- Home
- »
- Pharmaceuticals
- »
-
Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market 2033GVR Report cover
![Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market Size, Share & Trends Report]()
Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market (2026 - 2033) Size, Share & Trends Analysis Report By Action of Mechanism (Complement Inhibitors, Supportive Care, Others), By Route of Administration, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-890-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market Summary
The Japan paroxysmal nocturnal hemoglobinuria treatment market size was valued at USD 279.95 million in 2025 and is projected to reach USD 455.53 million by 2033, growing at a CAGR of 5.88% from 2026 to 2033. This growth is driven by continuous advancements in complement inhibitor therapies, including the development of next-generation C3 and Factor B/D inhibitors that improve clinical outcomes and address unmet needs such as residual anemia.
Key Market Trends & Insights
- Based on action of mechanism, the complement inhibitors segment dominated the market with a 91.95% revenue share in 2025.
- On the basis of route of administration, the parenteral segment held the largest revenue share of 96.13% in 2025.
- On the basis of end use, the hospitals & specialty clinics segment led the market with a revenue share of 93.65% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 279.95 Million
- 2033 Projected Market Size: USD 455.53 Million
- CAGR (2026-2033): 5.88%
Additionally, strong reimbursement policies and orphan drug incentives in Japan are supporting patient access to high-cost therapies and encouraging pharmaceutical innovation. Increasing disease awareness and improved diagnostic capabilities are also contributing to earlier detection and treatment, further expanding the treated patient population and sustaining market growth.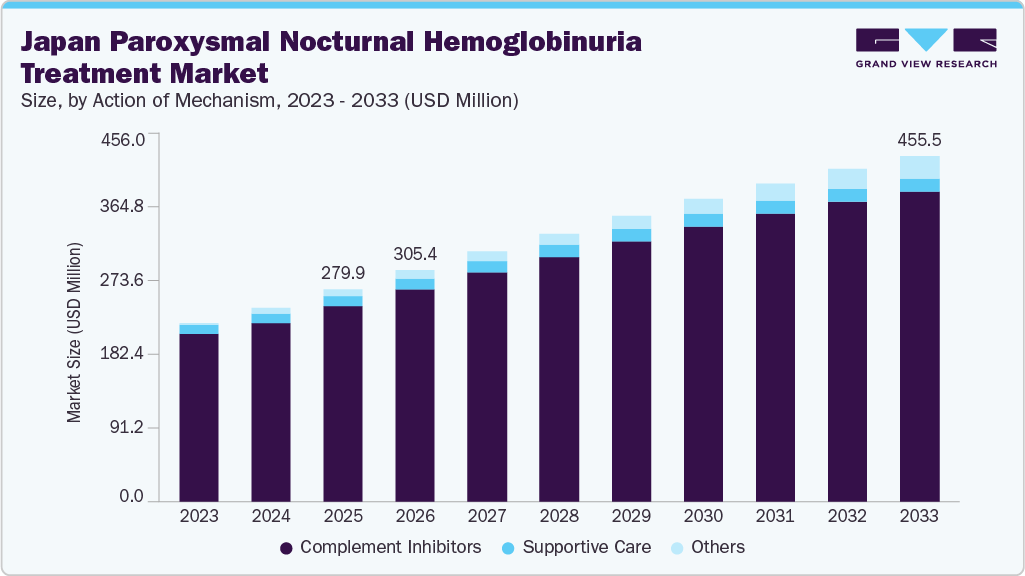
The market is primarily driven by significant advancements in complement inhibitor therapies that directly target the underlying mechanism of complement-mediated hemolysis. The transition from supportive care approaches, such as transfusions and anticoagulation, to targeted biologics has transformed clinical outcomes. Real-world evidence highlights this shift, as reported in February 2026 in the International Journal of Hematology, where 86.7% of 45 Japanese patients were treated with C5 inhibitors, resulting in a median increase in hemoglobin from 7.2 g/dL at diagnosis to 10.0 g/dL during treatment.
Innovation within the market is further strengthened by the evolution from first-generation C5 inhibitors to next-generation therapies, including upstream complement inhibitors targeting C3 and alternative pathway factors. These advancements address residual anemia associated with extravascular hemolysis, an unmet need in earlier treatments. A September 2025 review published in Drugs highlighted phase III trial data involving 441 patients, demonstrating that ravulizumab was non-inferior to eculizumab at 26 and 52 weeks. Additionally, findings from the APPLY-PNH and APPOINT-PNH trials showed that oral iptacopan delivered sustained reductions in hemolysis markers and improved hemoglobin levels compared with continued anti-C5 therapy, signaling a shift toward more comprehensive, patient-friendly treatment options.
