- Home
- »
- Medical Devices
- »
-
Drug Eluting Stent Market Size, Share, Industry Report 2033GVR Report cover
![Drug Eluting Stent Market Size, Share & Trends Report]()
Drug Eluting Stent Market (2026 - 2033) Size, Share & Trends Analysis Report By Type of Coating (Polymer-based Coating, Polymer-free Coating), By End Use (Hospitals, Specialty Cardiac/Vascular Centers, Ambulatory Surgical Centers), By Region, And Segment Forecasts
- Report ID: 978-1-68038-999-9
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Drug Eluting Stent Market Size & Trends
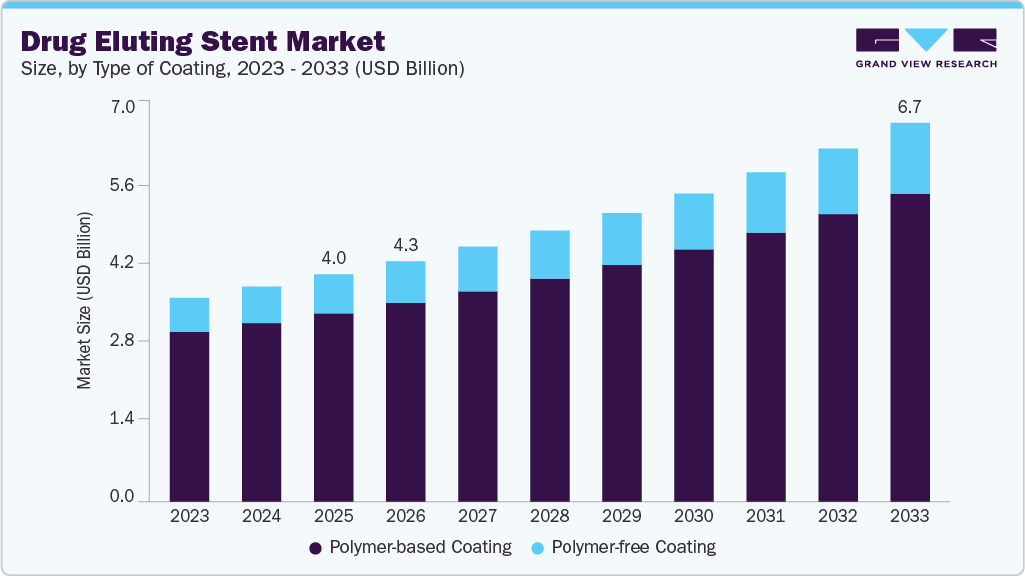
The global drug eluting stent market size was estimated at USD 4.0 billion in 2025 and is projected to reach USD 6.7 billion by 2033, growing at a CAGR of 6.7% from 2026 to 2033. Market growth is primarily driven by the rising CVD prevalence, fueled by obesity, hypertension, diabetes, aging populations, sedentary lifestyles, and unhealthy diets. In October 2024, the CDC reported 919,032 deaths in the U.S. from cardiovascular disease in 2023, with coronary heart disease causing 371,506 deaths. About 1 in 6 CVD deaths occurred in adults under 65, and approximately 805,000 heart attacks happened nationwide, including 605,000 first-time events.
Key Market Trends & Insights
- The North America drug eluting stents market dominated the global market in 2025 with a revenue share of 44.1%.
- The U.S. drug eluting stents market accounted for the largest share in North America in 2025.
- Based on the type of coating, the polymer-based coating segment held the largest share in 2025.
