- Home
- »
- Animal Health
- »
-
Laboratory Animal Medicine Market Size Report, 2033GVR Report cover
![Laboratory Animal Medicine Market Size, Share & Trends Report]()
Laboratory Animal Medicine Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Diagnostics & Health Surveillance Products, Therapeutics & Preventive Medicines), By Animal Model (Rodents, Rabbits), By Application, By Customer Type, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-876-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Laboratory Animal Medicine Market Summary
The global laboratory animal medicine market size was estimated at USD 7.2 billion in 2025 and is projected to reach USD 11.5 billion by 2033, growing at a CAGR of 6.1% from 2026 to 2033. The market growth is driven by the intensification of precision medicine & humanized model demand, rising burden of chronic disease research, integration of AI and digital health monitoring, and global health security & zoonotic surveillance.
Key Market Trends & Insights
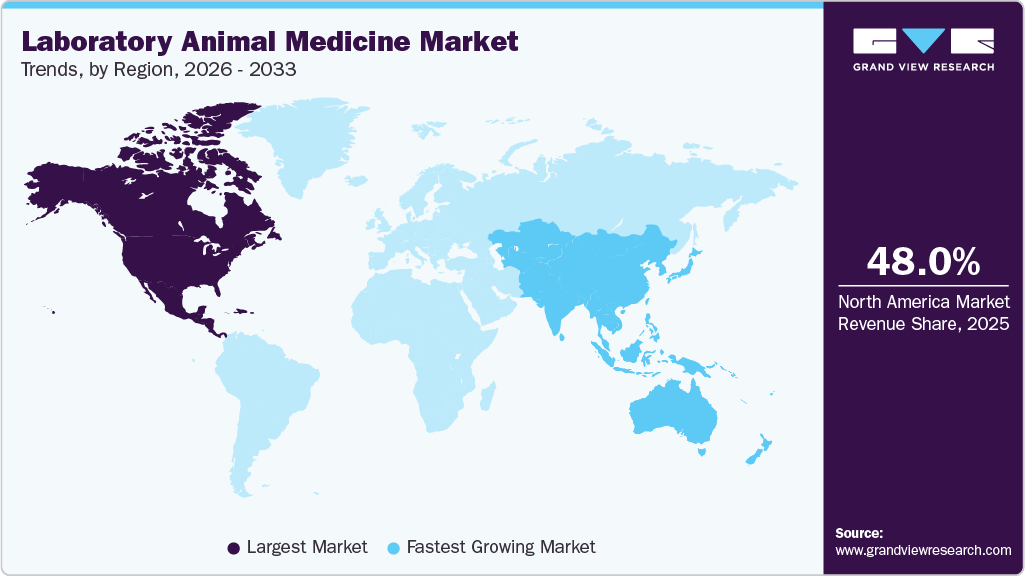
- North America dominated the global laboratory animal medicine market in 2025 with a share of more than 48%.
- The China laboratory animal medicine market is expected to grow the fastest globally at a rate of 10.9% over the forecast period.
- By product, the therapeutics & preventive medicines segment held the largest share of more than 26% in the market in 2025
- By animal model, the rodents segment is the largest as well as the fastest in the market in 2025.
- Based on the application, the chronic disease management & supportive care segment is expected to grow at the fastest rate of 6.5% over the forecast period.
