- Home
- »
- Medical Devices
- »
-
Implantable Tibial Neuromodulation Market Size Report, 2033GVR Report cover
![Implantable Tibial Neuromodulation Market Size, Share & Trends Report]()
Implantable Tibial Neuromodulation Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Leadless Tibial Implants, Lead-Based Tibial Implants, Hybrid/Accessory Systems), By Indication, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-886-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Implantable Tibial Neuromodulation Market Summary
The global implantable tibial neuromodulation market size was estimated at USD 54.46 million in 2025 and is projected to reach USD 87.37 million by 2033, growing at a CAGR of 6.88% from 2026 to 2033. The market is driven by the rising prevalence of conditions such as overactive bladder and urinary incontinence, particularly among the aging population.
Key Market Trends & Insights
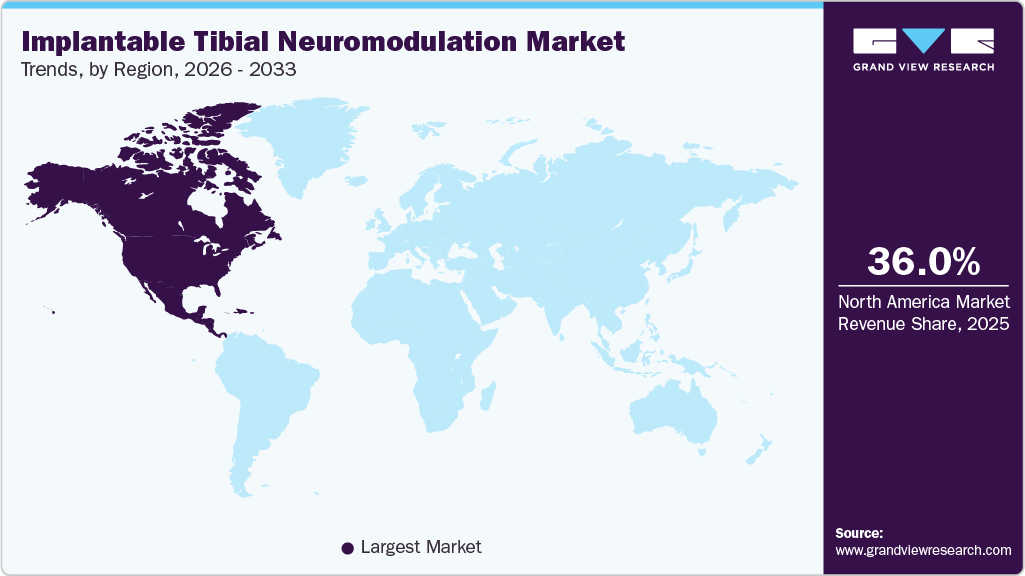
- North America dominated the global implantable tibial neuromodulation industry with the largest revenue share of 36.05% in 2025.
- The implantable tibial neuromodulation industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product, the leadless tibial implants segment led the market with the largest revenue share in 2025.
