage complex assembly processes, reagent-device integration, and large-volume production. The growth of at-home test kits and rapid diagnostics has further reinforced this trend, with market participants turning to partners capable of scaling output while maintaining high levels of accuracy, reliability, and regulatory compliance.
Service Insights
Based on service, the market is classified into contract manufacturing and contract development, packaging, labelling & sterilization, regulatory affairs, and others. The contract manufacturing segment accounted for the largest revenue share in the market with 49.65% in 2024. The segment's growth is due to the increasing demand for large-scale, high-precision production of diagnostic devices, reagents, and components. Diagnostic companies are increasingly outsourcing manufacturing to CDMOs to reduce capital expenditure, ensure consistent quality, and accelerate time-to-market.
The contract development segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven by the rising need for end-to-end development services, including prototype design, process optimization, analytical testing, and clinical validation. Diagnostic companies are increasingly outsourcing these activities to CDMOs to accelerate product innovation, reduce R&D costs, and manage operational risks.
Class Insights
Based on class segment, the market is segregated into Class I, Class II, and Class III. The Class II segment held the largest share in the market in 2024. The growth of the segment is due to the high demand for moderately complex diagnostic devices that require regulatory oversight but are not as stringent as Class III devices. Class II devices, which include blood glucose monitors, immunoassay analyzers, and certain molecular diagnostic kits, are widely used across hospitals, clinics, and point-of-care settings. Outsourcing the manufacturing and development of these devices to CDMOs allows diagnostic companies to leverage specialized expertise, maintain compliance with FDA and ISO standards, and scale production efficiently to meet growing clinical demand.
Class I segment is anticipated to grow at a considerable CAGR during the forecast period. The growth of the segment is due to the rising demand for low-risk, simple diagnostic devices, such as basic laboratory instruments, sample collection devices, and handheld testing kits, which are widely used in clinics and home settings. Companies are increasingly outsourcing the manufacturing of these devices to CDMOs to benefit from cost efficiency, streamlined production, and regulatory support, while ensuring consistent quality and faster market entry.
Application Insights
In terms of application, the market is segregated into infectious disease diagnostics, oncology diagnostics, cardiometabolic diagnostics, genetic & genomic testing, respiratory & critical care diagnostics, and others segments. The infectious disease diagnostics segment held the largest share in the market in 2024. The growth of the segment is due to the increasing prevalence of infectious diseases, including viral, bacterial, and parasitic infections, coupled with the rising demand for rapid and accurate diagnostic solutions. Frequent outbreaks, such as influenza, COVID-19, and other emerging pathogens, have intensified the need for large-scale production of diagnostic kits and devices, leading companies to rely on CDMOs for manufacturing and development expertise.
The genetic & genomic testing segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is due to the increasing adoption of precision medicine, personalized healthcare, and advanced molecular diagnostics, which require specialized and high-complexity testing. Rising awareness of hereditary diseases, advancements in next-generation sequencing (NGS) technologies, and the growing demand for companion diagnostics are fueling the need for outsourced development and manufacturing services from CDMOs.
End Use Insights
In terms of end use, the market is segregated into original equipment manufacturers (OEMs), pharmaceutical & biopharmaceutical companies, and others segments. The Original Equipment Manufacturers (OEMs) segment accounted for the largest revenue share in 2024 due to the increasing reliance of OEMs on CDMOs for outsourcing non-core activities such as device manufacturing, assembly, quality testing, and regulatory compliance. By partnering with CDMOs, OEMs can reduce capital expenditure, accelerate time-to-market, and maintain focus on R&D, product innovation, and commercialization.