Market Size & Forecast
- 2025 Market Size: USD 7.2 Billion
- 2033 Projected Market Size: USD 11.5 Billion
- CAGR (2026-2033): 6.1%
- North America: Largest market in 2025
- Asia Pacific: Fastest market from 2026 to 2033
The rising global burden of chronic conditions, specifically oncology, neurodegenerative diseases, and metabolic disorders, serves as a primary catalyst for the laboratory animal medicine sector. Unlike acute toxicity studies, chronic disease research necessitates longitudinal protocols that extend animal lifespans within a facility. This prolonged duration directly correlates with an increased requirement for sustained medical management and health monitoring. For instance, in oncology, the use of immunocompromised and humanized mouse models requires stringent diagnostics & health surveillance products. These animals are highly susceptible to opportunistic infections, driving the continuous use of serology & PCR test kits to maintain colony biosecurity and microbiology & parasitology assays to ensure pathogen-free status. Furthermore, the complexity of tumor progression studies mandates high-frequency clinical chemistry & hematology reagents/panels to monitor systemic physiological changes and organ function over time.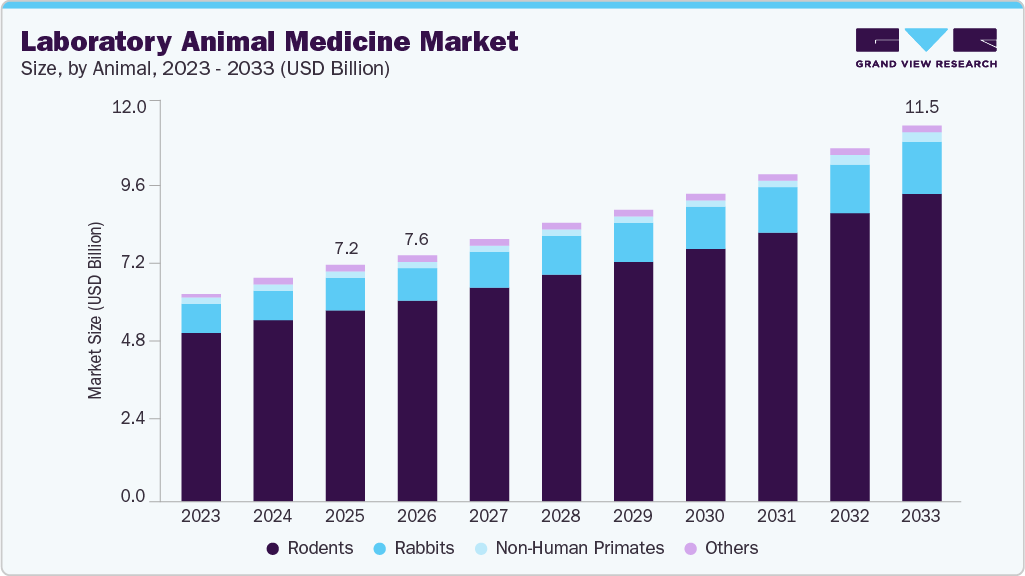
The demand for therapeutics & preventive medicines is similarly intensified by chronic modeling. Longitudinal studies for arthritis or cardiovascular diseases require long-term administration of analgesics & sedatives and anti-inflammatory & supportive care drugs to meet welfare standards and manage pain without compromising study data. Moreover, clinical care devices & equipment such as infusion & syringe pumps and ventilators & respiratory support systems are critical for the precise, metered delivery of experimental compounds and the maintenance of animal health during invasive procedures over several months.
Advanced chronic disease modeling also relies heavily on clinical imaging & diagnostic devices and animal patient monitoring devices to track disease markers non-invasively. As research institutions transition from short-term trials to multi-month chronic observations, the consumption of clinical & welfare consumables such as needles, syringes, catheters & vascular access sets, and bandages, wound care & surgical consumables increases. This shift from volume-based testing to high-value, long-term health management ensures a stable revenue market trajectory, as every additional day an animal remains in a study necessitates a corresponding unit of medical support, preventive care, and diagnostic oversight.
However, the accelerated adoption of New Approach Methodologies (NAMs), such as organ-on-a-chip and 3D bioprinting, introduces structural challenges for the laboratory animal medicine sector. As researchers pivot toward these high-fidelity alternatives, the absolute volume of animal cohorts used in toxicity and efficacy screening will decrease. This transition directly dampens the long-term demand for these products, as digital and cellular assays replace the biological need for systemic medical management and surgical support.
Market Concentration & Characteristics
The global laboratory animal medicine sector exhibits moderate concentration, dominated by a small cohort of multinational entities providing health products for models such as rodents, NHPs, and rabbits. Companies like Zoetis Inc., Merck Animal Health, and Boehringer Ingelheim leverage expansive distribution networks and M&A intensity to consolidate market share. Competitive influence remains concentrated among vendors offering comprehensive, validated hardware-software ecosystems and integrated health solutions that ensure regulatory compliance and data integrity.