- Based on end use, the hospitals segment held the largest share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.0 Billion
- 2033 Projected Market Size: USD 6.7 Billion
- CAGR (2026-2033): 6.7%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Rising Prevalence of Cardiovascular Diseases and Associated Risk Factors
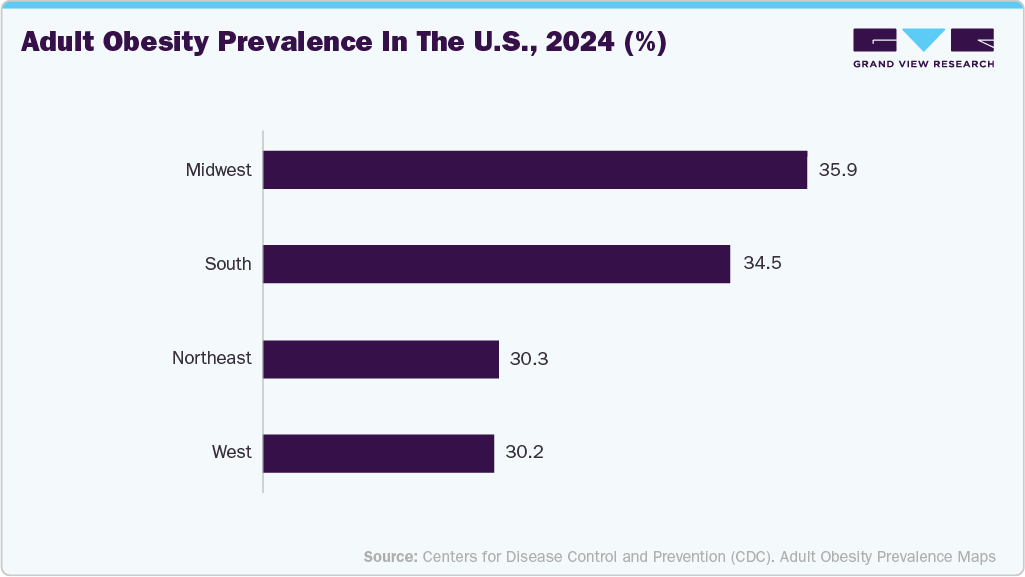
The rising prevalence of cardiovascular diseases (CVDs) is a primary driver of the market. Increasing cases of coronary artery disease, heart failure, and peripheral artery disease are largely driven by lifestyle-related risk factors such as obesity, diabetes, hypertension, and sedentary behavior. As the global population ages, the demand for interventional procedures such as stenting, angioplasty, and catheter-based therapies continues to grow. In December 2025, the CDC released the 2024 adult obesity prevalence data for all U.S. states and territories. Obesity, defined as a BMI ≥30 kg/m², increases the risk of chronic diseases such as heart disease, diabetes, and certain cancers. Rates ranged from 25% to over 40%, highlighting the urgent need for prevention and treatment strategies across the population.
Increasing Volume of Percutaneous Coronary Intervention (PCI) Procedures
PCI is widely used to treat coronary artery disease and restore blood flow in narrowed or blocked coronary arteries. The increasing prevalence of cardiovascular diseases, including coronary artery disease and acute coronary syndromes, is raising the number of patients requiring interventional treatment. Higher incidence of risk factors such as hypertension, diabetes, and obesity further contributes to procedural demand. In May 2025, the SCAI Scientific Sessions reported that 408,060 outpatient PCI procedures (2020-2022) were analyzed, with 1.8% performed in ambulatory surgery centers and 98.2% in hospital outpatient departments. PCI use in ASCs rose to 0.87 per 10,000 person‑years, and 30‑day adverse events, including mortality, stroke, and bleeding, remained below 2%.
Rising Product Innovations and Regulatory Approvals
Manufacturers are investing heavily in next-generation stents, catheters, and balloons to improve procedural outcomes and reduce complications. Innovations such as drug eluting stents with bioresorbable polymers, thinner strut designs, and improved deliverability are enhancing safety and efficacy in percutaneous interventions. In February 2026, a study published in the Indian Heart Journal reported that ultrathin-strut biodegradable polymer everolimus-eluting stents demonstrated clinical outcomes comparable to durable-polymer everolimus-eluting stents in routine percutaneous coronary intervention (PCI). At 24-month follow-up, major adverse cardiac event rates were similar (3.8% vs 3.7%) and there was no definite or probable stent thrombosis, supporting the safety and effectiveness of ultrathin biodegradable polymer stents in real-world practice.
