- Home
- »
- Pharmaceuticals
- »
-
Enclomiphene Market Size And Share, Industry Report, 2033GVR Report cover
![Enclomiphene Market Size, Share & Trends Report]()
Enclomiphene Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Type (Enclomiphene Citrate Tablets), By Dosage Strength (Medium Dose (12.5 mg - 25 mg)), By Indication, By Distribution Channel, By Region (North America, Europe, Asia Pacific, Middle East & Africa), And Segment Forecasts
- Report ID: GVR-4-68040-905-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Enclomiphene Market Summary
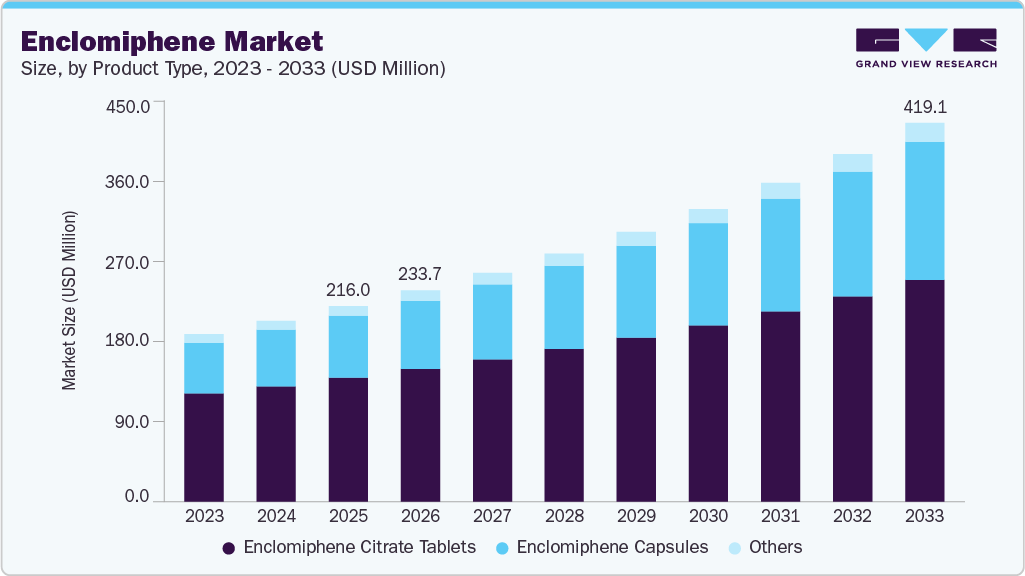
The global enclomiphene market size was estimated at USD 216.00 million in 2025 and is projected to reach USD 419.05 million by 2033, growing at a CAGR of 8.70% from 2026 to 2033. The growth is driven by the increasing prevalence of male hypogonadism and infertility, along with a growing preference for therapies that boost endogenous testosterone while preserving fertility.
Key Market Trends & Insights
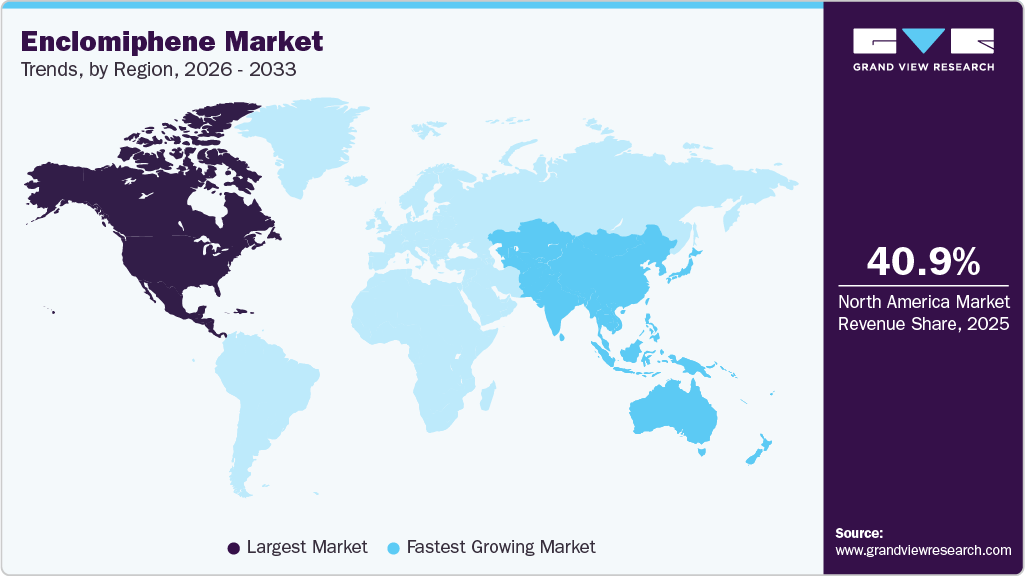
- North America enclomiphene market held the largest share of 40.97% of the global market in 2025
- The enclomiphene industry in the U.S. is expected to grow significantly over the forecast period.
- By product type, the enclomiphene citrate tablets segment held the highest market share of 63.20% in 2025.
- By dosage strength, the medium dose (12.5 mg - 25 mg) segment held the highest market share of 51.31% in 2025.
