- Home
- »
- Pharmaceuticals
- »
-
Drug Discovery Informatics Market, Industry Report, 2030GVR Report cover
![Drug Discovery Informatics Market Size, Share & Trends Report]()
Drug Discovery Informatics Market (2025 - 2030) Size, Share & Trends Analysis Report By Service (Molecular Modelling), By Application (Discovery Informatics), By End-use (Academic & Research Institutes), By Region, And Segment Forecasts
- Report ID: 978-1-68038-749-0
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Drug Discovery Informatics Market Summary
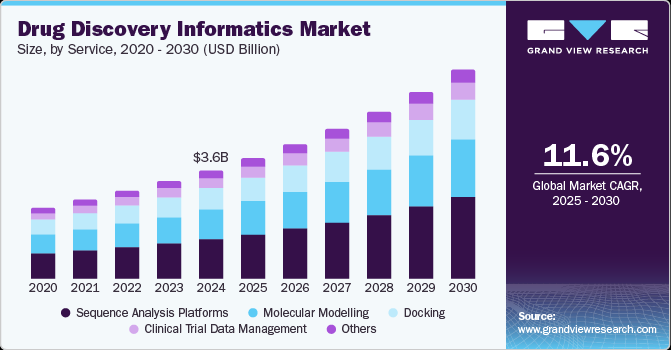
The global drug discovery informatics market size was estimated at USD 3,650.0 million in 2024 and is projected to reach USD 7,030.0 million by 2030, growing at a CAGR of 11.6% from 2025 to 2030. This can be attributed to various factors, including advancements in high-performance computing and web services, increased adoption of in-silico modeling tools, the growing volume of data in drug discovery, and rising demand for innovative drug molecules.
Key Market Trends & Insights
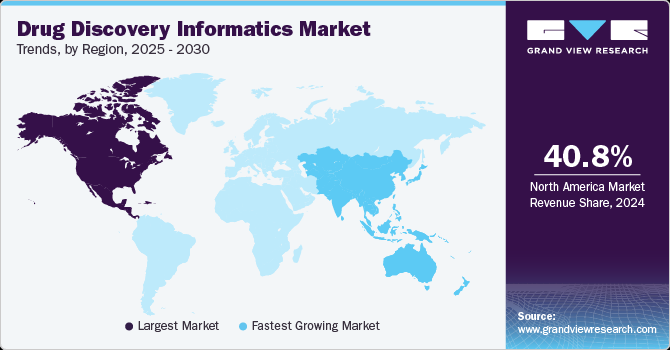
- In terms of region, North America was the largest revenue generating market in 2024.
- Country-wise, China is expected to register the highest CAGR from 2025 to 2030.
- In terms of service, sequence analysis platforms accounted for a revenue share of 36.4% in 2024.
- The molecular modeling segment is expected to grow at a CAGR of 11.8% over the forecast period.
Market Size & Forecast
- 2024 Market Size: USD 3,650.0 Million
- 2030 Projected Market Size: USD 7,030.0 Million
- CAGR (2025-2030): 11.6%
- North America: Largest market in 2024
Furthermore, introducing advanced information technology solutions for drug discovery is expected to intensify industry competition. In January 2021, Certara, Inc. unveiled the latest version of its Simcyp Physiologically-based Pharmacokinetic (PBPK) Simulator platform. This new platform incorporates models that assess and analyze drug actions during pregnancy and lactation.
Moreover, businesses are increasingly preferring to embrace informatics solutions to update and advance the drug discovery procedure, thereby fueling the market's growth. The market is poised for substantial growth in the coming years, driven by advancements in high-performance computing and web services, the increasing adoption of in-silico modeling tools, a growing volume of drug discovery data, and rising demand for innovative drug molecules. This growth is further fueled by the launch of advanced information technology solutions in drug discovery, leading to intensified competition among industry players. For example, Certara, Inc. introduced the Simcyp Physiologically-based Pharmacokinetic (PBPK) Simulator version 20, which provides new models for assessing drug action during pregnancy and lactation, aiding in critical decisions related to optimizing medicine safety and efficacy profiles.
Drug discovery informatics refers to the application of computational tools and methods to enhance the process of identifying and developing new drugs. This field leverages advanced technologies to streamline drug discovery, making it more efficient and cost-effective. Artificial intelligence (AI) is revolutionizing the pharmaceutical industry by transforming various aspects of drug discovery and development. AI algorithms play a crucial role in optimizing experimental designs and parameter selections, thereby reducing the time and resources needed for both preclinical and clinical studies. There is a growing trend among companies to adopt informatics solutions to streamline and shorten the drug discovery timeline. Curve Therapeutics Ltd. implemented the CDD Vault platform to manage its genetically encoded Microcycle platform. This hosted platform offers secure data management and seamless real-time data sharing, facilitating collaborative discoveries across the pharmaceutical, biotechnology, and academic sectors.