Market Concentration & Characteristics
The market exhibits a high degree of innovation, driven by continuous advancements in stent platform design, ultrathin strut architecture, and next-generation drug-polymer combinations that improve safety and long-term efficacy. For instance, in January 2025, BIOTRONIK revealed expanded indications in Europe for its Orsiro Mission drug eluting stent, including use with 1-month DAPT and treatment of complex calcified lesions, highlighting advancements toward safer and more flexible patient-specific coronary interventions.
The industry is witnessing a moderate level of M&A activity, with companies focusing on expanding their interventional cardiology portfolios and global reach. For instance, in July 2025, Teleflex Incorporated acquired the Vascular Intervention business of BIOTRONIK SE & Co. KG, including the Orsiro Mission drug eluting stent portfolio. This deal strengthens Teleflex’s presence in coronary interventions and enhances its global cardiovascular device offerings.
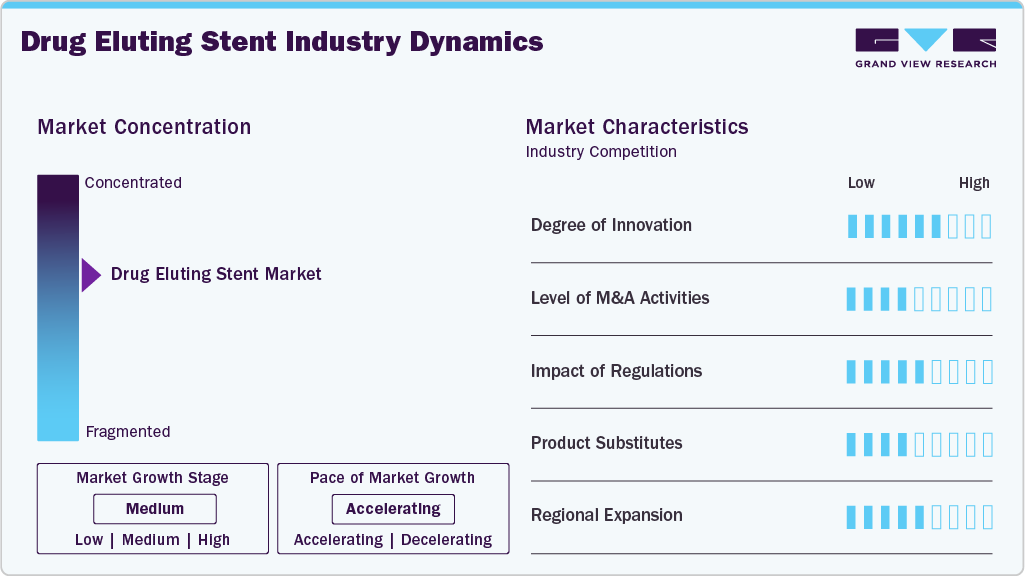
The drug eluting stents market operates in a stringent regulatory environment, requiring approvals such as U.S. FDA premarket approval (PMA) and CE marking under the EU MDR 2017/745 in Europe, along with compliance with evolving global medical device standards. These regulations ensure high safety, clinical efficacy, and long-term performance of DES, but also increase development costs and extend time to market due to extensive clinical trial requirements.
There are limited direct substitutes for DES; however, alternatives such as drug-coated balloons (DCBs) and conventional balloon angioplasty are used in similar settings. DCBs, in particular, may limit DES where a “leave nothing behind” approach is preferred.
The drug eluting stents pipeline product market is witnessing strong regional expansion, particularly across Asia-Pacific, Latin America, and Europe, as companies scale global commercialization and strengthen distribution networks. For instance, in October 2025, MicroPort Scientific Corporation announced that its coronary stent portfolio, including the Firehawk drug eluting stent, has expanded to over 90 countries and 3,000+ hospitals worldwide, reflecting significant penetration in emerging and developed markets.