Innovation intensity remains elevated globally, specifically regarding high-precision clinical imaging and diagnostic devices, automated animal patient monitoring, and AI-supported analytics. Vendors prioritize technologies that enhance research reproducibility while promoting animal welfare and minimizing stress. Product development increasingly focuses on comprehensive hardware-software ecosystems, incorporating digital sensors and automated data capture to support validated, application-specific health management workflows across research facilities.
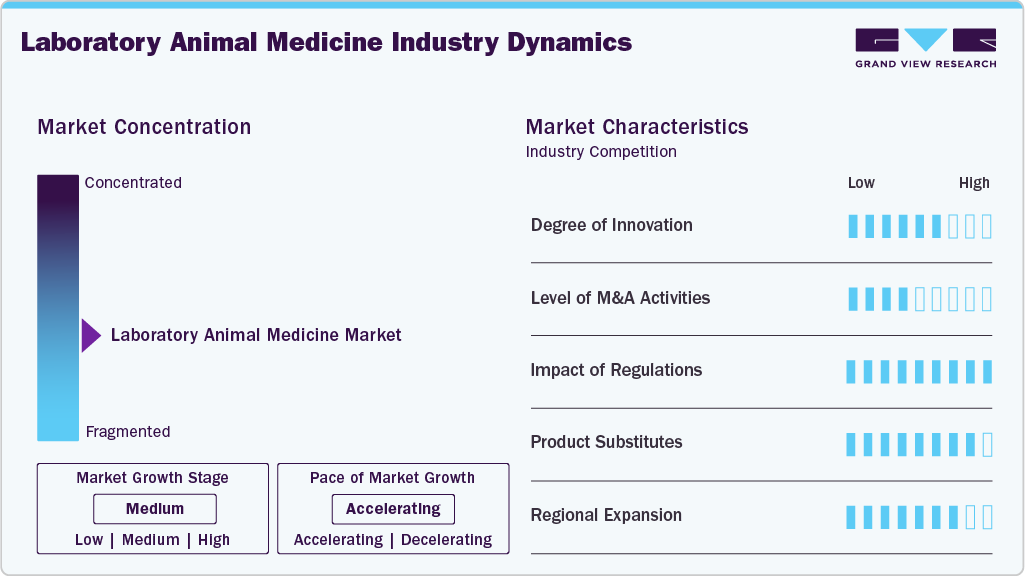
Mergers and acquisitions intensity remains moderate, characterized by strategic consolidation to enhance specialized capabilities and technical service infrastructure. Tier-1 players, such as Merck Animal Health and Zoetis Inc., actively pursue acquisitions of niche innovators in imaging, diagnostics, and aquaculture to broaden therapeutic depth and secure recurring revenue streams. This consolidation is particularly evident in high-value segments like molecular diagnostics and precision monitoring, where large-scale manufacturers aim to integrate proprietary technologies into comprehensive, validated product suites. This strategic movement gradually concentrates market influence among vendors capable of offering integrated healthcare ecosystems for institutional research facilities.
Stringent global regulatory frameworks, notably the FDA Modernization Act 2.0 and EU Directive 2010/63/EU, mandate the 3Rs-Replacement, Reduction, and Refinement. While these policies dampen long-term demand for high-volume consumables by lowering absolute animal counts in toxicity screening, they simultaneously intensify per-animal expenditure. Mandated refinement protocols necessitate advanced medical oversight, driving the adoption of specialized animal patient monitoring devices, precision analgesics & sedatives, and non-invasive clinical imaging & diagnostic devices to ensure humane endpoints and data integrity.
The sector faces increasing substitution from New Approach Methodologies (NAMs), including organ-on-a-chip, 3D bioprinting, and in-silico modeling. These technologies recapitulate human physiological responses more accurately than traditional biological assays, gradually displacing animal cohorts in early-stage efficacy and safety screening. This structural shift may dampen long-term demand for healthcare products in these animal models as research facilities transition toward digital and cellular alternatives in the near future.