Japan’s strong healthcare framework plays a crucial role in market expansion, particularly through universal coverage and supportive orphan drug policies. The government’s reimbursement system, which covers approximately 70% of medical costs, ensures broad access to high-cost biologics and promotes early treatment initiation. Regulatory support further accelerates innovation, with orphan drugs receiving priority review timelines of around 9 months compared to the standard 12-month pathways. These incentives have attracted both global and domestic players, fostering a robust pipeline of advanced therapies and reinforcing Japan as a key market for the development of rare disease treatments.
Increasing diagnosis rates and disease awareness are also contributing significantly to market growth. Improved use of high-sensitivity flow cytometry and standardized diagnostic protocols has enhanced early detection of PNH, particularly among patients with aplastic anemia. An October 2024 MDPI publication reported that PNH clones were identified in 40-60% of patients, with untreated mortality exceeding 35% within 5 years, underscoring the importance of timely diagnosis. Additionally, a March 2024 study in the International Journal of Hematology involving 59 Japanese patients on C5 inhibitors reported a mean hemoglobin level of 10.2 g/dL, highlighting persistent symptom burden and the need for continued therapeutic optimization. Together, these factors are expanding the diagnosed patient population and sustaining long-term growth in the Japan PNH treatment market.
Market Concentration & Characteristics
Innovation in the Japan Paroxysmal Nocturnal Hemoglobinuria (PNH) treatment market remains high, driven by continuous advancements in complement pathway-targeted therapies. The market is characterized by a strong shift from conventional supportive care toward highly specialized biologics, particularly C5 and emerging C3 inhibitors that directly address complement-mediated hemolysis. The transition from first-generation therapies such as eculizumab to next-generation agents, including ravulizumab and oral iptacopan, reflects rapid therapeutic evolution. For instance, a September 2025 Drugs review highlighted phase III data involving 441 patients where ravulizumab demonstrated non-inferiority to eculizumab, while APPLY-PNH and APPOINT-PNH trials showed improved hemoglobin levels with oral iptacopan. Additionally, long-acting formulations and oral therapies are improving patient convenience and adherence, reinforcing innovation-led market growth.
The market exhibits a highly concentrated, specialist-driven structure, dominated by a limited number of global biopharmaceutical companies due to the ultra-rare nature of PNH and the high entry barriers. Complement inhibitors account for over 91.95% of the market share, highlighting strong dependence on a single therapeutic class. Key players such as AstraZeneca (Alexion) maintain a dominant position with flagship products like Soliris (eculizumab) and Ultomiris (ravulizumab), while emerging players like Apellis are introducing C3 inhibitors to challenge existing standards. The competitive landscape is shaped by lifecycle innovation, pipeline expansion, and strategic collaborations to introduce next-generation and oral therapies. However, the small patient population and high development costs limit new entrants, reinforcing a concentrated competitive environment.