Type of Coating Insights
The polymer-based coating segment accounted for the largest revenue share of over 82.8% in 2025, driven by its ability to provide controlled and sustained drug release, thereby significantly reducing restenosis rates and improving long-term clinical outcomes in percutaneous coronary interventions. Technologically advanced product launch drives the growth of the market, for instance, in March 2026, Abbott Laboratories revealed the launch of its XIENCE Skypoint drug eluting stent in India, designed with enhanced flexibility and improved deliverability for treating complex coronary artery blockages while utilizing advanced polymer-based coating technology for effective drug release.
The polymer-free coating segment is expected to grow at the fastest CAGR over the forecast period, driven by the need to eliminate polymer-associated complications such as delayed endothelial healing and late adverse events. According to the Elsevier B.V. article in March 2026, a multicenter prospective registry study conducted across 33 centers in India evaluated the ISAR SUMMIT polymer-free everolimus-eluting stent in 500 patients enrolled between August 2022 and September 2023, where the device demonstrated a target lesion failure (TLF) rate of 1.0% at 12 months (5 patients), including cardiovascular death of 0.8%, target-vessel myocardial infarction of 0.4%, and target-lesion revascularization of 0.2%, along with definite or probable stent thrombosis of 0.6% (3 patients).
End Use Insights
By end use, the hospitals segment accounted for the largest revenue share of 68.3% in 2025, driven by the high volume of percutaneous coronary intervention (PCI) procedures performed in hospital settings. The growth is also supported by advanced catheterization laboratories and the presence of skilled interventional cardiologists for managing acute and complex coronary artery disease cases. Supporting this dominance, as per the article NCBI article published in October 2024, about 9,700 patients underwent PCI with drug eluting stent implantation across multiple clinical centers, representing sustained clinical outcomes with continued monitoring up to 10 years, highlighting the extensive utilization of DES in hospital-based settings.
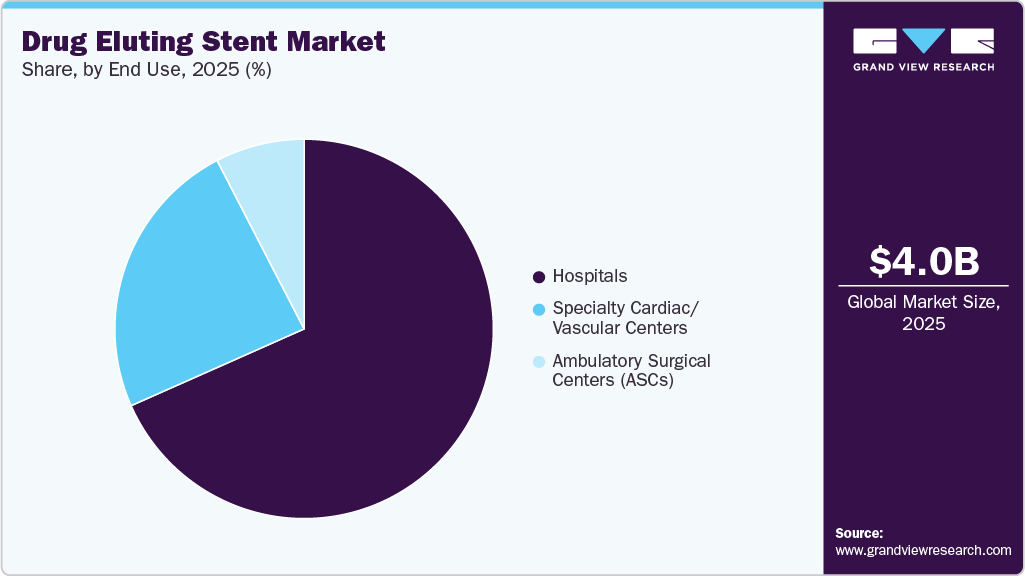
The ambulatory surgical centers segment is expected to grow at the fastest CAGR over the forecast period, driven by the increasing shift of low-risk percutaneous coronary intervention (PCI) procedures toward outpatient settings, supported by cost efficiency, shorter recovery times, and advancements in minimally invasive technologies. The expansion of reimbursement coverage for outpatient PCI procedures has further accelerated adoption, enabling select drug eluting stent (DES) interventions to be safely performed outside traditional hospital environments.
