- Home
- »
- Medical Devices
- »
-
Gastrointestinal Endoscopic Devices Market Report, 2030GVR Report cover
![Gastrointestinal Endoscopic Devices Market Size, Share & Trends Report]()
Gastrointestinal Endoscopic Devices Market (2025 - 2030) Size, Share & Trends Analysis Report By Type (Rigid GI Endoscopes, Flexible GI Endoscopes, Disposable GI Endoscopes), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-494-4
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
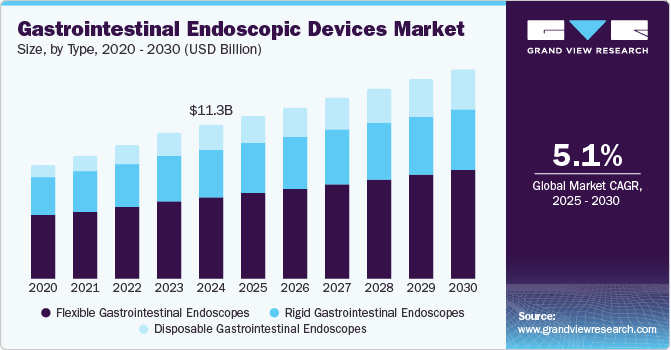
The global gastrointestinal endoscopic devices market size was estimated at USD 11.27 billion in 2024 and is projected to grow at a CAGR of 5.1% from 2025 to 2030. According to the American Cancer Society, approximately 26,380 new cases of stomach cancer were reported in the U.S. in 2022, highlighting the ongoing need for enhanced diagnostic tools such as endoscopy. Moreover, a multinational study by the University of Gothenburg found that over 40% of individuals globally suffer from functional gastrointestinal (GI) disorders, further driving demand for endoscopic procedures.
Technological advancements in endoscopic technology are also playing a significant role in driving market growth. Innovations such as advanced imaging systems, robotic-assisted devices, and enhanced visualization tools facilitate more accurate diagnoses and effective treatments. These advancements make procedures safer and more efficient, driving their adoption in clinical settings. Furthermore, the integration of artificial intelligence and robotic systems into endoscopy is enhancing procedural accuracy and efficiency, providing opportunities for improved patient outcomes.
The preference for minimally invasive surgeries is another key driver of the market. Studies have shown that laparoscopic techniques are used in nearly 80% of colorectal surgeries in North America, reflecting a broader shift toward less invasive options that reduce recovery times and complications. The increasing adoption of minimally invasive techniques is expected to enhance patient outcomes and satisfaction.
Furthermore, public health initiatives in countries such as Brazil have led to a rise in colonoscopy screenings, with reports showing an increase of approximately 25% in screening rates over the past five years. The U.S. Preventive Services Task Force recommends regular screening for colorectal cancer starting at age 45, contributing to a projected increase in screening participation rates among adults by 15% by 2025 as awareness grows. These initiatives are expected to drive demand for endoscopic procedures and enhance patient outcomes.
Type Insights
Flexible gastrointestinal endoscopes dominated the market and accounted for a share of 53.2% in 2024, driven by their pivotal role in diagnosing and treating various gastrointestinal disorders. These minimally invasive procedures provide real-time visualization, enhance patient outcomes, and reduce recovery times, thus supporting the growing need for early disease detection.
Disposable gastrointestinal endoscopes are expected to grow at the fastest CAGR of 7.8% over the forecast period, aided by increasing concerns over infection control and patient safety. Single-use devices eliminate cross-contamination risks, making them a preferred choice in healthcare settings. Convenience and reduced sterilization needs further drive their adoption.
End Use Insights
Outpatient facilities led the market and accounted for a share of 54.6% in 2024 due to the trend towards minimally invasive procedures that allow patients to undergo diagnostics and treatments without prolonged hospital stays. This shift enhances patient convenience, reduces healthcare costs, and makes outpatient settings increasingly popular for endoscopic interventions.
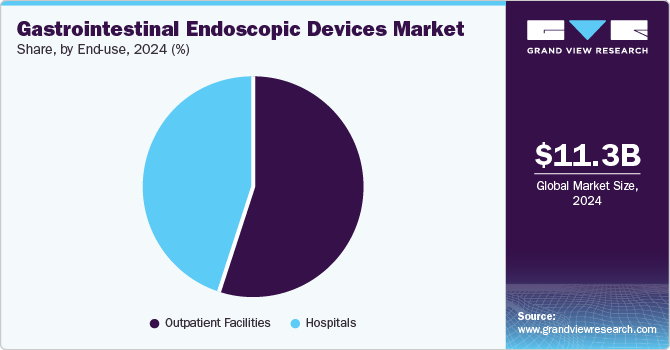
The hospitals sector is projected to grow lucratively over the forecast period, fueled by their comprehensive capabilities in managing complex cases and emergencies. Equipped with advanced technology and skilled personnel, hospitals provide a wide range of diagnostic and therapeutic procedures, driving the need for sophisticated endoscopic equipment to address various gastrointestinal disorders effectively.