- By indication, the male hypogonadism segment held the highest market share of 49.10% in 2025.
- By distribution channel, the hospital pharmacies segment held the highest market share of 49.50% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 216.00 Million
- 2033 Projected Market Size: USD 419.05 Million
- CAGR (2026-2033): 8.70%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Rising awareness of men’s health, improved diagnosis rates, and expanding access to fertility and endocrinology care are further supporting demand. In addition, the shift toward selective estrogen receptor modulators over traditional testosterone replacement therapy, coupled with ongoing clinical research, product development, and lifestyle factors such as delayed parenthood and hormonal imbalances, is accelerating market growth, although regulatory constraints and competition from established treatments like clomiphene citrate may moderate adoption.The enclomiphene segment has seen increasing clinical and patient interest as a non‑steroidal medium dose (12.5 mg - 25 mg) therapy that stimulates the body’s own testosterone production while preserving fertility, positioning it as an alternative for men with hypogonadism who wish to avoid the suppressive effects of traditional testosterone replacement therapy (TRT). Enclomiphene works by blocking estrogen receptors in the hypothalamus and boosting luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which, in turn, elevate endogenous testosterone levels without shutting down the hypothalamic-pituitary-gonadal axis. Recent clinical data, including a 2025‑- 2026 systematic review/meta-analysis of randomized controlled trials, confirmed that selective estrogen receptor modulators like enclomiphene significantly improve testosterone, LH, and FSH levels in men with functional hypogonadism, offering potential benefits over placebo and comparable hormonal effects to topical testosterone, with the added advantage of fertility preservation.
The segment’s growth is driven by a rising prevalence of male hypogonadism, increasing demand for fertility‑sparing testosterone therapies, and patient preference for medium dose (12.5 mg - 25 mg) treatments that avoid injections and the systemic suppression associated with traditional TRT. Metabolic and lifestyle factors such as obesity and type‑2 diabetes, which contribute to secondary hypogonadism, are further fueling interest in alternatives like enclomiphene that target underlying hormonal axis dysfunction. Clinical studies and meta-analyses have demonstrated that enclomiphene and similar selective estrogen receptor modulators (SERMs) not only effectively raise testosterone levels but also preserve spermatogenesis better than conventional testosterone therapies. These advantages make it a preferred option for men seeking both efficacy and fertility preservation. Growing awareness among healthcare providers and patients regarding long-term safety and quality-of-life benefits is further supporting adoption. As more real-world use and incremental research emerge, the segment continues to expand, emphasizing patient-centered hormonal management.
Market Concentration & Characteristics
The market is characterized by a high degree of innovation, driven by its unique mechanism as a selective estrogen receptor modulator that stimulates endogenous testosterone production while preserving fertility. Unlike traditional testosterone replacement therapies, it offers medium dose (12.5 mg - 25 mg) administration and reduced systemic suppression, addressing unmet needs in male hypogonadism. Ongoing research focuses on optimizing dosing regimens, combination therapies, and improving patient adherence. This innovative approach positions the segment as a rapidly evolving niche within men’s hormonal health.
The enclomiphene industry is characterized by a high degree of innovation, driven by its unique mechanism as a selective estrogen receptor modulator that stimulates endogenous testosterone production while preserving fertility. However, barriers to market entry are significant, including stringent regulatory approvals, limited clinical data, and the need for specialized manufacturing expertise. Unlike traditional testosterone therapies, it requires careful formulation and monitoring to ensure safety and efficacy. These factors make the market highly specialized, favoring established players with clinical and regulatory capabilities.
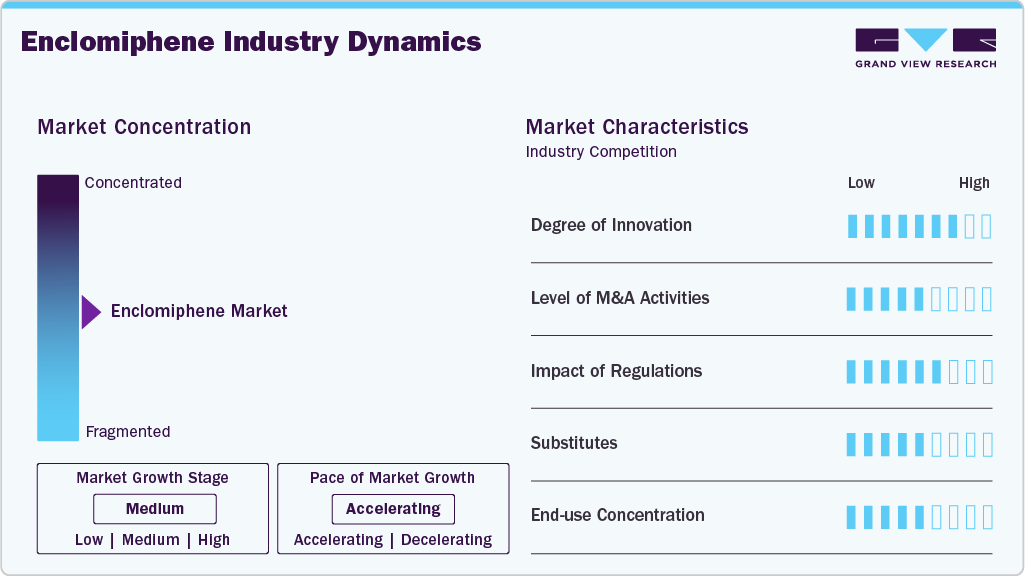
The market is significantly influenced by regulatory frameworks, as the therapy is often used off‑label and is not yet FDA‑approved, requiring careful oversight in prescription and distribution. Regulatory guidelines on clinical trials, safety monitoring, and hormone therapy protocols shape product development and market entry. Compliance with international standards is essential for compounding pharmacies and digital health platforms offering enclomiphene. These regulations ensure patient safety but also affect the pace of adoption and investment in this emerging segment.