- By indication, the overactive bladder (OAB) segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 54.46 Million
- 2033 Projected Market Size: USD 87.37 Million
- CAGR (2026-2033): 6.88%
- North America: Largest market in 2025
The increasing preference for minimally invasive, long-term treatment solutions over conventional therapies, along with technological advancements in neuromodulation devices, is further supporting market growth. In addition, growing awareness and improved access to urological care are accelerating adoption rates globally.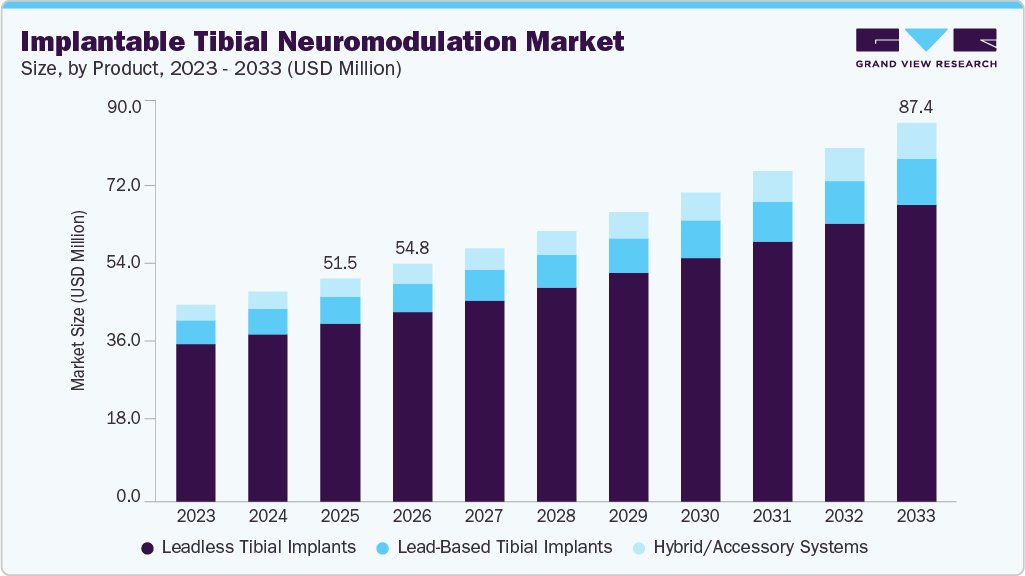
The increasing prevalence of bladder disorders such as overactive bladder and urinary incontinence is a key driver of the implantable tibial neuromodulation industry, as these conditions require effective long-term management solutions. As patient numbers rise globally, demand for minimally invasive, durable therapies is increasing. This trend is accelerating the adoption of implantable neuromodulation devices that offer improved symptom control and quality of life. According to the National Center for Biotechnology Information, in August 2024, the prevalence of urinary incontinence in men was approximately half that observed in women. It was estimated that 11% to 34% of older men experience urinary incontinence, with 2% to 11% reporting daily symptoms, while the risk is notably higher among individuals undergoing prostate surgery.
Prevalence of Urinary Incontinence (2024)
Category
Data / Statistics
Global prevalence (age ≥20)
~423 million individuals affected worldwide
U.S. (overall)
~13 million individuals affected
Nursing facility residents
≥50% prevalence
Long-term care residents (>100 days)
>75% prevalence
Homebound older adults
53% reported incontinent
Hospitalized older patients
11% at admission; 23% at discharge
Women (overall)
24%-45% prevalence
Women aged 20-39
7%-37% experience incontinence
Women aged >60 (daily)
9%-39% report daily incontinence
Source: NCBI
The growing preference for minimally invasive therapies is another key driver of the implantable tibial neuromodulation industry, as patients seek treatments with lower surgical risk, faster recovery, and fewer complications. Implantable tibial neuromodulation offers an effective alternative to more invasive procedures while improving comfort and convenience. This shift in treatment preference is accelerating adoption among both patients and healthcare providers. For instance, in March 2026, the American Urological Association highlighted that implantable tibial neuromodulation devices can be placed through a small incision in a single-stage outpatient procedure under local anesthesia, reducing the need for repeated hospital visits and complex surgeries. This shift toward minimally invasive, low-burden treatment approaches is significantly driving the adoption of implantable tibial neuromodulation in clinical practice.
Furthermore, technological advancements in neuromodulation are significantly driving the implantable tibial neuromodulation industry, with innovations such as miniaturized implants, leadless designs, and wireless control systems improving treatment precision and patient comfort. The development of rechargeable and long-lasting devices is also enhancing therapy convenience and reducing the need for repeat procedures. These advancements are increasing clinical acceptance and accelerating the adoption of implantable solutions.
"The Altaviva device represents a significant advancement for patients with urge urinary incontinence who often weigh the invasiveness of a therapy and the potential side effects when considering treatment options," said Dr. Kevin Benson, Urogynecologist at Sanford Health and investigator for the Altaviva device pivotal study (TITAN 2). "I'm excited to offer my patients this simple experience and believe the Altaviva device has the potential to transform lives and bring new hope to more people."