North America continues to dominate in terms of animals used for research owing to sophisticated research infrastructure and a high concentration of biopharmaceutical headquarters. However, a significant geographic expansion pivot toward the Asia-Pacific (APAC) region is underway. Countries like China and India are experiencing the fastest growth as global pharmaceutical companies and CROs relocate preclinical pipelines to leverage cost-efficiencies and expand laboratory networks, making APAC the decisive frontier for future market expansion.
Product Insights
The therapeutics & preventive medicines segment accounted for the highest market share of more than 26% in 2025 and is expected to grow at the fastest rate over the forecast period. This dominance is owed to broad therapeutic necessity across all major animal model categories, where routine vaccine administration, anti-infective protocols, and antiparasitic regimens constitute non-discretionary expenditure within GLP-compliant facilities. Stringent regulatory mandates governing colony health documentation, perioperative analgesia, and anesthesia administration further drive sustained procurement volumes. In addition, rising NHP utilization in biopharmaceutical preclinical pipelines has intensified demand for species-specific therapeutics, while expanding GLP-certified laboratory networks across North America and the Asia Pacific are standardizing preventive care protocols at higher intervention frequencies.
Diagnostics & health surveillance products is expected to be the second-largest segment due to its foundational role in maintaining colony health integrity and regulatory compliance across research facilities. Mandatory sentinel testing, serological screening, and PCR-based pathogen surveillance programs generate consistent, high-frequency procurement of diagnostic kits and reagents. Expansion of GLP-certified laboratories and public diagnostic network infrastructure is elevating baseline testing expectations, driving broader adoption of clinical chemistry and hematology panels. Increasing biosecurity requirements within NHP and rodent colonies, particularly across pharmaceutical CRO networks, further sustain demand for confirmatory and routine microbiology and parasitology assays.
Animal Model Insights
Rodents held the highest market share in 2025 and are expected to maintain the fastest growth from 2026 to 2033. This dominance is driven by rodents' established position as the default animal model across preclinical toxicology, oncology, immunology, and neuroscience research globally. Their short reproductive cycles, genetic tractability, and availability of validated transgenic and knockout strains make them operationally indispensable across pharmaceutical, biopharmaceutical, and academic institutions. High colony densities inherent to rodent facilities necessitate intensive health surveillance, routine vaccination, and antiparasitic management at scale. Expanding preclinical CRO capacity across North America, China, and India is further amplifying per-facility rodent healthcare expenditure.
The other animals segment, consisting of guinea pigs, hamsters, ferrets, pigs, dogs, sheep, and fish, etc., is expected to register the second-fastest growth rate from 2026 to 2033 due to their expanding utility across specialized preclinical applications where rodent models demonstrate translational limitations. Guinea pigs and ferrets serve respiratory and infectious disease research; swine and canine models address cardiovascular and surgical validation requirements; aquatic models support toxicology and developmental biology screening. Regulatory acceptance of these species in specific submission pathways, combined with growing exotic and aquatic model adoption across Asian and European research institutions, sustains diversified healthcare product demand.
Application Insights
The disease surveillance & colony health monitoring segment dominated the market with a share of around 18% in 2025, due to its position as the primary, non-negotiable operational requirement across all laboratory animal facility types globally. Continuous pathogen monitoring, sentinel animal programs, and routine serological screening constitute mandatory compliance activities under IACUC, GLP, and equivalent regulatory frameworks. Rising facility scale, multi-site CRO network expansion, and stricter biosecurity documentation expectations collectively intensify testing frequency and diagnostic reagent consumption per animal cohort, sustaining segment leadership independent of broader research activity fluctuations.
The chronic disease management & supportive care segment is anticipated to grow at the fastest CAGR of more than 6.5% over the coming years. This growth is driven by increasing utilization of longitudinal animal models in metabolic disorder, oncology, and neurodegeneration research, where extended study durations demand sustained pharmaceutical intervention and clinical monitoring. GLP compliance requirements mandate documented pain management, anti-inflammatory administration, and supportive care protocols throughout prolonged in vivo studies. Expanding biopharmaceutical preclinical pipelines globally, particularly within CRO networks in North America and the Asia Pacific, are generating higher per-animal therapeutic consumption across multi-month study timelines.