The market faces competition from various type substitutes, including traditional testosterone replacement therapies such as gels, injections, and patches, as well as other selective estrogen receptor modulators like clomiphene citrate. While these alternatives also aim to restore testosterone levels, enclomiphene differentiates itself by preserving fertility and offering convenient medium-dose (12.5 mg - 25 mg) administration. The availability of multiple substitutes drives innovation and choice within the market, encouraging developers to enhance efficacy, safety, and patient adherence. This dynamic makes the segment highly competitive yet ripe for growth in men’s hormonal health.
The enclomiphene is witnessing strategic expansion, with growing adoption in North America and Europe driven by high awareness of male hypogonadism and advanced healthcare infrastructure. Emerging markets in Asia-Pacific and Latin America are also showing potential, driven by the increasing prevalence of lifestyle-related hypogonadism and rising access to telemedicine platforms. Companies are leveraging digital health channels and local partnerships to improve availability and patient education. This global outreach is accelerating market penetration and supporting broader acceptance of enclomiphene therapies.
Product Type Insights
The enclomiphene citrate tablets segment dominated the market with the largest revenue share of 63.20% in 2025, driven by strong patient preference for oral formulations that offer convenient administration and improved compliance compared to injectable or topical testosterone therapies. The segment benefits from its mechanism of stimulating endogenous testosterone production while preserving spermatogenesis, making it particularly attractive for men seeking fertility-preserving treatment options. Clinical evidence continues to support its efficacy, with a 2025 meta-analysis reporting significant increases in testosterone, LH, and FSH levels among treated patients, reinforcing its therapeutic value. Additionally, studies indicate that oral enclomiphene can raise testosterone levels by approximately 150-340 ng/dL within weeks, highlighting its rapid clinical response and effectiveness. For instance, in September 2025, digital health platform expansions introduced personalized oral enclomiphene tablet regimens combined with adjunct therapies, improving accessibility and adherence to treatment protocols. Furthermore, ongoing developments in compounding practices and tailored dosing strategies are enhancing product availability and customization, supporting continued growth of the tablet segment within male hormonal therapy.
The enclomiphene capsules segment is projected to grow at a CAGR of 10.62% over the forecast period. driven by increasing demand for personalized and flexible dosing formats enabled through compounding pharmacies and telehealth-led treatment models. Capsules are particularly favored for their ability to support precise dose titration and combination therapies, making them suitable for long-term management of secondary hypogonadism and fertility-focused care. The segment is also benefiting from the rising adoption of compounded medications, as enclomiphene remains prescription-based and not widely commercially standardized, allowing capsule formulations to be tailored to individual patient needs. Clinical evidence indicates that enclomiphene therapies can increase testosterone levels while preserving sperm production, reinforcing their role in fertility-conscious treatment approaches. For instance, in November 2025, telehealth platform Strut Health expanded access to custom-compounded men’s health formulations, including enclomiphene-based therapies, emphasizing personalized dosing and capsule-based delivery through physician-guided prescriptions. Furthermore, the increasing shift toward subscription-based digital health models and individualized hormone therapy protocols continues to strengthen the adoption of capsule formulations within this segment
Dosage Strength Insights
The medium dose (12.5mg -25 mg) segment accounted for the largest revenue share of 51.31% in 2025, driven by its optimal balance of efficacy and tolerability, making it the most commonly prescribed dosing range in clinical practice. This dosage range is widely adopted as it consistently stimulates endogenous testosterone production while minimizing adverse effects, with studies showing both 12.5 mg and 25 mg doses significantly increase testosterone, LH, and FSH levels comparable to topical therapies. Clinical protocols and position statements published in 2026 indicate that treatment typically begins within this range and is titrated based on patient response, reinforcing its standardization in therapeutic use. Additionally, dose-finding studies demonstrate that the 12.5-25 mg range achieves stable hormonal responses, with peak steady-state testosterone levels observed at 25 mg, supporting its dominance. The segment is further driven by increasing preference for fertility-preserving therapies, as this dosing range effectively maintains spermatogenesis while restoring hormone levels. For instance, in December 2025, a published meta-analysis of randomized controlled trials confirmed that SERM therapies, including enclomiphene, significantly increased testosterone by ~273.76 ng/dL while preserving fertility outcomes, reinforcing the clinical preference for standard dosing ranges. Furthermore, ongoing developments in personalized hormone therapy protocols and physician-guided dose titration strategies continue to strengthen the adoption of medium-dose enclomiphene across treatment settings.