"It's an honor to bring the Altaviva device to market," said Emily Elswick, president of the Pelvic Health business, which is part of the Neuroscience Portfolio at Medtronic. "For too long, society has told people it is normal to eventually lose bladder control. We say, "enough". What is common does not mean it is normal. The Altaviva device is designed to provide a simple and effective experience to treat urge urinary incontinence, empowering patients with a technology that supports them in their daily lives and opens the door to renewed hope and improved quality of life."
"Too many people silently struggle with the challenges of urgency and loss of bladder control, often believing that relying on diapers or pads is their only option," said Brett Wall, executive vice president and president of the Medtronic Neuroscience Portfolio. "The Altaviva device represents a transformative milestone and ushers in a new bladder control therapy with advanced features. Medtronic has earned its position as the global leader in neuromodulation by consistently pushing the boundaries of innovation, and this FDA approval underscores our unwavering commitment to delivering therapies that truly change lives."
Market Concentration & Characteristics
The degree of innovation in the market for implantable tibial neuromodulation is moderate to high, driven by advancements in miniaturized, leadless, and long-lasting implantable devices that enable minimally invasive treatment. Emerging technologies such as closed-loop stimulation and wireless power systems are enhancing therapy precision and patient convenience. For instance, in September 2025, the National Center for Biotechnology Information published a study highlighting significant innovation in implantable tibial neuromodulation, particularly through the development of advanced implantable percutaneous tibial nerve stimulation (iPTNS) devices. These innovations include variability in device design, power sources, and implantation techniques, as well as emerging concepts such as closed-loop neuromodulation to improve therapeutic efficacy and enable personalized treatment approaches.
The level of mergers and acquisitions in the implantable tibial neuromodulation industry is growing, as larger medtech companies acquire innovative startups to expand their neuromodulation portfolios. For instance, in January 2026, Boston Scientific Corporation announced the acquisition of Valencia Technologies to enter the implantable tibial nerve stimulation segment and strengthen its urology pipeline.
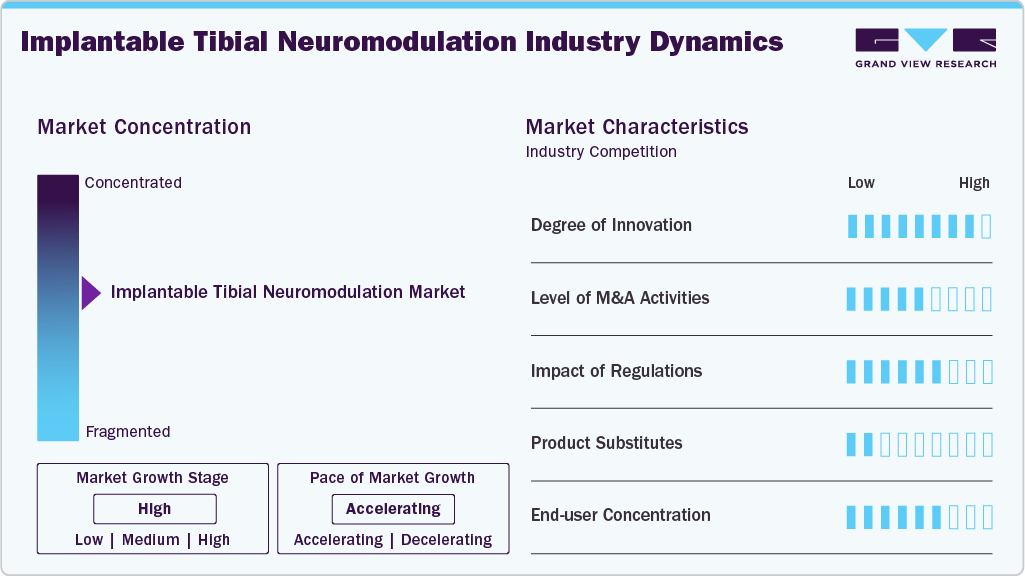
Regulatory frameworks have a significant impact on the market, as stringent approval processes by agencies such as the U.S. Food and Drug Administration ensure safety, efficacy, and long-term reliability of implantable devices. While these regulations may extend product development timelines, they also enhance clinical validation and build patient trust. In addition, evolving regulatory pathways for minimally invasive and neuromodulation technologies are gradually supporting faster adoption and innovation in the market.