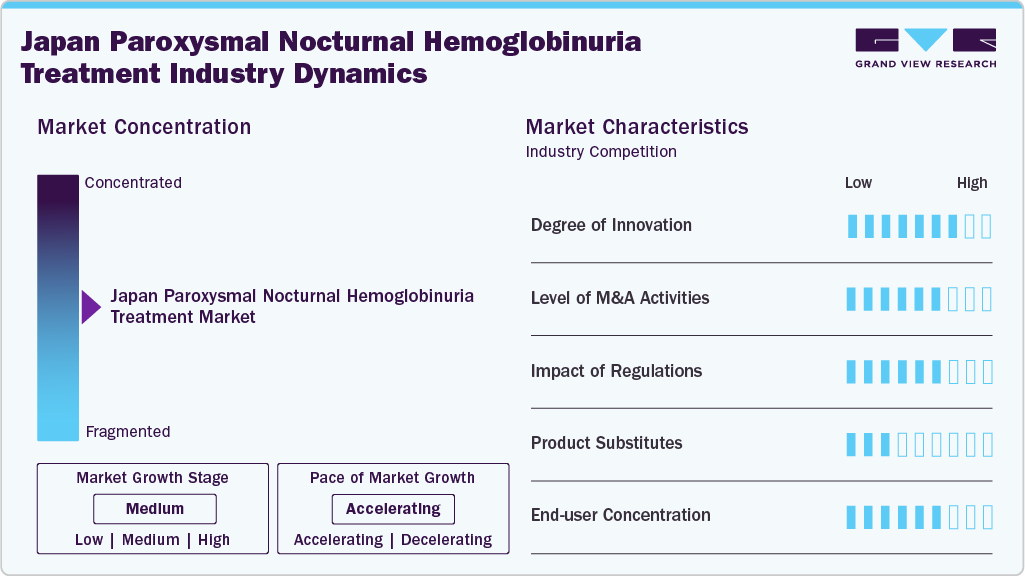
The regulatory and reimbursement framework in Japan plays a critical role in shaping market dynamics, acting as both a facilitator of innovation and a stabilizing force. Strong universal healthcare coverage, which reimburses approximately 70% of treatment costs, ensures widespread access to high-cost biologics and supports sustained demand. Additionally, orphan drug incentives such as tax benefits, research subsidies, and extended exclusivity encourage pharmaceutical investment in rare disease therapies. Accelerated approval pathways, with priority review timelines of around nine months, enable faster market entry for innovative treatments, strengthening Japan’s position as a favorable launch market for advanced PNH therapies.
The market demonstrates low to moderate substitutability, as complement inhibitors remain the standard of care with limited alternative treatment options beyond supportive therapies. While internal substitution is occurring between C5, C3, and emerging Factor B/D inhibitors, these therapies largely complement rather than replace each other within treatment protocols. The dominance of parenteral administration persists, although gradual adoption of subcutaneous and oral therapies is reshaping treatment pathways and enabling a shift toward decentralized care settings. Overall, the Japan PNH treatment market is defined by high-value therapies, limited patient volume, strong clinical dependence on targeted biologics, and innovation-driven competition focused on improving efficacy, convenience, and long-term disease management.
Action of Mechanism Insights
The complement inhibitors segment dominated the market, accounting for 91.95% of revenue in 2025. The market includes therapies targeting key components of the complement system, primarily C5 and C3, to reduce hemolysis and control disease symptoms. C5 inhibitors, such as eculizumab and ravulizumab, block C5 cleavage, improving outcomes like anemia and thrombosis, with growth driven by rising diagnosis rates, favorable orphan drug policies, and strong government support. C3 inhibitors act earlier in the pathway, addressing residual hemolysis in patients not fully responsive to existing treatments. Increasing demand for more effective and personalized therapies, coupled with pharmaceutical expansions and regulatory approvals, is further fueling adoption. These inhibitors are increasingly integrated into hospital and specialty clinic protocols. Collectively, these factors support sustained market growth for complement-targeted therapies in Japan.
The others segment is expected to grow at the fastest CAGR over the forecast period. The segment includes pharmacological treatment approaches such as androgen therapy and other residual drug-based interventions. Rising awareness among physicians about alternative treatment options for patients who are ineligible for or intolerant to complement inhibitors is driving adoption in this segment. The increasing number of diagnosed PNH cases with varying disease severities creates a need for tailored therapies, encouraging the use of these residual pharmacological approaches. Additionally, advancements in clinical research and the ongoing off-label use of these drugs offer further growth potential. Hospitals and specialty clinics are integrating these therapies into individualized care plans, ensuring broader access.
Route of Administration Insights
The parenteral segment in the Japan paroxysmal nocturnal hemoglobinuria treatment market held the largest revenue share of 96.13% in 2025. The Parenteral route of administration encompasses both Intravenous (IV) infusion and Subcutaneous (SC) injection therapies, each catering to different patient needs. IV infusion therapies, including complement inhibitors, are delivered directly into the bloodstream in clinical settings, offering rapid therapeutic action and predictable pharmacokinetics. Their use is supported by long-term real-world data and integration of digital health tools for outpatient monitoring, reducing hospital stays. Concurrently, SC injection therapies provide patient-centric options that can be self-administered or delivered in shorter visits, supported by nurse-led education and home-based treatment programs. Advancements in formulation technologies, such as lower-volume or longer-acting injections, enhance patient comfort and adherence. The combination of IV and SC approaches allows flexible treatment strategies, with SC therapies complementing IV regimens to meet diverse clinical and lifestyle needs.