The low-dose (≤12.5 mg) segment is projected to grow at a CAGR of 10.47% over the forecast period, driven by an increasing preference for conservative, patient-tailored hormone therapy approaches that minimize side effects while maintaining therapeutic efficacy. Lower doses such as 6.25 mg and 12.5 mg have demonstrated the ability to significantly increase testosterone, LH, and FSH levels while reducing the risk of adverse events, making them suitable for early-stage hypogonadism and long-term maintenance therapy. This segment is particularly favored among younger patients and those prioritizing fertility preservation, as enclomiphene maintains sperm production while restoring hormonal balance.
Additionally, improved tolerability profiles are supporting adoption, with a 2024 clinical study reporting lower adverse event rates than with alternative therapies, reinforcing the clinical value of lower-dosing strategies. The segment is also benefiting from the rise of personalized medicine and compounding pharmacy practices that enable precise low-dose capsule and tablet formulations. For instance, in May 2025, a clinical and compounding-focused publication highlighted increasing physician adoption of dose-flexible enclomiphene regimens starting at 6.25 mg, emphasizing individualized titration strategies to optimize safety and outcomes. Furthermore, ongoing developments in telehealth-driven hormone management and customized dosing protocols continue to strengthen the adoption of low-dose enclomiphene therapies across diverse patient populations.
Indication Insights
The male hypogonadism segment dominated the market with the largest revenue share of 49.10% in 2025, driven by the rising prevalence of secondary hypogonadism associated with obesity, metabolic syndrome, and type-2 diabetes, particularly among aging male populations. Enclomiphene is increasingly preferred due to its ability to stimulate endogenous testosterone production while preserving spermatogenesis, making it highly suitable for fertility-conscious patients. Clinical research continues to validate its role, with studies highlighting improved hormonal balance and reduced suppression of the hypothalamic-pituitary-gonadal axis compared to traditional testosterone therapies. Additionally, increasing awareness and screening for testosterone deficiency are supporting earlier diagnosis and treatment adoption across healthcare settings. The segment is further benefiting from a shift toward personalized, fertility-preserving hormonal management. For instance, in February 2025, the European Association of Urology (EAU) highlighted in its updated Sexual and Reproductive Health Guidelines that comprehensive hormonal evaluation, including testosterone and gonadotropin assessment, is essential for diagnosing male infertility and hypogonadism. The guidelines also emphasize targeted, etiology-based treatment approaches, supporting the clinical relevance of fertility-preserving therapies such as selective estrogen receptor modulators, which underpin the emerging interest in agents like enclomiphene.
The male infertility segment is projected to grow at a CAGR of 10.74% over the forecast period, driven by the rising global incidence of male reproductive disorders, declining sperm counts, and increasing awareness of male-factor infertility, which contributes to nearly 40-50% of infertility cases globally in 2024. Enclomiphene is gaining traction in this segment due to its ability to stimulate endogenous testosterone while preserving spermatogenesis, making it particularly suitable for men seeking fertility restoration rather than suppression. Clinical studies have demonstrated that enclomiphene not only improves hormonal profiles but also enhances sperm parameters, including motility and total motile sperm count, supporting its role in fertility-focused treatment strategies. Additionally, increasing adoption of assisted reproductive technologies (ART) and pre-treatment hormonal optimization is driving demand for therapies that improve natural fertility potential. The segment is further supported by the growing integration of hormonal therapies into fertility clinic protocols and personalized treatment pathways. For instance, in April 2023, a clinical study published in the American Urological Association journals reported that enclomiphene use was associated with improved pregnancy outcomes compared to alternative therapies in men undergoing infertility treatment, highlighting its expanding clinical relevance in fertility-focused care.
Distribution Channel Insights
The hospital pharmacies segment accounted for the largest revenue share of 49.50% in 2025, driven by the need for physician-supervised hormone therapy initiation, dose titration, and continuous monitoring in patients with male hypogonadism and infertility. Hospital pharmacies enable integrated care delivery through collaboration between endocrinologists, urologists, and fertility specialists, ensuring accurate diagnosis and optimized treatment outcomes. The segment is further supported by increasing demand for complex hormone therapies and compounded formulations that require controlled dispensing environments and clinical oversight. Additionally, the rise in hospital-based fertility treatments and endocrine disorders is increasing patient reliance on in-house pharmacy services. Pharmacists routinely provide clinical pharmacy services to a majority of inpatients in over 75% of hospitals, highlighting the expanding role of hospital pharmacies in direct patient care and complex therapy management. For instance, in July 2024, AdventHealth began construction on a USD 220 million expansion project at its Daytona Beach hospital, including the addition of over 240,000 square feet, 104 inpatient beds, and expanded support services such as a new and enlarged pharmacy and laboratory infrastructure, aimed at enhancing integrated care delivery and supporting advanced medication management within hospital settings.