Regional Insights
North America gastrointestinal endoscopic devices market dominated the global market with a revenue share of 39.5% in 2024. The region experiences advanced healthcare infrastructure, high prevalence of gastrointestinal disorders, and increasing adoption of minimally invasive surgical techniques. The region’s emphasis on technological innovation and patient safety drives demand.
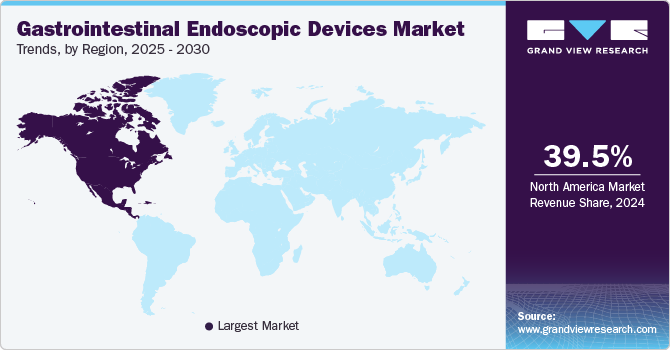
U.S. Gastrointestinal Endoscopic Devices Market Trends
The gastrointestinal endoscopic devices market in the U.S. dominated the North America gastrointestinal endoscopic devices market with the largest revenue share in 2024. The country experiences a significant rise in chronic diseases, particularly gastrointestinal disorders, and an aging population. The growing preference for minimally invasive procedures and continuous advancements in endoscopic technologies enhance diagnostic and therapeutic capabilities, boosting market growth.
Europe Gastrointestinal Endoscopic Devices Market Trends
Europe gastrointestinal endoscopic devices market held substantial market share in 2024, owing to increasing awareness about gastrointestinal health, rising incidences of colorectal cancer, and supportive healthcare policies promoting early diagnosis. The region’s focus on advanced medical technologies and robust healthcare systems contributes to its growth in the global market.
The gastrointestinal endoscopic devices market in Germany is expected to grow rapidly in the forecast period, supported by its strong healthcare infrastructure, high investment in medical technology, and a significant number of skilled healthcare professionals. The increasing prevalence of gastrointestinal diseases and proactive approach to preventive care enhance demand for endoscopic devices.
Asia Pacific Gastrointestinal Endoscopic Devices Market Trends
Asia Pacific gastrointestinal endoscopic devices market is expected to register significant growth over the forecast period. Rising healthcare expenditures, increasing awareness of gastrointestinal health, and a growing geriatric population in the region are driving market growth. The expansion of healthcare infrastructure and technological advancements drive demand for endoscopic procedures across the region.
The gastrointestinal endoscopic devices market in India is projected to grow at the fastest rate of 5.6% in the Asia Pacific market over the forecast period. Increased investments in healthcare infrastructure, growing awareness about preventive health measures, and government initiatives to improve access to diagnostic services propel market growth.
Key Gastrointestinal Endoscopic Devices Insights
Some key companies operating in the market include Olympus Corporation; Boston Scientific Corporation; Medtronic; Johnson & Johnson Services, Inc. (Ethicon Inc.); KARL STORZ SE & Co. KG; among others. Major players are leveraging product innovation and strategic partnerships to strengthen their market presence, focusing on advanced technologies such as robotic-assisted endoscopes and disposable devices.
-
Boston Scientific Corporation is a premier provider of gastrointestinal endoscopic devices, featuring an extensive portfolio that includes stents, balloons, clips, and retrieval instruments. Their advanced technologies, such as the AXIOS Stent, significantly enhance minimally invasive procedures and improve patient outcomes.
-
Fujifilm Holdings Corporation is a key competitor in the gastrointestinal endoscopic devices sector, specializing in endoscopic systems and imaging technologies. Their extensive product range focuses on advanced visualization and diagnostics, enhancing procedural efficiency and patient care in endoscopy.
Key Gastrointestinal Endoscopic Devices Companies:
The following are the leading companies in the gastrointestinal endoscopic devices market. These companies collectively hold the largest market share and dictate industry trends.