In addition, the integration of AI with cloud computing and advanced analytics presents numerous opportunities for growth in the drug discovery informatics market. Cloud-based technologies offer low-cost data storage and processing capabilities, enabling the analysis of larger datasets. This integration enhances the drug discovery process by making it more cost-effective. AI also enhances clinical trial design by optimizing participant recruitment and predicting potential side effects, leading to more targeted and efficient trials.
As AI continues to evolve, it is poised to further transform the pharmaceutical sector. Its applications in drug design, polypharmacology, and drug repurposing are particularly promising. AI-driven technologies are expediting target identification and facilitating drug design, which are critical components of the drug development process. Moreover, AI aids in patient stratification and drug combination analysis, allowing for more tailored treatment strategies.
Service Insights
The sequence analysis platforms dominated the market and accounted for the largest revenue share of 36.4% in 2024. Bioinformatics solutions are widely employed for the development of primary and secondary databases of nucleic acids, proteins, and other biomolecule sequences. In addition, informatics software enables the mining and warehousing of genome sequencing data to identify genes and targeted proteins, thereby facilitating the development of potential drugs. Furthermore, different databases and analysis techniques are employed in basic drug discovery research, with sequence analysis tools like CLUSTALW, BLAST, and FASTA playing a crucial role. Moreover, the launch of new and advanced software in this field contributes to the growth of the segment.
Informatics platforms also play a significant role in molecular modeling, which is increasingly popular in the drug discovery process. These tools enable the modeling and simulation of biological systems and small molecules, providing insights into their behavior at a molecular level. Additionally, they contribute to the cost-efficiency of hit discovery and hit-to-lead optimization, accelerating the drug development process. The molecular modeling segment is expected to grow at a CAGR of 11.8% over the forecast period. The integration of AI and machine learning with molecular modeling tools accelerates drug discovery processes, making them more precise and cost-effective. This integration also fosters innovation in drug design and development.
Application Insights
The discovery informatics led the global drug discovery informatics industry and accounted for the largest revenue share of 60.2% in 2024, with a major focus on developing precision medicines. Precision medicine revolves around providing personalized medical care tailored to an individual's genetic and molecular profile. Companies such as PerkinElmer, Inc. offer innovative solutions utilizing analytics to accelerate drug discovery. Furthermore, in several developing countries, governments are actively encouraging the adoption of bioinformatics to expedite drug discovery processes.
Bio content management is anticipated to grow at a CAGR of 10.9% over the forecast period, driven by the increasing need for robust data storage and retrieval systems. This demand arises from managing large datasets, including gene sequences and clinical trial information. Furthermore, pharmaceutical companies and research institutions are focusing on collaborative and data-accessible solutions to streamline drug discovery workflows. Moreover, the integration of advanced IT applications and AI enhances data management efficiency, fostering market growth.
End Use Insights
Pharmaceutical and biotechnology companies held the dominant position in the market, with the largest revenue share of 48.4% in 2024. This growth is attributed to increased R&D spending, particularly in precision medicine. In addition, informatics solutions streamline drug development processes, enhance efficiency, and reduce costs. Furthermore, technological advancements and strategic collaborations fuel this growth by facilitating data-driven drug development and accelerating innovation.
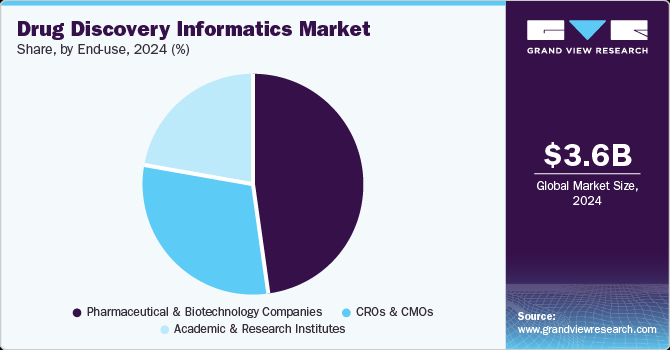
Academic and research institutes are expected to grow at the fastest CAGR of 12.9% from 2025 to 2030, owing to the need for efficient data management and analysis tools. Informatics solutions enhance collaboration among researchers, streamline workflows, and accelerate discovery processes. Furthermore, increased funding for research projects and the integration of AI and machine learning technologies supports this growth.