The oral segment is expected to grow at the fastest CAGR during the forecast period, driven by the shift toward more convenient and patient-friendly treatment options. Oral therapies offer a non-invasive alternative to traditional parenteral treatments, reducing the burden associated with frequent hospital visits or infusion procedures. This segment is gaining traction as patients and healthcare providers prioritize ease of administration and improved quality of life. The growing acceptance of oral medications in chronic disease management is also contributing to its expansion. Additionally, continuous innovation in drug development is enabling the introduction of targeted oral therapies for rare hematologic conditions. As a result, the oral segment is expected to witness steady growth within the broader PNH treatment landscape in Japan.
End Use Insights
Hospitals & specialty clinics led the Japan paroxysmal nocturnal hemoglobinuria treatment market, with a 93.65% market share in 2025. The segment includes hospitals and specialty clinics that act as the main centers for the diagnosis, initiation of treatment, administration of therapies, monitoring, and continuous management of PNH. For market sizing purposes, it includes patients whose primary care is delivered in these healthcare settings, along with the revenue generated from these services in Japan. Additionally, the segment captures the cost of specialized procedures, infusion services, and follow-up consultations provided within these facilities. It also reflects the role of multidisciplinary care teams in improving patient outcomes and ensuring adherence to long-term treatment protocols. For instance, in January 2024, AstraZeneca announced that Japan’s Ministry of Health, Labour and Welfare approved Voydeya (danicopan) as an add-on therapy for Paroxysmal Nocturnal Hemoglobinuria.
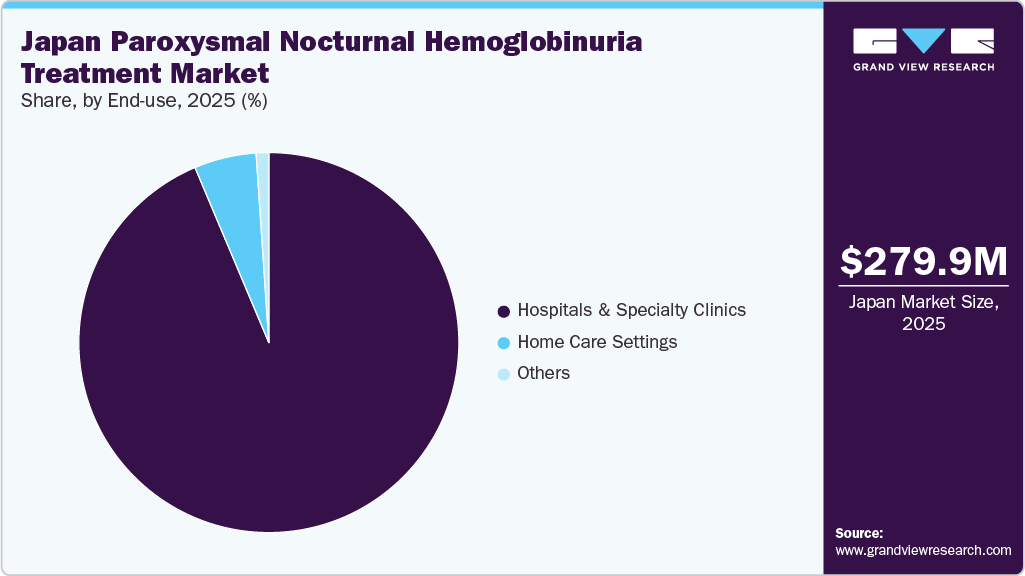
Home care settings in the Japan Paroxysmal Nocturnal Hemoglobinuria (PNH) treatment market are emerging as an important alternative to hospital-based care, particularly for patients requiring long-term therapy and routine monitoring. These settings enable the administration of selected therapies, follow-up care, and supportive services within the patient’s residence, improving convenience and reducing the need for frequent hospital visits. Growth is supported by Japan’s aging population and the increasing burden of rare chronic diseases, which are driving demand for decentralized care models. In addition, advancements in drug formulations that allow less frequent dosing or easier administration are facilitating the shift toward home-based care. The rising focus on patient-centric treatment approaches further supports this trend.