Regional Insights
The North America drug eluting stents industry dominated with a share of 44.1% in 2025. Rising CVD cases, advanced healthcare infrastructure, and high awareness of interventional cardiology treatments, which support the widespread adoption of DES across hospitals and outpatient settings, fuel market growth. Key manufacturers maintain a strong presence, continuously focusing on product innovation and regulatory approvals to strengthen their market position. For instance, in November 2025, the Merck Foundation launched the Collaborative for Equity in Cardiac Care, a USD 22 million, five-year initiative aimed at improving access to timely and high-quality cardiovascular interventions in the U.S.
U.S. Drug Eluting Stent Market Trends
The drug eluting stents industry in the U.S. accounted for the largest market share in North America in 2025. Growing awareness among patients and healthcare providers about early diagnosis and timely intervention is further driving higher procedure volumes across hospitals, specialty cardiac centers, and outpatient facilities. In April 2024, Michigan Medicine reported that over 500,000 Americans undergo PCI annually, highlighting the large addressable patient pool for DES technologies. The study emphasized the need to reduce avoidable mortality, supporting the adoption of advanced device-based interventions.
Europe Drug Eluting Stent Market Trends
The drug eluting stents industry in Europe is expected to grow notably over the forecast period. Rising CVD incidence and adoption of minimally invasive cardiovascular procedures continue to accelerate the demand for interventional cardiology interventions across Europe. Hospitals are integrating advanced technologies to improve lesion assessment and optimize stent deployment. In February 2026, an article published in the European Heart Journal discussed the future of coronary revascularization, highlighting a potential shift from traditional metallic coronary stents toward “metal-less” treatment strategies.
The UK drug eluting stent industry is projected to grow considerably over the forecast period. Growing awareness among patients and clinicians regarding early diagnosis and timely intervention is supporting procedure volumes across NHS hospitals and cardiac centers in the UK, fueling market growth. For instance, a June 2024 study published in the BMJ analyzed cardiovascular trends among 22 million individuals, reporting that while major atherosclerotic conditions such as coronary heart disease declined by ~30%, the cardiovascular burden remained stable due to rising cases of arrhythmias and thromboembolic disorders, thereby sustaining the need for interventional procedures such as PCI and continued adoption of drug eluting stents in clinical practice.
The drug eluting stent industry in Germany is growing over the forecast period, driven by the high prevalence of cardiovascular diseases and strong adoption of interventional cardiology procedures maintained by advanced healthcare infrastructure and specialized cardiac centers. Patients increasingly benefit from minimally invasive procedures such as percutaneous coronary intervention (PCI), contributing to sustained demand for DES. For instance, in December 2025, the Robert Koch Institute reported 119,795 deaths from coronary heart disease (CHD) in 2023, with an age-standardized mortality rate of 120 per 100,000. Mortality was higher in men (172) than women (82.2) and rose sharply with age, reaching 1,476 per 100,000 in those ≥80 years, indicating a large patient pool for PCI procedures and supporting continued DES adoption.
Asia Pacific Drug Eluting Stent Market Trends
The drug eluting stents industry in the Asia-Pacific region has witnessed a growing demand for drug eluting stents, driven by the increasing prevalence of cardiovascular diseases and technological advancements in the region, which fuel market growth. Companies in the Asia Pacific region, particularly in Japan, South Korea, China, and India, have been developing innovative cardiology devices. These include advanced stents, catheters, and balloons, used in procedures such as angioplasty & stent placement. For instance, in March 2026, Abbott launched Xience Skypoint, its latest everolimus-eluting drug eluting stent (DES), in India to address complex coronary blockages, including calcified lesions, long lesions, and small vessels.