Regional Insights
North America drug discovery informatics market dominated the global market with a revenue share of 40.8% in 2024, primarily driven by the increasing investments in research and development (R&D) activities focused on drug discovery and the advancements in data mining and analytics technologies. These developments have significantly contributed to the expansion of the market by enabling more efficient and effective analysis of large volumes of data in the drug discovery process. By leveraging advanced informatics tools and techniques, researchers and pharmaceutical companies can extract valuable insights and make informed decisions, leading to accelerated drug discovery and development efforts.
U.S. Drug Discovery Informatics Market Trends
The growth of the drug discovery informatics market in the U.S. is driven by significant investments in R&D, particularly in precision medicine and rare disease research. In addition, advanced data mining and analytics technologies enhance the efficiency of drug discovery processes, allowing researchers to make informed decisions.
Asia Pacific Drug Discovery Informatics Market Trends
Asia Pacific drug discovery informatics market is expected to grow at the fastest CAGR of 12.3% during the forecast period. This is owing to the rapid growth of pharmaceutical and biopharmaceutical industries and the expanding number of CROs in the region. China and India are the major countries offering promising growth opportunities over the forecast period. Furthermore, strategic partnerships between local companies and international players enhance technological capabilities, contributing to the region's high growth rate. For instance, In November 2024, Schrödinger and Novartis formed a partnership to enhance drug discovery using computational tools. Novartis paid USD 150 million upfront and could pay up to USD 2.3 billion in milestones and royalties. The collaboration aimed to boost research efficiency across Novartis's therapeutic areas.
Europe Drug Discovery Informatics Market Trends
Europe drug discovery informatics is expected to witness substantial CAGR over the forecast period, driven by increased R&D spending and the integration of AI and machine learning technologies. Pharmaceutical companies leverage these advancements to accelerate drug development timelines and improve precision in drug targeting.
Key Drug Discovery Informatics Company Insights
Major players in the global drug discovery informatics industry include Certara, Infosys Ltd., Charles River Laboratories, and others. To speed up drug development, key players in the industry are actively collaborating with other companies, research institutes, and clinical research organizations. These partnerships aim to enhance efficiency and accelerate the delivery of valuable data.
-
Certara offers a range of products, including D360, which accelerates drug discovery by streamlining the design-make-test-analyzing cycle for small molecules and biologics.
-
Eurofins DiscoverX specializes in cell-based and biochemical assays for drug discovery, offering a comprehensive suite of products that support early-stage research and development. Its solutions help researchers identify and validate drug targets, optimize lead compounds, and assess drug efficacy and safety.
Key Drug Discovery Informatics Companies:
The following are the leading companies in the drug discovery informatics market. These companies collectively hold the largest market share and dictate industry trends.
- Certara
- Boehringer Ingelheim International GmbH
- Infosys Ltd.
- Charles River Laboratories
- Collaborative Drug Discovery, Inc.
- Eurofins DiscoverX Products
- Jubliant Biosys
- Selvita
- Novo Informatics Pvt. Ltd.
- ChemAxon Ltd.
- Albany Molecular Research Inc.
- Oracle
- Accenture
- Agilent Technologies, Inc.
- Illumina, Inc.
Recent Developments
-
In January 2025, Charles River Laboratories expanded its Apollo ecosystem to offer bespoke client experiences, enhancing access to services and visibility around teams & processes.
-
In October 2024, Certara, a leader in model-informed drug development, finalized its acquisition of Chemaxon, a scientific informatics company, to enhance drug discovery and development.
Drug Discovery Informatics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 4.06 billion
Revenue forecast in 2030
USD 7.03 billion
Growth rate
CAGR of 11.6% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Service, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S., Canada, Mexico, Germany, UK, France, Italy, Spain, Denmark, Sweden, Norway, China, Japan, India, Australia, Thailand, South Korea, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait
Key companies profiled
Certara; Boehringer Ingelheim International GmbH; Infosys Ltd.; Charles River Laboratories; Collaborative Drug Discovery, Inc.; Eurofins DiscoverX Products; Jubliant Biosys; Selvita; Novo Informatics Pvt. Ltd.; ChemAxon Ltd.; Albany Molecular Research Inc.; Oracle; Accenture; Agilent Technologies, Inc.; Illumina, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Drug Discovery Informatics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global drug discovery informatics market report based on service, application, end use, and region:
-
Service Outlook (Revenue, USD Million, 2018 - 2030)
-
Sequence Analysis Platforms
-
Molecular Modelling
-
Docking
-
Clinical Trial Data Management
-
Others
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Discovery Informatics
-
Biocontent Management
-
-
End Use Channel Outlook (Revenue, USD Million, 2018 - 2030)
-
Pharmaceutical & Biotechnology Companies
-
CROs & CMOs
-
Academic & Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