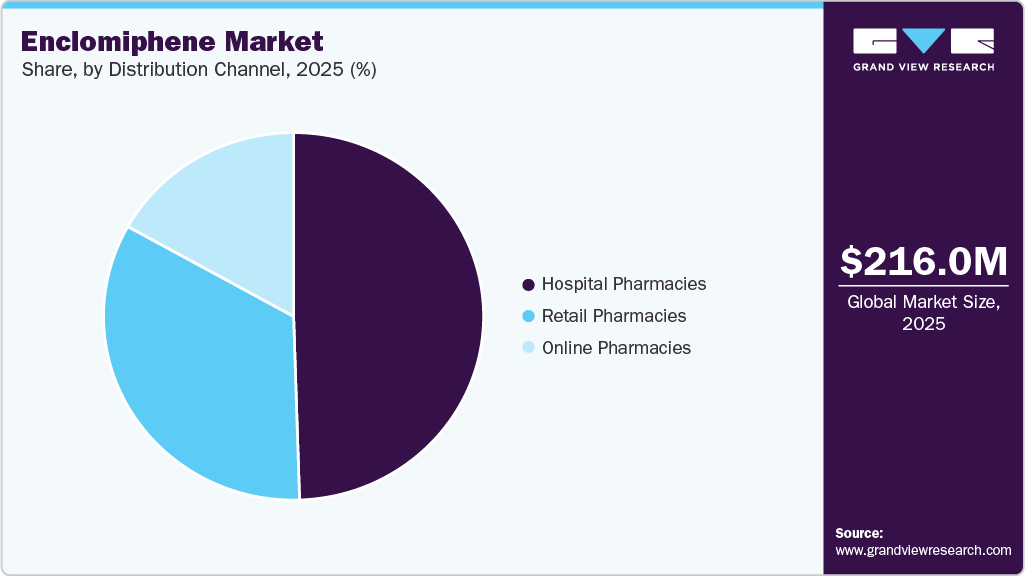
The online pharmacies segment is expected to grow at the fastest CAGR during the forecast period. The market is gaining significant traction, driven by increasing patient preference for convenient, discreet, and home-delivered access to hormone therapies, particularly for conditions such as male hypogonadism and infertility. The segment is supported by the growing adoption of telemedicine platforms that enable virtual consultations, e-prescriptions, and seamless medication fulfillment, thereby improving access to specialized hormonal treatments. Additionally, online pharmacies facilitate personalized treatment pathways, subscription-based medication delivery, and better adherence through digital monitoring tools, making them highly suitable for long-term therapies. The increasing burden of endocrine disorders and rising acceptance of remote healthcare services are further accelerating adoption. Moreover, integration of digital health ecosystems and patient engagement tools is enhancing continuity of care and treatment outcomes. For instance, in January 2024, CVS Health announced enhancements to its digital pharmacy and loyalty platform, including its ExtraCare Plus membership, which offers free same-day delivery of prescriptions and health products, strengthening its online pharmacy capabilities and improving patient access to medications through digital channels.
Regional Insights
The North America enclomiphene market held the largest market share of 40.97% in 2025, driven by rising awareness of male hypogonadism and increasing patient preference for fertility-preserving testosterone therapies. Medium Dose (12.5 mg - 25 mg) administration and reduced systemic suppression compared to traditional TRT make enclomiphene a favorable option among men seeking convenient and safer hormonal management. Advanced healthcare infrastructure, widespread clinical adoption, and growing physician familiarity with selective estrogen receptor modulators (SERMs) further support market expansion. Additionally, the prevalence of lifestyle-related conditions such as obesity and type-2 diabetes, which contribute to secondary hypogonadism, is increasing demand for innovative treatment options. With ongoing research into optimized dosing and combination therapies, the North American market continues to evolve with a focus on efficacy, safety, and patient quality of life.
U.S. Enclomiphene Market Trends
The U.S. enclomiphene market is experiencing steady growth, driven by increasing patient and clinician interest in non‑steroidal, fertility‑preserving alternatives to traditional testosterone replacement therapy (TRT) for male hypogonadism. Although enclomiphene is not currently FDA‑approved and is often accessed through compounding pharmacies, its mechanism of stimulating endogenous testosterone production while maintaining spermatogenesis appeals to men who want to avoid injections and preserve fertility, boosting demand in specialized endocrinology and men’s health practices. Rising awareness of lifestyle‑related secondary hypogonadism linked to obesity and metabolic syndrome is also contributing to broader consideration of innovative hormonal therapies. Enhanced digital health platforms and telemedicine services in the U.S. are increasing access to personalized hormonal care, enabling more men to explore tailored treatment options. Clinicians are progressively discussing enclomiphene as part of the comprehensive management of low testosterone symptoms, despite regulatory limitations and the need for more long‑term data. For instance, in September 2025, Hims & Hers Health expanded its men’s hormone care offerings by enabling providers to prescribe tailored compounded enclomiphene doses through its platform, alongside dual‑action treatment plans for sexual symptoms, signaling growing commercial interest in encapsulating enclomiphene into mainstream testosterone care models in the U.S. market.