Customer Insights
The pharmaceutical & biopharmaceutical companies segment held the highest share of the market in 2025, driven by their position as the largest institutional operators of in-house preclinical facilities and primary sponsors of contract research globally. Regulatory submission requirements mandate extensive in vivo safety, efficacy, and toxicology data, generating non-discretionary, high-volume healthcare product procurement. Expanding biologics and advanced therapy pipelines have intensified animal study volumes, driving sustained demand for diagnostics, therapeutics, anesthesia, and perioperative consumables within pharmaceutical research operations.
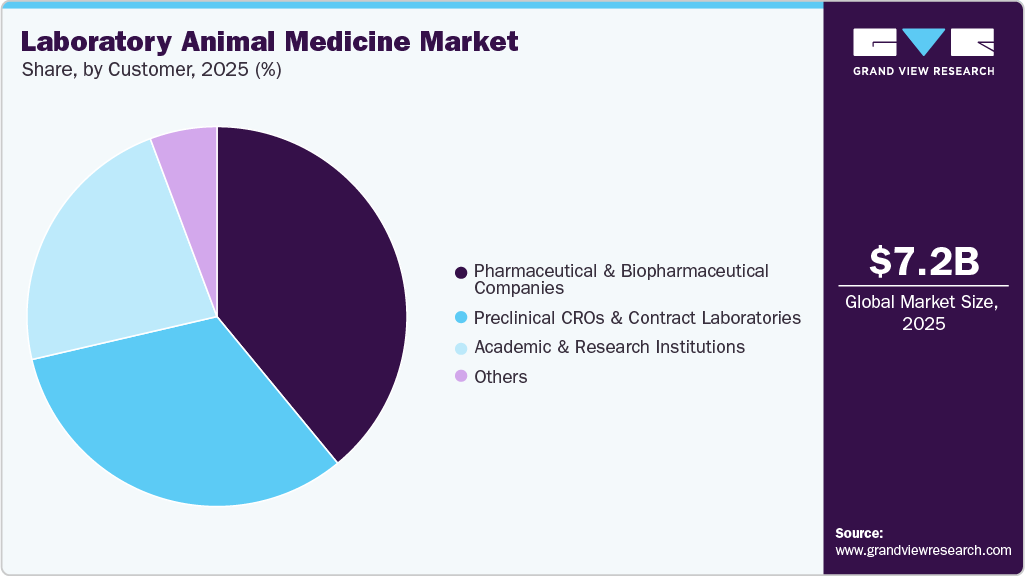
The preclinical CROs & contract laboratories segment is estimated to grow at the fastest CAGR in the forecast years. This growth is driven by the accelerating outsourcing of preclinical research by pharmaceutical and biopharmaceutical sponsors seeking operational flexibility and regulatory expertise. CRO network expansion across lower-cost, high-capability markets in China, India, and Eastern Europe is increasing aggregate animal study volumes. Multi-client facility operations necessitate rigorous, continuous health surveillance and standardized therapeutic protocols, generating proportionally higher healthcare product consumption relative to single-sponsor in-house research operations.
Regional Insights
North America dominated the global laboratory animal medicine market in 2025 with a share of more than 48%. The growth is driven by the region's dense concentration of pharmaceutical and biopharmaceutical manufacturers, top-tier academic research institutions, and established preclinical CRO networks, primarily across the U.S. Stringent USDA, IACUC, and FDA regulatory frameworks mandate comprehensive veterinary oversight, documented preventive care, and validated diagnostic protocols, elevating healthcare intensity per animal. Canada's expanding GLP-certified laboratory infrastructure and Mexico's growing contract research capacity further contribute regionally. Sustained NIH funding allocations and large-scale private R&D investments continuously support high-volume, compliance-driven animal healthcare procurement.
U.S. Laboratory Animal Medicine Market Trends
The laboratory animal medicine market in the U.S. dominated North America in 2025, holding the largest revenue share of around 71.5%. Rising institutional prioritization of laboratory animal welfare standards is generating measurable demand for professional veterinary oversight, structured husbandry programs, advanced diagnostics, and comprehensive care protocols. Regulatory expectations governing documented welfare outcomes are elevating healthcare intensity across facility types globally. Concurrently, veterinary teams are increasingly dependent on rapid, accurate diagnostic testing, spanning hematology, clinical chemistry, microbiology, cytology, and histology, to enable early pathogen detection, recovery management, and continuous welfare assessment. This diagnostic precision requirement is driving capital investment toward centralized, high-throughput laboratory infrastructure capable of delivering fast, actionable results across multi-species research populations.