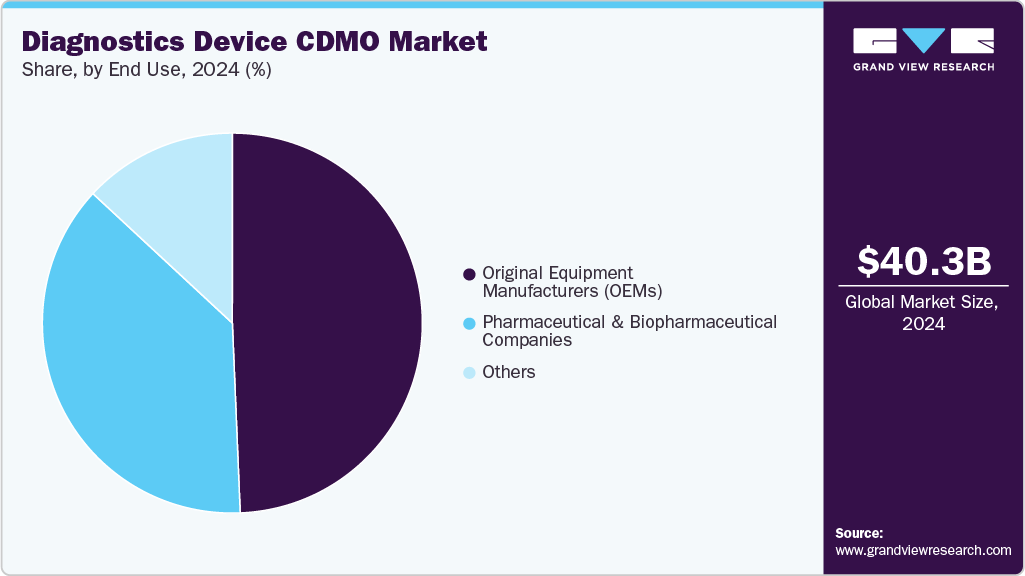
The pharmaceutical & biopharmaceutical companies segment is anticipated to grow at a considerable CAGR during the forecast period. This growth is due to the increasing involvement of these companies in developing advanced diagnostic solutions, including companion diagnostics, molecular testing kits, and personalized medicine platforms. Outsourcing to CDMOs enables pharmaceutical and biopharmaceutical companies to leverage specialized expertise in device development, manufacturing, and regulatory compliance while minimizing capital investment and operational complexity.
Regional Insights
North America Diagnostics Device CDMO Market Trends
North America diagnostics device CDMO industry accounted for the largest revenue share of 48.59% in 2024. The market is driven by a strong regulatory framework, advanced R&D infrastructure & rising demand for innovative healthcare technologies. Most medtech companies are increasingly outsourcing design, development, and manufacturing to optimize costs, accelerate time-to-market, and access specialized expertise.
U.S. Diagnostics Device CDMO Market Trends
The diagnostics device CDMO industry in the U.S. held the largest share in 2024. The country’s growth is due to the rising demand for advanced medical technologies, cost optimization needs, and stringent regulatory requirements. In addition, OEMs in the country are increasingly outsourcing medical device design, prototyping, component production, and assembly to CDMOs to enhance efficiency & reduce time-to-market.
Europe Diagnostics Device CDMO Market Trends
The diagnostics device CDMO industry in Europe is expected to grow significantly due to increasing investments in biotechnology and pharmaceutical R&D, supportive government initiatives promoting advanced therapies, and a strong focus on personalized medicine across the region.
The diagnostics device CDMO industry in Germany held a significant share in 2024, owing to the country’s strong pharmaceutical and biotechnology industry, advanced manufacturing infrastructure, and robust investment in research and development. Germany’s supportive regulatory framework, skilled workforce, and strategic location within Europe make it an attractive hub for CDMO services.
The diagnostics device CDMO industry in the UK held a significant share in 2024. The growth of the market is due to country’s strong biopharmaceutical sector, substantial investments in cutting-edge research, and government initiatives supporting innovation in advanced therapies.
Asia Pacific Diagnostics Device CDMO Market Trends
Asia Pacific diagnostics device CDMO industry is anticipated to witness the fastest CAGR over the estimated timeline. The regional growth is due to the region’s rapidly growing pharmaceutical and biotechnology sectors, cost-effective manufacturing capabilities, and increasing investments in R&D infrastructure. In addition, rising demand for personalized medicine and biologics, supportive government policies, and the presence of emerging CDMOs with advanced technological capabilities are driving market expansion.
The diagnostics device CDMO industry in China held the largest share in 2024. The growth is due to the country’s rapidly expanding pharmaceutical and biotechnology industries, significant government support for innovation, and increasing investments in advanced manufacturing infrastructure. China’s large patient population, cost-efficient production capabilities, and growing number of clinical trials further boost demand for outsourcing services.