Europe Enclomiphene Market Trends
The Europe enclomiphene market is shaped by increasing clinical awareness of male hypogonadism and the need for fertility‑preserving hormonal therapies across the region, with diverse adoption patterns influenced by varying regulatory frameworks and reimbursement policies. While traditional testosterone replacement therapies remain the primary treatment in most countries, selective estrogen receptor modulators like enclomiphene are gaining interest among endocrinologists and urologists seeking alternatives that stimulate endogenous testosterone without compromising fertility. Rising prevalence of aging male populations, lifestyle‑related comorbidities such as obesity and metabolic syndrome, and growing awareness of male reproductive health are key growth drivers. European healthcare systems’ focus on specialist care and evidence-based practice supports integration of novel hormonal treatments within comprehensive men’s health programs. Additionally, increasing patient preference for medium dose (12.5 mg - 25 mg) and less invasive therapies is encouraging exploration of enclomiphene in clinical practice. Overall, the market is gradually expanding as clinicians and patients seek personalized, fertility-conscious solutions for testosterone deficiency.
The UK enclomiphene market is shaped by a unique regulatory and clinical landscape, where the compound remains unlicensed for general prescription use and is typically accessed through specialist or investigational pathways rather than routine NHS prescribing. Enclomiphene’s mechanism stimulating endogenous testosterone production while preserving spermatogenesis draws interest among clinicians and patients seeking alternatives to traditional TRT, particularly for men with secondary hypogonadism who wish to maintain fertility. However, its unapproved status means uptake in the UK is cautious, with clinicians often limiting use to specialist settings or as part of research protocols under Medicines and Healthcare Products Regulatory Agency (MHRA) guidance, and experienced endocrine teams monitor patient response closely. Demand is further influenced by ongoing clinical discourse around alternatives to conventional testosterone therapies, the prevalence of lifestyle‑associated hypogonadism, and evolving private healthcare practices in men's health. For instance, in February 2026, the British Society of Sexual Medicine issued a position statement recognising enclomiphene as a promising medium dose (12.5 mg - 25 mg) therapy for men with male hypogonadism-particularly for those wishing to preserve fertility or who are intolerant to injectable or topical treatments while emphasising its unlicensed status and recommending use under specialist supervision
The Germany enclomiphene market reflects a broader and evolving clinical context for male hypogonadism care, where testosterone deficiency is increasingly recognized as a significant health issue among aging male populations and those with metabolic comorbidities. Although traditional testosterone replacement therapies (TRT) such as gels, injectables, and undecanoate formulations dominate therapeutic use due to established clinical infrastructure and reimbursement pathways, alternatives like enclomiphene - a selective estrogen receptor modulator that stimulates endogenous testosterone while preserving fertility are gaining awareness among specialist endocrinologists and urologists as part of more personalized hormonal management strategies. Germany’s advanced healthcare system and structured endocrinology practices support relatively high diagnosis rates and integration of diverse treatment approaches, but recent observational research highlights varied implementation patterns and underdiagnosis of functional and secondary hypogonadism in routine practice, indicating room for broader adoption of innovative therapies. For instance, in December 2025, a comprehensive observational study on male hypogonadism diagnosis and management in Germany reported that both incidence and prevalence of hypogonadism increased between 2015 and 2021, with conventional testosterone therapies implemented widely but substantial gaps remaining in standardized care pathways underscoring the need for enhanced clinician awareness and guideline‑based management that could include emerging alternatives like enclomiphene therapies. This regional focus on improving diagnostic precision and expanding therapeutic options continues to influence how clinicians and patients view novel hormonal treatments within the German market.
The France enclomiphene market is shaped by evolving clinical interest in male hypogonadism and fertility‑preserving hormonal treatments, although traditional testosterone replacement therapies remain more commonly prescribed given established clinical guidelines and reimbursement pathways. In France, rising awareness of testosterone deficiency and male reproductive health has led to more routine screening and endocrinology referrals, particularly among aging men and those with metabolic comorbidities that exacerbate hypogonadal states. Clinicians are increasingly considering alternatives like selective estrogen receptor modulators (SERMs) to traditional TRT to preserve fertility while managing symptoms of low testosterone in select patient populations, though formal regulatory approvals for enclomiphene itself remain limited in Europe. French healthcare infrastructure, with its emphasis on evidence‑based practice and specialist care, supports the integration of tailored hormonal therapies within comprehensive men’s health programs. For instance, in February 2024, French policymakers discussed expanding infertility screening initiatives, including sperm assessments for men in early adulthood as part of broader reproductive health strategies, underscoring growing attention to male hormonal and fertility issues that indirectly influence interest in therapies like enclomiphene
Asia Pacific Enclomiphene Market Trends
The Asia‑Pacific enclomiphene market is expected to grow at a CAGR of 10.67% during the forecast period, propelled by rising awareness of testosterone deficiency and increasing healthcare access across key economies such as China, India, Japan, and South Korea. Improving diagnostic infrastructure and a growing elderly male population susceptible to hormonal imbalance and metabolic disorders are expanding the addressable cohort for treatments like enclomiphene. Cultural shifts and rising participation in routine health screenings are gradually reducing stigma around sexual health concerns, helping to drive demand for non‑invasive, medium dose (12.5 mg - 25 mg) hormonal therapies. Although adoption levels vary widely across the region due to economic and regulatory diversity, improving physician familiarity with selective estrogen receptor modulators and patient preference for fertility‑preserving alternatives to injectable testosterone are supporting market expansion. Additionally, telemedicine and digital health platforms are enhancing access to endocrinology specialists in both urban and semi‑urban areas, further facilitating uptake of innovative therapies. As healthcare systems in the Asia‑Pacific continue to strengthen and awareness campaigns gain traction, the regional market for enclomiphene and related hypogonadism treatments is expected to grow steadily, increasing its contribution to the global hormonal health landscape.