Europe Laboratory Animal Medicine Market Trends
The laboratory animal medicine market in Europe is estimated to be the second-largest region in this market. The growth is driven by Europe's mature regulatory architecture governing laboratory animal research, anchored by Directive 2010/63/EU, which mandates rigorous welfare standards, documented veterinary oversight, and structured health monitoring across all member states. The ECLAM actively advances professional competency standards, elevating veterinary care intensity across institutional facilities. Simultaneously, the region's robust pharmaceutical and biotechnology manufacturing base sustains high preclinical research volumes. Public funding through Horizon Europe and ECVAM-led 3Rs initiatives is paradoxically sustaining demand for higher-precision, welfare-supportive healthcare products within remaining animal-based research workflows, particularly across Germany, France, and the UK.
The UK laboratory animal medicine market dominated Europe in 2025, driven by its position as Europe's largest preclinical research hub, housing a dense concentration of pharmaceutical manufacturers, academic research institutions, and contract research organizations. The Home Office licensing framework under the Animals (Scientific Procedures) Act 1986 mandates comprehensive named veterinary surgeon oversight, structured health surveillance, and documented welfare protocols, elevating per-animal healthcare intensity. The UK government's published roadmap, accelerating the adoption of alternative methods, is simultaneously driving investment toward higher-precision diagnostics and welfare-supportive therapeutics within remaining animal-based research programs.
The laboratory animal medicine market in Germany is another major market in Europe, driven by its position as one of Europe's largest pharmaceutical and biotechnology manufacturing bases, sustaining substantial in-house preclinical research activity across major corporations and public research institutions. Germany's rigorous implementation of EU directives through the Animal Welfare Act mandates structured veterinary oversight, documented health monitoring, and standardized perioperative care protocols. The country's extensive network of Helmholtz and Leibniz research centers, alongside leading academic institutions, generates consistent, high-volume procurement of diagnostics, therapeutics, and clinical care consumables across diversified animal model populations.
Asia Pacific Laboratory Animal Medicine Market Trends
The laboratory animal medicine market in the Asia Pacific is estimated to be the fastest-growing region. The growth is driven by the rapid expansion of pharmaceutical and biopharmaceutical manufacturing infrastructure across China, India, South Korea, and Japan, generating proportionally higher preclinical research volumes and in-house animal study requirements. China's biotech boom has intensified NHP utilization, driving specialized therapeutic and diagnostic procurement, while simultaneously prompting regulatory authorities to formalize laboratory animal welfare guidelines. India's growing CRO sector and progressive regulatory engagement with non-animal alternatives are paradoxically elevating healthcare standards within existing animal research operations. South Korea's structured investment in advanced preclinical capabilities and Japan's established GLP-compliant research infrastructure further sustain regional demand for diagnostics, therapeutics, and clinical monitoring solutions across diversified animal model populations.
China laboratory animal medicine market dominates the Asia Pacific in terms of share and growth rate, driven by its position as the world's fastest-expanding biotech and pharmaceutical research base, with surging domestic drug development pipelines generating substantial in-house and CRO-based preclinical study volumes. Escalating NHP demand, reflected in sharply rising prices for cynomolgus macaques, signals deepening utilization of specialized animal models requiring intensive veterinary oversight and species-specific therapeutics. Regulatory formalization of laboratory animal welfare standards and GLP certification requirements is elevating healthcare intensity per animal. Government-backed biologics manufacturing expansions and growing CDMO capacity are further sustaining high-volume, compliance-driven animal healthcare procurement across institutional and commercial research facilities.