End-use concentration in the market is high, with hospitals and specialized urology clinics accounting for most procedures due to the need for skilled professionals and surgical infrastructure. Ambulatory surgical centers are gradually emerging as alternative settings, supported by the shift toward minimally invasive implantation techniques. However, adoption remains concentrated in developed healthcare systems where access to advanced neuromodulation therapies is higher.
Product Insights
Leadless tibial implants dominated the market in 2025, driven by their minimally invasive nature, compact design, and reduced risk of lead-related complications. These systems offer improved patient comfort and simplified implantation procedures, making them highly preferred among clinicians. Their growing clinical adoption and strong performance outcomes have positioned them as the leading segment in the market.
Hybrid/Accessory Systems are expected to experience the fastest growth during the forecast period, driven by increasing demand for customizable, patient-specific treatment solutions. These systems enhance device functionality, improve implantation ease, and support better therapy outcomes, making them increasingly preferred for advanced neuromodulation procedures.
Indication Insights
Overactive bladder (OAB) dominated the market in 2025, driven by its high global prevalence and significant impact on quality of life. The increasing number of patients seeking long-term, minimally invasive treatment options has led to greater adoption of neuromodulation therapies for OAB management. As a result, this segment accounted for the largest share, driven by strong clinical demand and expanding treatment awareness.
Fecal Incontinence (FI) is expected to witness the fastest growth during the forecast period, driven by the rising prevalence of bowel disorders and increasing awareness of neuromodulation as an effective treatment option. The shift toward minimally invasive therapies and growing clinical evidence supporting tibial nerve stimulation for FI management are further accelerating adoption.
End Use Insights
Hospitals dominated the market in 2025, as they serve as the primary settings for device implantation due to the availability of skilled specialists and advanced surgical infrastructure. The need for clinical evaluation, precision procedures, and post-implantation monitoring further supports hospital-based adoption. As a result, hospitals accounted for the largest share owing to higher patient inflow and established treatment capabilities.
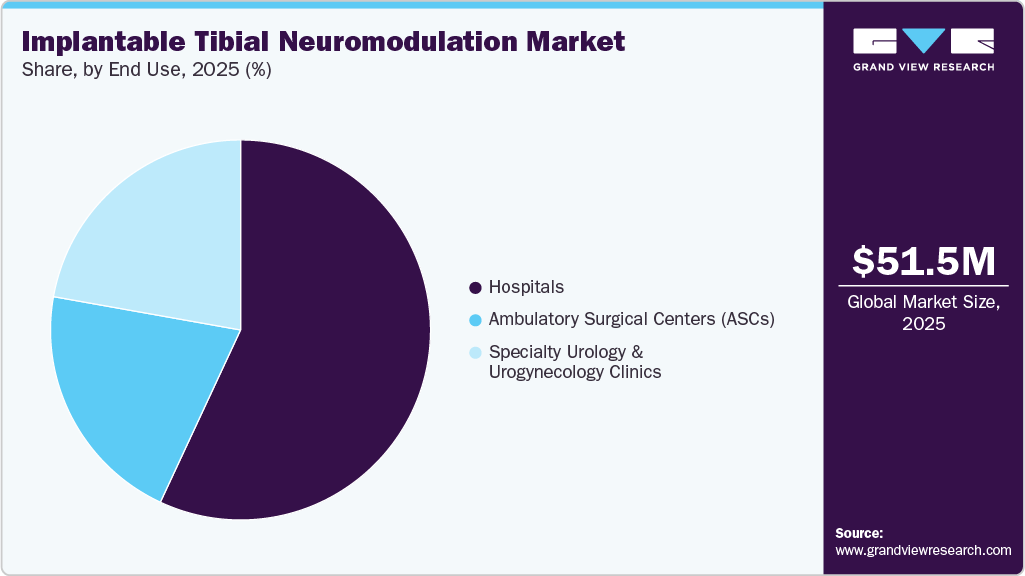
Ambulatory Surgical Centers (ASCs) are expected to witness the fastest CAGR during the forecast period, driven by the increasing shift toward outpatient procedures and cost-effective treatment settings. The minimally invasive nature of tibial neuromodulation, along with shorter recovery times, makes it well-suited for ASCs, supporting higher patient throughput and improved accessibility to care.