The Japan enclomiphene market is still in the early stages of development, shaped largely by the broader clinical focus on male hypogonadism and hormone deficiency therapies, with conventional testosterone replacement remaining the dominant treatment approach under recently updated Japanese practice guidelines emphasizing symptom‑oriented diagnosis and management strategies for late‑onset hypogonadism. While enclomiphene itself has not seen formal regulatory approval in Japan, interest in selective estrogen receptor modulators as fertility‑preserving alternatives to testosterone therapies exists within academic and clinical contexts, drawing on international clinical evidence supporting their ability to stimulate endogenous testosterone production. For instance, in October 2025, a study of Japanese men following the 2022 hypogonadism guidelines highlighted evolving treatment practices that integrate tailored hormone management strategies, underscoring a growing clinical awareness of individualized approaches to testosterone deficiency a trend that may indirectly support future interest in enclomiphene‑like modalities. As awareness of male reproductive health issues and diagnostic screening increases in Japan, demand for therapies that both improve testosterone levels and preserve spermatogenesis may gradually rise, especially among men concerned about fertility and quality‑of‑life outcomes.
The China enclomiphene market is still nascent and largely influenced by the broader testosterone and hormone therapy landscape, where conventional testosterone replacement therapies (TRT) remain the primary treatment for male hypogonadism and testosterone deficiency. China’s aging male population, rising awareness of hormonal health issues, and increasing diagnosis of low‑testosterone conditions are driving demand for advanced hormonal treatment options, including fertility‑preserving approaches. Additionally, growing healthcare infrastructure and expanding access to endocrinology services in urban centers are improving awareness of alternative therapeutic strategies that could include selective estrogen receptor modulators in the future. While enclomiphene itself is not yet widely available or routinely prescribed in China, the overall trend toward personalized hormone management and fertility‑focused care supports potential interest in non‑suppressive medium dose (12.5 mg - 25 mg) therapies. Patient and clinician education on male reproductive health and tailored treatment paradigms is expected to accelerate interest over the coming years, particularly as the hormone treatment market expands alongside broader acceptance of hormone therapy for aging‑related deficiencies.
Latin America Enclomiphene Market Trends
In Latin America, the Enclomiphene market is still emerging, with much of the treatment landscape for male hypogonadism centered on traditional therapies and selective estrogen receptor modulators like clomiphene being more commonly discussed and studied as alternatives to conventional testosterone replacement therapy. While specific commercial adoption of enclomiphene itself is limited due to regulatory and development challenges in many regions, the therapeutic concepts behind medium dose (12.5 mg - 25 mg) SERMs that stimulate endogenous testosterone production and preserve fertility are gaining attention among clinicians in endocrinology and urology communities. Healthcare infrastructure variability across countries such as Brazil, Argentina, Mexico, and Chile means uptake of novel hormonal therapies is influenced by access to endocrinologists, compounded prescription availability, and patient demand for fertility‑sparing options. Additionally, the overall regional testosterone deficiency treatment market is growing alongside increasing recognition of hypogonadism prevalence tied to lifestyle and metabolic factors, suggesting potential future interest in alternatives like enclomiphene as awareness and clinical experience expand.
The Brazil enclomiphene market is evolving within the broader context of male hormonal health, where increasing recognition of hypogonadism and male infertility is influencing treatment interest and patient demand. Brazil’s growing population of middle‑aged and aging men, coupled with rising prevalence of obesity, metabolic syndrome, and lifestyle‑related hormonal imbalances, is driving demand for innovative therapies that go beyond conventional testosterone replacement to include fertility‑preserving options. Clinical focus on tailored hormone management and fertility‑oriented care at endocrinology and urology clinics is contributing to heightened awareness of alternatives such as selective estrogen receptor modulators. Improved healthcare infrastructure, expanding access to specialized reproductive and endocrine services in urban centers, and a rising number of diagnostic screenings are further supporting interest in diversified hormonal treatments. Additionally, patient preference for medium dose (12.5 mg - 25 mg) or less invasive treatments that maintain endogenous hormonal axis function is a key driver for adopting next‑generation therapies. As the landscape of hormone therapy in Brazil continues to shift toward personalized and quality‑of‑life‑focused care, the groundwork for enclomiphene‑like therapies to find their niche within male hypogonadism and fertility treatment pathways is strengthening.