The China drug eluting stent industry accounted for the largest market share in the Asia Pacific region in 2025. Continuous investments in cardiovascular infrastructure and physician training, which have strengthened interventional capabilities across major hospitals. The increasing use of minimally invasive procedures for complex lesions, including chronic total occlusions and calcified arteries, is contributing to rising PCI volumes and sustained DES demand, fueling market growth. For instance, in October 2025, MicroPort Scientific Corporation reported cumulative shipments and implantations of over 10 million for its coronary stent systems globally, reflecting strong adoption of DES platforms such as Firehawk Liberty and Firesorb across 90+ countries, with significant hospital penetration in China.
The drug eluting stent industry in India is expected to grow at a significant CAGR over the forecast period, driven by the increasing demand for interventional cardiology devices such as stents and balloons used in angioplasty procedures, along with expanding catheterization laboratory infrastructure across public and private hospitals. Rising awareness, improving access to cardiac care, and a growing burden of coronary artery disease are further supporting procedural volumes and DES adoption. For instance, between May and August 2025, findings published in the Journal of the Practice of Cardiovascular Sciences highlighted a steady rise in interventional cardiology procedures in India, driven by expanding cath lab capacity and increasing adoption of advanced devices, including drug eluting stents.
Latin America Drug Eluting Stent Market Trends
The drug eluting stents industry in Latin America is expected to witness significant growth over the forecast period, driven by the rising incidence of cardiovascular diseases and the increasing adoption of minimally invasive interventional procedures across key countries such as Brazil and Argentina. Expanding access to cardiac care, improving healthcare infrastructure, and growing awareness of early diagnosis and treatment are further supporting market expansion.
The Brazil drug eluting stent industry is expected to witness significant growth over the forecast period. The rising prevalence of cardiovascular diseases, supporting strong demand for interventional cardiology procedures such as percutaneous coronary intervention (PCI), drives market growth. For instance, in June 2025, a study published in The Lancet Regional Health Americas reported that age-standardized cardiovascular mortality in Brazil declined by 31%, with reductions reaching up to 43% in larger municipalities in the South, reflecting improvements in access to cardiovascular care and interventional treatments, thereby supporting continued growth in DES utilization across the country.
Middle East & Africa Drug Eluting Stent Market Trends
The drug eluting stents industry in the Middle East and Africa is growing significantly over the forecast period, driven by the rising burden of cardiovascular diseases and increasing prevalence of risk factors such as obesity, diabetes, and hypertension. Countries including Saudi Arabia, UAE, South Africa, and Kuwait are strengthening cardiac care infrastructure, with hospitals and specialized centers expanding their percutaneous coronary intervention (PCI) capabilities. In addition, key players such as Medtronic, BIOTRONIK, and B. Braun SE are focusing on regional expansion through physician training and clinical support programs, improving procedural expertise and patient access to advanced DES technologies, thereby supporting long-term market growth across the MEA region.
The South Africa drug eluting stent industry is growing significantly over the forecast period. The increasing burden of cardiovascular diseases, which is increasing the demand for interventional cardiology procedures such as percutaneous coronary intervention, is driving market growth. According to a May 2024 NCBI article, cardiovascular disease accounts for ~17.3% of deaths in South Africa, with over 200 deaths per day, driving demand for revascularization procedures and increased adoption of drug eluting stents.
Key Drug Eluting Stent Company Insights
Leading players in the drug eluting stents market, such as Boston Scientific Corporation, B. Braun SE and Medtronic, have strategically employed innovative approaches, including mergers and acquisitions, market penetration initiatives, partnerships, and distribution agreements.