Regional Insights
North America Implantable Tibial Neuromodulation Market Trends
The implantable tibial neuromodulation industry in North America held the largest share and accounted for 36.05% of global revenue in 2025. Driven by advanced healthcare systems, high awareness of bladder disorders, and increasing preference for minimally invasive treatment options. The presence of leading medical device companies and ongoing technological innovations is further supporting market growth, while expanding reimbursement coverage is improving patient access across the region.
U.S. Implantable Tibial Neuromodulation Market Trends
The U.S. implantable tibial neuromodulation industry is witnessing strong momentum due to early adoption of innovative implantable therapies and a well-established urology care ecosystem. The increasing preference for minimally invasive treatments, along with growing clinical acceptance among specialists, is driving adoption across hospitals and ambulatory settings. In addition, continuous product innovation and improving patient awareness are supporting wider adoption, positioning the U.S. as a key market for advanced neuromodulation solutions.
Prevalence of Urinary Incontinence in U.S. (2024)
Category
Prevalence / Data
Total affected population (U.S.)
~13 million individuals
Nursing facility residents
≥50% prevalence
Long-term care residents (>100 days)
>75% prevalence
Source: NCBI
Europe Implantable Tibial Neuromodulation Market Trends
The Europe implantable tibial neuromodulation industry is witnessing significant growth, driven by increasing adoption of minimally invasive therapies and a strong focus on improving outcomes in urological care. Rising clinical research activity and growing acceptance among healthcare professionals are supporting the uptake of implantable solutions, while expanding access to advanced treatments and gradual improvements in reimbursement frameworks are further contributing to market expansion across the region.
The UK implantable tibial neuromodulation industry is witnessing gradual growth, driven by rising interest in minimally invasive therapies and a strong public healthcare framework. Adoption is evolving cautiously due to ongoing clinical evaluations and evidence requirements, but rising awareness of treatments for overactive bladder is encouraging uptake. In addition, advancements in device technology and a shift toward outpatient care settings are expected to support future market expansion.
Urinary Incontinence Statistics in the UK (2025)
Category
Data / Statistics
People affected (overall)
1 in 5 individuals (~14 million people)
Women with urinary incontinence
~34%
Men with urinary incontinence
Over 2 million
Source: The Urology Foundation
The France implantable tibial neuromodulation industry is experiencing gradual growth, driven by increasing focus on minimally invasive treatment approaches and improving access to specialized urology care. Growing awareness of bladder disorders and rising demand for long-term therapeutic solutions are supporting adoption, while ongoing advancements in implantable device technology are further strengthening market development.
Asia Pacific Implantable Tibial Neuromodulation Market Trends
The Asia Pacific implantable tibial neuromodulation industry is experiencing strong growth, driven by improving healthcare infrastructure, rising awareness of advanced urological treatments, and a large untreated patient population. Increasing investments in healthcare and the gradual adoption of minimally invasive technologies are supporting market expansion across key countries such as China, India, and Japan.
The China implantable tibial neuromodulation industry is growing significantly, driven by rising demand for minimally invasive treatments and the growing burden of urological disorders among the aging population. Expansion of hospital infrastructure and greater adoption of advanced medical technologies are supporting market development, while improving awareness and access to specialized care are further contributing to adoption.
Latin America Implantable Tibial Neuromodulation Market Trends
The Latin America implantable tibial neuromodulation industry is experiencing gradual growth, supported by improving healthcare access and increasing awareness of minimally invasive treatments for urological conditions. Adoption is rising in urban healthcare centers, particularly in countries such as Brazil and Mexico, while ongoing investments in healthcare infrastructure are further driving market expansion despite cost-related challenges.