Middle East & Africa Enclomiphene Market Trends
The Middle East & Africa enclomiphene market is gradually emerging as part of the broader hormonal and male reproductive health landscape, with growing interest in fertilitypreserving testosterone therapies across the region’s evolving healthcare systems. While traditional testosterone replacement therapies remain the dominant focus in many countries, increasing awareness of male hypogonadism and infertility is contributing to demand for alternatives that maintain spermatogenesis, such as enclomiphene‑based approaches. The market is shaped by rising prevalence of hormonal imbalance and male infertility, driven by lifestyle factors like obesity and diabetes, alongside expanding fertility clinic services and hormone therapy centers in urban hubs like Saudi Arabia, the UAE, and South Africa. Regulatory complexity and limited clinician familiarity with enclomiphene therapies present challenges, but strategic partnerships between regional manufacturers and global API suppliers, coupled with digital health platform expansion, are enhancing supply chain resilience and physician awareness. Investments in healthcare infrastructure and telemedicine are improving access to specialized endocrinology care, indirectly supporting enclomiphene demand. As education on male reproductive health grows and tailored treatment protocols gain traction, the MEA region is poised for incremental adoption of enclomiphene within comprehensive hypogonadism and fertility management strategies.
The Saudi Arabia enclomiphene market is emerging with the country’s expanding focus on male reproductive health and broader fertility care, driven by growing awareness of hormonal imbalances, lifestyle‑related fertility issues, and an increase in infertility diagnosis and treatment services. The country’s investment in fertility clinics, specialized endocrinology services, and healthcare infrastructure under national development initiatives is enhancing access to advanced reproductive therapies that could extend to selective estrogen receptor modulators like enclomiphene as clinician familiarity grows. Rising prevalence of male fertility concerns associated with obesity, metabolic syndrome, and delayed parenthood is encouraging interest in therapies that preserve fertility while addressing testosterone deficiency, complementing existing assisted reproductive services. Public and private sector expansion of reproductive health networks and diagnostics is creating a more supportive environment for comprehensive hormone management. For instance, in July 2025, the Global Fertility Network completed a major acquisition of fertility centers in Riyadh and Jeddah to double IVF and reproductive service capacity in Saudi Arabia, highlighting broader growth in fertility care infrastructure that indirectly supports demand for male hormonal and fertility‑preserving treatments. As this ecosystem evolves, tailored hormonal therapies with a focus on male reproductive outcomes are likely to see increasing engagement from healthcare providers and patients alike.
Key Enclomiphene Company Insights
Repros Therapeutics Inc., Sandoz International GmbH, and Fresenius Kabi AG are actively strengthening the enclomiphene market through continuous innovation in targeted oral formulations that enhance drug stability, bioavailability, and hormonal regulation efficiency. Companies such as Sun Pharmaceutical Industries Ltd. and Aurobindo Pharma Ltd. are expanding their hormone therapy portfolios by investing in advanced oral delivery technologies and cost-effective manufacturing solutions tailored for male hypogonadism and fertility-preserving treatments. Zydus Lifesciences Ltd. and Fresenius Kabi AG are focusing on medium-dose (12.5 mg - 25 mg), controlled-release, and optimized oral enclomiphene formulations, supporting the development of next-generation dosage forms with improved patient adherence and consistent therapeutic outcomes.
Meanwhile, Lupin Limited leverages its expertise in specialty chemistries to enhance formulation consistency, pharmacokinetic performance, and long-term efficacy of enclomiphene therapies. Hetero Labs Limited continues to expand its large-scale API production and cost-efficient manufacturing capabilities, addressing the growing demand for accessible hormone therapies, while Sandoz International GmbH plays a critical role in developing patient-friendly and scalable enclomiphene formulations that improve treatment convenience and compliance. Collectively, these companies are driving advancements in formulation innovation, scalability, and regulatory alignment, supporting the evolving needs of modern enclomiphene market development.
Key Enclomiphene Companies:
The following key companies have been profiled for this study on the enclomiphene market.
- Repros Therapeutics Inc.
- Viatris Inc.
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Zydus Lifesciences Ltd.
- Lupin Limited
- Hetero Labs Limited
- Sandoz International GmbH
- Fresenius Kabi AG
Recent Developments
-
In September 2025, Hims & Hers Health Inc. expanded its men’s health portfolio by introducing compounded enclomiphene as part of its personalized low-testosterone treatment offerings.
-
In March 2026, Rugiet expanded into testosterone replacement therapy (TRT), introducing enclomiphene-based and other personalized hormone treatments, marking its entry into the broader men’s hormone health market.
Enclomiphene Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 233.70 million
Revenue forecast in 2033
USD 419.05 million
Growth rate
CAGR of 8.70% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product type, dosage strength, indication, distribution channel, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Repros Therapeutics Inc.; Viatris Inc.; Teva Pharmaceutical Industries Ltd.; Sun Pharmaceutical Industries Ltd.; Aurobindo Pharma Ltd.; Zydus Lifesciences Ltd.; Lupin Limited; Hetero Labs Limited; Sandoz International GmbH; Fresenius Kabi AG.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Enclomiphene Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global enclomiphene market report based on product type, dosage strength, indication, distribution channel, and region:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Enclomiphene Citrate Tablets
-
Enclomiphene Capsules
-
Others
-
-
Dosage Strength Outlook (Revenue, USD Million, 2021 - 2033)
-
Low Dose (≤12.5 mg)
-
Medium Dose (12.5 mg - 25 mg)
-
High Dose (>25 mg)
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Male Hypogonadism
-
Secondary Hypogonadism
-
Male Infertility
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital pharmacies
-
Retail pharmacies
-
Online pharmacies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