Key Drug Eluting Stent Companies:
The following key companies have been profiled for this study on the drug eluting stent market.
- Boston Scientific Corporation
- Medtronic
- Abbott
- Teleflex Incorporated (BIOTRONIK)
- Terumo Corporation
- B. Braun SE
- Translumina
- MicroPort Scientific Corporation
- Biosensors International Group, Ltd
- Lepu Medical
- iVascular S.L.U.
- SMT
- Meril Life Sciences Pvt. Ltd.
- Concept Medical
- Cardionovum GmbH
Recent Developments
-
In October 2025, Orchestra BioMed initiated enrollment in the Virtue SAB clinical trial, evaluating a sirolimus-based coronary therapy against conventional treatments for in-stent restenosis, highlighting continued advancements in DES-related technologies.
-
In July 2025, Teleflex Incorporated completed the acquisition of substantially all of the vascular intervention business of BIOTRONIK for approximately (€760 Million) USD 895 Million, significantly expanding its presence in interventional cardiology and peripheral vascular markets. The transaction added a broad portfolio of products used in catheterization labs, including drug-coated balloons, drug eluting stents, covered stents, and balloon catheters, strengthening Teleflex’s global cath lab portfolio and market positioning.
-
In November 2024, Abbott Laboratories reported positive long-term clinical outcomes for its Esprit drug eluting scaffold, exhibiting sustained effectiveness in vascular interventions and supporting continued innovation in DES-related platforms.
Drug Eluting Sten Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.3 billion
Revenue forecast in 2033
USD 6.7 billion
Growth rate
CAGR of 6.7% from 2026 to 2033
Actual Data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million, volume in thousand units, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type of coating, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; Indonesia; Malaysia; Vietnam; South Korea; Brazil; Argentina; Colombia; Chile; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Boston Scientific Corporation; Medtronic; Abbott; Teleflex Incorporated (BIOTRONIK); Terumo Corporation; B. Braun SE; Translumina; MicroPort Scientific Corporation; Biosensors International Group, Ltd; Lepu Medical; iVascular S.L.U.; SMT; Meril Life Sciences Pvt. Ltd.; Concept Medical; Cardionovum GmbH
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Drug Eluting Stent Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis of the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global drug eluting stents market report based on type of coating, end use, and region:
-
Type of Coating Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Polymer-based Coating
-
Non-biodegradable Polymers
-
Biodegradable Polymers
-
-
Polymer-free Coating
-
Micro porous Surface
-
Micro structured Surface
-
Slotted tubular Surface
-
Nanoporous Surface
-
-
-
End Use Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Cardiac/Vascular Centers
-
Ambulatory Surgical Centers (ASCs)
-
-
Regional Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
Indonesia
-
Malaysia
-
Vietnam
-
-
Latin America
-
Brazil
-
Argentina
-
Colombia
-
Chile
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the drug eluting stent market include Boston Scientific Corporation; Medtronic; B. Braun SE; Concept Medical; Abbott; Koninklijke Philips N.V.; Teleflex Incorporated (BIOTRONIK); Translumina; MicroPort Scientific Corporation; Cardionovum GmbH; iVascular S.L.U.; Lepu Medical; Meril Life Sciences Pvt. Ltd.; Cordis; Acotec Scientific (Acotec / Acotec Scientific AG); SMT
b. Key factors that are driving the market growth include sedentary lifestyles, unhealthy food habits, and lack of exercise leading to a high risk of developing arterial diseases.
b. The global drug eluting stent market size was estimated at USD 4.0 billion in 2025 and is expected to reach USD 4.3 billion in 2026.
b. The global drug eluting stent market is expected to grow at a compound annual growth rate of 6.7% from 2026 to 2033 to reach USD 6.7 billion by 2033.
b. North America dominated the drug eluting stent market with a share of 44.2% in 2025. This is attributable to supportive reimbursement coverage coupled with a high incidence rate of artery diseases.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