The laboratory animal medicine market in India is the second-fastest-growing country in the region, driven by its rapidly expanding CRO and contract laboratory sector, attracting pharmaceutical outsourcing from global sponsors. Regulatory engagement by CDSCO toward reducing animal testing dependence is simultaneously elevating welfare and diagnostic standards within active research operations. Growing academic and institutional preclinical infrastructure, combined with LASA India-led professional veterinary competency initiatives, is increasing structured healthcare product adoption across rodent and NHP model populations.
Latin America Laboratory Animal Medicine Market Trends
The laboratory animal medicine market in Latin America is growing at a lucrative rate, driven by expanding pharmaceutical manufacturing activity across Brazil and Argentina, where regulatory modernization and bilateral medical approval frameworks are accelerating preclinical research volumes. Brazil's progressive animal welfare legislative reforms are elevating institutional veterinary oversight requirements and diagnostic protocol standardization. Growing academic research infrastructure across Argentina and Colombia, combined with increasing CRO establishment in the region, is generating rising procurement demand for therapeutics, diagnostics, and clinical care consumables.
Brazil laboratory animal medicine market dominates the region in terms of share and growth rate, driven by its position as Latin America's largest pharmaceutical and biotechnology manufacturing base, sustaining substantial preclinical research activity across institutional and commercial facilities. CONCEA-governed animal welfare regulations mandate structured veterinary oversight and documented health protocols, elevating per-animal healthcare intensity. Growing CRO establishment, expanding academic research infrastructure, and Brazil's recent alliance with Mexico to streamline medical approvals are collectively amplifying preclinical study volumes and associated animal healthcare product procurement.
Middle East & Africa Laboratory Animal Medicine Market Trends
The laboratory animal medicine market in the MEA region is growing at a lucrative rate, owing to expanding biomedical research infrastructure across Gulf Cooperation Council states, particularly Saudi Arabia and the UAE, where government-backed healthcare diversification strategies are driving investment in preclinical and veterinary research capabilities. Saudi Arabia's localization initiatives for veterinary vaccine and drug production are generating domestic research activity. Africa's growing academic and public health research networks, combined with increasing biosecurity requirements across regional laboratory animal facilities, are sustaining rising diagnostic and therapeutic procurement demand.
South Africa laboratory animal medicine market dominates the regional market in terms of share. The country is the region’s most developed biomedical research hub, housing established academic institutions, public health research organizations, and GLP-compliant preclinical facilities. Robust rabbit and NHP model utilization across immunology, infectious disease, and radiation countermeasure research generates consistent therapeutic and diagnostic procurement. Structured animal welfare governance frameworks and veterinary oversight requirements elevate per-animal healthcare intensity, sustaining South Africa's dominant procurement share within the broader MEA market.
The laboratory animal medicine market in Kuwait is growing steadily because of expanding government investment in biomedical and pharmaceutical research infrastructure, aligned with Kuwait's broader healthcare sector diversification agenda. Rising institutional capacity across university-affiliated research centers and public health laboratories is generating increasing demand for structured veterinary oversight and diagnostic protocols. Growing awareness of international laboratory animal welfare standards among Kuwaiti research institutions is progressively elevating procurement of therapeutics, health surveillance products, and clinical care consumables.
Key Laboratory Animal Medicine Company Insights
The global market exhibits moderate-to-high consolidation, with a few large multinational manufacturers controlling core pharmaceutical, diagnostic, and anesthesia supply channels. Competitive differentiation centers on validated workflows, regulatory compliance credentials, and integrated product portfolios spanning therapeutics, diagnostics, and clinical devices, while specialized niche firms maintain defensible positions in imaging, monitoring, and species-specific solutions.
Key Laboratory Animal Medicine Companies:
The following key companies have been profiled for this study on the laboratory animal medicine market.
- Zoetis Services LLC
- Boehringer Ingelheim International GmbH
- Merck & Co., Inc.
- Dechra
- Elanco Animal Health
- Kent Scientific Corporation
- Harvard Apparatus
- Starr
- QIAGEN
- Neogen Corporation
- Mediso Ltd.
- IITCINC
- Thermo Fisher Scientific Inc.
- Plas-Labs, Inc.
- TSE Systems
- CH Technologies (USA), Inc.