MEA Implantable Tibial Neuromodulation Market Trends
The MEA implantable tibial neuromodulation industry is growing gradually, supported by improving healthcare infrastructure and an increasing focus on advanced treatment options in urban healthcare centers. Growing awareness of minimally invasive therapies and expanding access to specialized urology care are driving early adoption, while ongoing investments in healthcare systems are expected to support future market growth.
Key Implantable Tibial Neuromodulation Company Insights
The implantable tibial neuromodulation industry is concentrated, with a few leading medical device companies driving innovation and commercialization, while emerging players contribute with niche technologies. Market share is influenced by product effectiveness, clinical evidence, and physician adoption, with companies focusing on expanding their presence through new product launches and strategic collaborations. Competition is increasing as firms invest in advanced, minimally invasive solutions to strengthen their positioning.
Key Implantable Tibial Neuromodulation Companies:
The following key companies have been profiled for this study on the implantable tibial neuromodulation market.
- Valencia Technologies (Boston Scientific Corporation)
- BlueWind Medical
- Medtronic
Recent Developments
-
In January 2026, Boston Scientific Corporation announced that it had entered into a definitive agreement to acquire Valencia Technologies Corporation, a firm focused on developing innovative solutions for bladder dysfunction. The company’s eCoin System is an implantable tibial nerve stimulation device designed to treat urge urinary incontinence, a key symptom of overactive bladder.
-
In January 2026, Greater Boston Urology announced the first implantation of the Altaviva device in Norwood, Massachusetts. The device, implanted near the ankle above the tibial nerve, is approved by the U.S. Food and Drug Administration for the treatment of urge urinary incontinence.
-
In September 2025, Medtronic plc announced that it had received U.S. Food and Drug Administration approval for its Altaviva device. The minimally invasive implantable tibial neuromodulation therapy is placed near the ankle and is designed to treat urge urinary incontinence.
-
In February 2025, BlueWind Medical Ltd. announced the publication of two-year results from its pivotal OASIS study evaluating the Revi System for the treatment of urgency urinary incontinence. The findings were featured in the March 2025 issue of the Journal of Urology.
Implantable Tibial Neuromodulation Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 54.85 million
Revenue forecast in 2033
USD 87.37 million
Growth rate
CAGR of 6.88% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, indication, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Norway; Denmark; Sweden; Spain; China; Japan; India; South Korea; Australia; Brazil; KSA; UAE; South Africa
Key companies profiled
Valencia Technologies (Boston Scientific Corporation); BlueWind Medical; Medtronic
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Implantable Tibial Neuromodulation Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global implantable tibial neuromodulation market report based on product, indication, end use, and region:

-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Lead-Based Tibial Implants
-
Leadless Tibial Implants
-
Hybrid/Accessory Systems
-
-
Indication Outlook (Revenue, USD Million; 2021 - 2033)
-
Overactive Bladder (OAB)
-
Urge Urinary Incontinence (UUI)
-
Fecal Incontinence (FI)
-
Chronic Pelvic Pain/Interstitial Cystitis
-
Neurogenic Bladder Disorders
-
-
End Use Outlook (Revenue, USD Million; 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Specialty Urology & Urogynecology Clinics
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global implantable tibial neuromodulation market is expected to grow at a compound annual growth rate of 6.88% from 2026 to 2033, reaching USD 87.37 million by 2033.
b. North America dominated the implantable tibial neuromodulation market in 2025.
b. Some key players operating in the implantable tibial neuromodulation market include Valencia Technologies (Boston Scientific Corporation), BlueWind Medical, and Medtronic.
b. The implantable tibial neuromodulation market is driven by the rising prevalence of overactive bladder, urinary incontinence, and other urological disorders along with the growing demand for minimally invasive and drug-free treatment options.
b. The global implantable tibial neuromodulation market size was valued at USD 54.46 million in 2025 and is expected to reach USD 54.85 million in 2026.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