Key Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Company Insights
Some of the leading players operating in the Japan paroxysmal nocturnal hemoglobinuria treatment market include AstraZeneca, Novartis AG, Sanofi, and F. Hoffmann-La Roche Ltd. Companies focus on capturing the market by increasing their presence using various business initiatives, such as partnerships & collaborations with government. Moreover, these companies have well established action of mechanism portfolio, which helps capture major market share. BioCryst Pharmaceuticals, Johnson & Johnson, Pfizer Inc. are some of the emerging market participants in the Japan paroxysmal nocturnal hemoglobinuria treatment market. Developing & launching new and improved diagnostic tools that offer faster results, higher accuracy, and easier usability are observed as prevalent operating strategies by these companies. The players may face challenges in penetrating the market due to competition and regulatory hurdles.
Key Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Companies:
- AstraZeneca
- Apellis Pharmaceuticals
- BioCryst Pharmaceuticals
- Novartis AG
- F. Hoffmann-La Roche Ltd
- Amgen Inc.
- Regeneron Pharmaceuticals
- Sanofi
- Pfizer Inc.
- Johnson & Johnson
Recent Development
-
In February 2026, Sanofi completed the global acquisition of Dynavax, directly expanding Sanofi Japan’s local vaccine portfolio. The integration adds next-generation adult vaccines to their local market offerings for respiratory and infectious diseases.
-
In February 2026, Shionogi announced a basic policy for an absorption-type merger with Torii Pharmaceutical, following Shionogi's acquisition of the company as a wholly-owned subsidiary to centralize domestic rare disease and immunology commercialization in Japan.
-
In July 2025, Apellis strengthened its Japan ties through a USD 275 million royalty repurchase agreement with Sobi. Sobi handles the Japanese commercialization of Empaveli, which remains a key alternative for PNH patients refractory to C5 treatment.
-
In August 2024, Novartis AG launched Fabhalta (iptacopan) in Japan as the first oral monotherapy for adults with PNH. It targets Factor B to prevent red blood cell destruction without requiring regular intravenous infusions.
Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market Report Scope
Attribute
Details
Market size in 2026
USD 305.40 million
Revenue forecast in 2033
USD 455.53 million
Growth rate
CAGR 5.88% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD Million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Action of Mechanism, Route of Administration, End Use
Key companies profiled
AstraZeneca; Apellis Pharmaceuticals; BioCryst Pharmaceuticals; Novartis AG; F. Hoffmann-La Roche Ltd; Amgen Inc.; Regeneron Pharmaceuticals; Sanofi; Pfizer Inc.; Johnson & Johnson
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Japan Paroxysmal Nocturnal Hemoglobinuria Treatment Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the Japan paroxysmal nocturnal hemoglobinuria treatment market on the basis of action of mechanism, route of administration, and end use:
-
Action of Mechanism Outlook (Revenue, USD Million, 2021 - 2033)
-
Complement Inhibitors
-
C5 Inhibitors
-
C3 Inhibitors
-
-
Supportive Care
-
Blood Transfusions
-
Immunosuppressant drugs
-
Anticoagulants
-
Others
-
-
Others
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Parenteral
-
Intravenous (IV) Infusion
-
Subcutaneous (SC) Injection
-
-
Oral
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Specialty Clinics
-
Home Care Settings
-
Others
-
Frequently Asked Questions About This Report
b. The Japan paroxysmal nocturnal hemoglobinuria treatment market size was valued at USD 279.95 million in 2025 and is anticipated to reach USD 305.40 million in 2026.
b. The Japan paroxysmal nocturnal hemoglobinuria treatment market is expected to witness a compound annual growth rate of 5.88% from 2026 to 2033 to reach USD 455.53 million by 2033.
b. Based on Action of Mechanism, the complement inhibitors segment dominated the market with the largest revenue share of 91.95% in 2025. The market includes therapies targeting key components of the complement system, primarily C5 and C3, to reduce hemolysis and control disease symptoms.
b. Some of the key players in Japan paroxysmal nocturnal hemoglobinuria treatment market are AstraZeneca, Apellis Pharmaceuticals, BioCryst Pharmaceuticals, Novartis AG, F. Hoffmann-La Roche Ltd
b. The Japan paroxysmal nocturnal hemoglobinuria treatment market is driven by continuous advancements in complement inhibitor therapies, including the development of next-generation C3 and Factor B/D inhibitors that improve clinical outcomes and address unmet needs such as residual anemia..
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