Recent Developments
-
In February 2026, Qiagen introduced the QIAsprint Connect system to automate DNA and RNA sample preparation in laboratories. This technology supports biomedical and preclinical research that often uses laboratory animals such as mice, rats, and other model organisms.
-
In November 2025, according to a Bloomberg article, China is expanding large-animal experiments, including gene-edited pigs and monkeys, to accelerate drug discovery and strengthen its position as a global biotechnology leader. These practices face fewer regulatory barriers than in the U.S. and Europe, which allows faster research progress.
Laboratory Animal Medicine Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 7.6 billion
Revenue forecast in 2033
USD 11.5 billion
Growth rate
CAGR of 6.1% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, animal, testing category, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Zoetis Services LLC; Boehringer Ingelheim International GmbH; Merck & Co., Inc.; Dechra; Elanco Animal Health; Kent Scientific Corporation; Harvard Apparatus; Starr; QIAGEN; Neogen Corporation; Mediso Ltd.; IITCINC; Thermo Fisher Scientific Inc.; Plas-Labs, Inc.; TSE Systems; CH Technologies, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Laboratory Animal Medicine Market Report Segmentation
This report forecasts revenue growth at the regional and country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global laboratory animal medicine market based on product, animal model, application, customer, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Diagnostics & Health Surveillance Products
-
Serology & PCR Test Kits
-
Microbiology & Parasitology Assays
-
Clinical Chemistry & Hematology Reagents / Panels
-
Others
-
-
Therapeutics & Preventive Medicines
-
Vaccines
-
Anti-infectives (Antibiotics, Antivirals, Antifungals)
-
Antiparasitics (Endo- & Ecto-parasite Control)
-
Analgesics & Sedatives
-
Anesthetics
-
Anti-inflammatory & Supportive Care Drugs
-
Others
-
-
Clinical Care Devices & Equipment
-
Animal Patient Monitoring Devices
-
Anesthesia Machines & Vaporizers
-
Ventilators & Respiratory Support Systems
-
Infusion & Syringe Pumps
-
Clinical Imaging & Diagnostic Devices
-
Others
-
-
Clinical & Welfare Consumables
-
Needles, Syringes, Catheters & Vascular Access Sets
-
Bandages, Wound Care & Surgical Consumables
-
Disinfectants, Detergents & Sanitation Products
-
Personal Protective Equipment (PPE) & Barrier Consumables
-
Others
-
-
Others
-
-
Animal Model Outlook (Revenue, USD Million, 2021 - 2033)
-
Rodents
-
Rabbits
-
Non-Human Primates (NHPs)
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Disease Surveillance & Colony Health Monitoring
-
Preventive Care & Biosecurity
-
Perioperative Care (Anesthesia, Analgesia, Surgical Support)
-
Chronic Disease Management & Supportive Care
-
Welfare & Behavioral Assessment
-
End-of-Life & Euthanasia Management
-
Training, Education & Compliance Support
-
Others
-
-
Customer Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Preclinical CROs & Contract Laboratories
-
Academic & Research Institutions
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global laboratory animal medicine market size was estimated at USD 7.2 billion in 2025 and is expected to reach USD 7.6 billion in 2026.
b. The global laboratory animal medicine market is expected to grow at a compound annual growth rate of 6.1% from 2026 to 2033 to reach USD 11.5 billion by 2033.
b. On the basis of animal model, rodents held the highest market share in 2025 and are expected maintain the fastest growth over 2026-2033. This dominance is driven by rodents' established position as the default animal model across preclinical toxicology, oncology, immunology, and neuroscience research globally.
b. Some key players operating in the laboratory animal medicine market include Zoetis Services LLC; Boehringer Ingelheim International GmbH; Merck & Co., Inc.; Dechra; Elanco Animal Health; Kent Scientific Corporation; Harvard Apparatus; Starr; QIAGEN; Neogen Corporation; Mediso Ltd.; IITCINC; Thermo Fisher Scientific Inc.; Plas-Labs, Inc.; TSE Systems; and CH Technologies, Inc.
b. Key factors that are driving the market growth include intensification of precision medicine & humanized model demand, rising burden of chronic disease research, integration of ai and digital health monitoring, and global health security & zoonotic surveillance.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