The diagnostics device CDMO industry in Japan is expected to grow over the forecast period. The country’s growth is due to the country’s strong pharmaceutical industry, advanced technological capabilities, and significant investments in biopharmaceutical R&D.
The diagnostics device CDMO industry in India is anticipated to grow at a lucrative CAGR over the forecast period. The country’s market growth is due to the lower operational costs, skilled scientific workforce, and growing R&D investments from both domestic and multinational companies.
Key Diagnostics Device CDMO Company Insights
The major players operating across the market are focused on adopting inorganic strategic initiatives such as mergers, partnerships, acquisitions, etc. Moreover, companies focus on technological innovations to augment their market position.
Key Diagnostics Device CDMO Companies:
The following are the leading companies in the diagnostics device CDMO market. These companies collectively hold the largest market share and dictate industry trends.
- Jabil Inc.
- Thermo Fisher Scientific Inc.
- Integer Holdings Corporation
- FLEX Ltd.
- Sanmina Corporation
- Celestica Inc.
- Phillips-Medisize
- Plexus Corp.
- Nipro Corporation
- Viant Technology LLC
Recent Developments
-
In February 2025, Jabil Inc. acquired Pharmaceutics International, Inc. a CDMO with expertise in aseptic filling, lyophilization, and oral solid dose manufacturing - expanding Jabil’s CDMO‑service offerings.
-
In March 2025, Flex Ltd. announce the launch of a new Product Introduction (NPI) center near Boston aimed at serving healthcare customers - including diagnostics and medical‑device firms - from concept through to production transfer.
Diagnostics Device CDMO Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 44.30 billion
Revenue forecast in 2033
USD 113.12 billion
Growth rate
CAGR of 12.43% from 2025 to 2033
Actual data
2018 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Service, class, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE; Oman; Qatar
Key companies profiled
Jabil Inc.; Thermo Fisher Scientific Inc.; Integer Holdings Corporation; FLEX Ltd.; Sanmina Corporation; Celestica Inc.; Phillips-Medisize; Plexus Corp.; Nipro Corporation; Viant Technology LLC
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Diagnostics Device CDMO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global diagnostics device CDMO market report based on service, class, application, end use, and region.
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Manufacturing
-
Accessories Manufacturing
-
Assembly Manufacturing
-
Component Manufacturing
-
Device Manufacturing
-
-
Contract Development
-
Product Design and Development Services
-
Testing & Validation
-
Quality Management
-
Others
-
-
Packaging, Labelling & Sterilization
-
Regulatory Affairs
-
Others
-
-
Class Outlook (Revenue, USD Million, 2021 - 2033)
-
Class I
-
Class II
-
Class III
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Infectious Disease Diagnostics
-
Oncology Diagnostics
-
Cardiometabolic Diagnostics
-
Genetic & Genomic Testing
-
Respiratory & Critical Care Diagnostics
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Original Equipment Manufacturers (OEMs)
-
Pharmaceutical & Biopharmaceutical Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global diagnostics device CDMO market size was estimated at USD 40.33 billion in 2024 and is expected to reach USD 44.30 billion in 2025.
b. The global diagnostics device CDMO market is expected to grow at a compound annual growth rate of 12.43% from 2025 to 2033 to reach USD 113.12 billion by 2033.
b. North America dominated the diagnostics device CDMO market with a share of 48.59% in 2019. This is attributable its strong regulatory framework, advanced R&D infrastructure & rising demand for innovative healthcare technologies. Most medtech companies are increasingly outsourcing design, development, and manufacturing to optimize costs, accelerate time-to-market, and access specialized expertise.
b. Some key players operating in the diagnostics device CDMO market include Jabil Inc., Thermo Fisher Scientific Inc., Integer Holdings Corporation, FLEX Ltd., Sanmina Corporation, Celestica Inc., Phillips-Medisize, Plexus Corp., Nipro Corporation, Viant Technology LLC.
b. Key factors that are driving the market growth include rising burden of chronic & infectious diseases, shift towards point-of-care and rapid diagnostics, cost efficiency and faster time-to-market, and technological advancements in manufacturing.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