- Olympus Corporation
- Boston Scientific Corporation
- Medtronic
- Johnson & Johnson Services, Inc. (Ethicon Inc.)
- KARL STORZ SE & Co. KG
- Fujifilm Holdings Corporation
- Stryker
- HOYA Corporation (PENTAX Medical)
- Smith+Nephew
- Cook
- CONMED Corporation
- Ambu A/S
- Richard Wolf GmbH
- STERIS plc
Recent Developments
-
In December 2024, PENTAX Medical received FDA 510(k) clearance for its new i20c Video Endoscope Series models, enhancing detection, diagnosis, and therapy through advanced imaging and improved ergonomics for healthcare professionals.
-
In October 2024, Cook Medical launched the U.S. distribution of PillSense, an FDA-approved GI bleed detection system, allowing rapid identification of upper gastrointestinal bleeding in under 10 minutes for efficient patient triage.
-
In September 2024, Olympus Latin America launched the EVIS X1 endoscopy system in Brazil, hosting events featuring expert lectures on colorectal cancer and demonstrations of advanced visualization technologies.
-
In August 2024, Ambu announced CE mark approval for its new generation duodenoscopy solution, Ambu aScope Duodeno 2 and aBox 2, enhancing workflow efficiency and infection control in ERCP procedures.
-
In April 2024, Medtronic unveiled its AI innovations for endoscopic care at the Genius Summit, introducing ColonPRO software and announcing a strategic collaboration with Modernizing Medicine to enhance patient workflow efficiency.
-
In January 2024, KARL STORZ acquired British AI firm Innersight Labs to enhance its healthcare portfolio with innovative 3D modeling software, supporting more efficient and effective surgical procedures for medical professionals.
Gastrointestinal Endoscopic Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 11.88 billion
Revenue forecast in 2030
USD 15.27 billion
Growth rate
CAGR of 5.1% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S., Canada, Mexico, UK, Germany, France, Italy, Spain, Denmark, Sweden, Norway, China, Japan, India, Australia, South Korea, Thailand, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait
Key companies profiled
Olympus Corporation; Boston Scientific Corporation; Medtronic; Johnson & Johnson Services, Inc. (Ethicon Inc.); KARL STORZ SE & Co. KG; Fujifilm Holdings Corporation; Stryker; HOYA Corporation (PENTAX Medical); Smith+Nephew; Cook; CONMED Corporation; Ambu A/S; Richard Wolf GmbH; STERIS plc
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Gastrointestinal Endoscopic Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global gastrointestinal endoscopic devices market report based on type, end use, and region:

-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Rigid Gastrointestinal Endoscopes
-
Gastroscope (Upper GI Endoscope)
-
Enteroscope
-
Sigmoidoscope
-
Duodenoscope
-
-
Flexible Gastrointestinal Endoscopes
-
Gastroscope (Upper GI Endoscope)
-
Enteroscope
-
Sigmoidoscope
-
Duodenoscope
-
Colonoscope
-
-
Disposable Gastrointestinal Endoscopes
-
Gastroscope (Upper GI Endoscope)
-
Enteroscope
-
Sigmoidoscope
-
Duodenoscope
-
Colonoscope
-
-
-
End Use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals
-
Outpatient Facilities
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global gastrointestinal endoscopic devices market was estimated at USD 11.3 billion in 2024 and is expected to reach USD 11.9 billion in 2025 and is expected to witness a significant growth owing to changing lifestyle coupled with unhealthy eating habits that has given rise to numerous gastrointestinal (GI) disorders.
b. The global gastrointestinal endoscopic devices market is expected to grow at a compound annual growth rate of 5.1% from 2025 to 2030 to reach USD 15.3 billion by 2030. Growing weight consciousness especially among women leads to adoption of weight management treatments options such as banding and bypass surgery utilizing gastrointestinal endoscopic devices is expected to augment the market growth.
b. North America dominated the gastrointestinal endoscopic devices market with a share of 39.5% in 2024. This is attributable to increasing healthcare expenditure, technological advancement such as flexible scopes and wireless capsules and rising patient awareness levels pertaining early disease diagnostics.
b. Some key players operating in the gastrointestinal endoscopic devices market include Covidien Ltd.; Stryker Corporation; Boston Scientific Corporation; Johnson & Johnson Ltd.; Fujinon Ltd. and Conmed Corporation.
b. Key factors that are driving the market growth include growing geriatric population with long term medical conditions such as GI disorders and increasing prevalence of colon & rectum cancer due to sedentary lifestyle, overweight & tobacco consumption.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










